Abstract
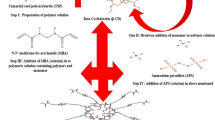
Psyllium has been explored for various food and pharmaceutical applications. Recently, its therapeutic action against cancer has been reported. Hence, herein in this research it has been modified to develop hydrogels through graft copolymerization reaction for use as sustained drug delivery carriers for anti-cancer drug “5-fluorouracil” (FU) to enhance the therapeutic potential. Synthesis of hydrogels was carried out by grafting of poly(2-hydroxypropyl methacrylate) and poly(acrylamide) onto psyllium in the presence of crosslinker. Polymers were characterized by cryo-SEM, AFM, XRD, 13C-NMR, and TGA techniques, beside evaluation of swelling, drug release, blood compatibility, mucoadhesion, antioxidant properties and gel strength of the hydrogels. The porous network structure with rough surface was observed in cryo-SEM and AFM images of the samples. Release of FU from psy-cl-poly(HPMA-co-AAm) polymer occurred with non-Fickian diffusion in sustained manner. These hydrogels were mucoadhesive (work of adhesion = 8.45 ± 0.32 N mm), non-haemolytic (haemolytic index = 1.05 ± 0.07%), and antioxidant (with 19.12 ± 0.14% inhibition in DPPH assay) in nature, therefore they could be used as slow drug delivery carriers for ailments associated GIT system.







Similar content being viewed by others
REFERENCES
L. Tong, C. Ahn, E. Symanski, D. Lai, and X. L. Du, Ann. Oncol. 25, 1234 (2014).
R. F. Donnelly, P. A. McCarron, M. M. Tunney, and A. D. Woolfson, J. Photochem. Photobiol., B 86, 59 (2007).
S. Singh, S. Jain, M. S. Muthu, S. Tiwari, and R. Tilak, AAPS PharmSciTech 9, 660 (2008).
S. R. Abulateefeh, S. G. Spain, K. J. Thurecht, J. W. Aylott, W. Chan, M. C. Garnett, and C. Alexander, Biomater. Sci. 1, 434 (2013).
T. Nagai and Y. Machida, Adv. Drug Delivery Rev. 11, 179 (1993).
Y. Sudhakar, K. Kuotsu, and A. K. Bandyopadhyay, J. Controlled Release 114, 15 (2006).
N. S. Madhav, A. K. Shakya, P. Shakya, and K. Singh, J. Controlled Release 140, 2 (2009).
H. Tu, Y. Qu, X. Hu, Y. Yin, H. Zheng, P. Xu, and F. Xiong, Carbohydr. Polym. 82, 440 (2010).
K. M. Rao, S. Nagappan, D. J. Seo, and C. S. Ha, Appl. Clay Sci. 97, 33 (2014).
M. Shahid, S.A. Bukhari, Y. Gul, H. Munir, F. Anjum, M. Zuber, T. Jamil, and K. M. Zia, Int. J. Biol. Macromol. 62, 172 (2013).
Y. S. R. Krishnaiah, V. Satyanarayana, B. D. Kumar, and R. S. Karthikeyan, Eur. J. Pharm. Sci. 16, 185 (2002).
I. Kavianinia, P. G. Plieger, N. J. Cave, G. Gopakumar, M. Dunowska, N. G. Kandile, and D. R. K. Harding, Int. J. Biol. Macromol. 85, 539 (2016).
M. Prabaharan, Int. J. Biol. Macromol. 49, 117 (2011).
Y. Yin, Y. Yang, and H. Xu, J. Appl. Polym. Sci. 83, 2835 (2002).
B. B. Mandal, S. Kapoor, and S. C. Kundu, Biomaterials 30, 2826 (2009).
B. Mukherjee, A. Samanta and S. C. Dinda, Trends Appl. Sci. Res. 1, 309 (2006).
V. U. R. M. Sowdhamini, B. Sirisha, and A. Pratyusha, Int. J. Pharm. Invest. 2, 35 (2015).
F. Fernandez-Banares, J. Hinojosa, J. L. Sanchez-Lombrana, E. Navarro, J. F. Martinez-Salmeron, A. Garcia-Puges, F. Gonzalez-Huix, J. Riera, V. Gonzalez-Lara, F. Dominguez-Abascal, J. J. Gine, J. Moles, F. Gomollon, and M. A. Gassull, Am. J. Gastroenterol. 94, 427 (1999).
M. R. Clausen, H. Bonnen, and P. B. Mortensen, Gut 32, 923 (1991).
J. A. Market and M. H. Fischer, Proc. Nutr. Soc. 62, 207 (2003).
J. F. Kennedy, J. S. Sandhu, and D. A. T. Southgate, Carbohydr. Res. 75, 265 (1979).
B. Singh, Int. J. Pharm. 334, 1 (2007).
N. Zhang, Y. Yin, S. J. Xu, and W. S. Chen, Molecules 13, 1551 (2008).
N. A. Peppas and N. K. Mongia, Eur. J. Pharm. Biopharm. 43, 51 (1997).
G. Chang, Y. Chen, Y. Li, S. Li, F. Huang, Y. Shen, and A. Xie, Carbohydr. Polym. 122, 336 (2015).
S. Tanpichai and K. Oksman, Composites, Part A 88, 226 (2016).
P. L. Ritger and N. A. Peppas, J. Controlled Release 5, 23 (1987).
P. L. Ritger and N. A. Peppas, J. Controlled Release 5, 37 (1987).
M. H. Shoaib, J. Tazen, H. A. Merchant, and R. I. Yousuf, Pak. J. Pharm. Sci. 19, 119 (2006).
A. G. Sullad, L. S. Manjeshwar, and T. M. Aminabhavi, Ind. Eng. Chem. Res. 49, 7323 (2010).
K. S. C. R. D. Santos, J. F. J. Coelho, P. Ferreira, I. Pinto, S. G. Lorenzetti, E. I. Ferreira, O. Z. Higa, and M. H. Gil, Int. J. Pharm. 310, 37 (2006).
American Society for Testing Materials, ASTM F 756-00 (Philadelphia, 2000).
G. Gorczyca, R. Tylingo, P. Szweda, E. Augustin, M. Sadowska, and S. Milewski, Carbohydr. Polym. 102, 901 (2014).
M. Curcio, F. Puoci, F. Lemma, O. I. Parisi, G. Cirillo, U. G. Spizzirri, and N. Picci, J. Agric. Food Chem. 57, 5933 (2009).
N. Thirawong, J. Nunthanid, S. Puttipipatkhachorn, and P. Srimornsak, Eur. J. Pharm. Biopharm. 67, 132 (2007).
N. Buchtova, A. D’orlando, P. Judeinstein, O. Chauvet, P. Weiss, and J. Le Bideau, Carbohydr. Polym. 202, 404 (2018).
M. Chang, X. Liu, L. Meng, X. Wang, and J. Ren, Pharmaceutics 10, 261 (2018).
M. Munz, Appl. Surf. Sci. 279, 300 (2013).
D. Das, R. Das, P. Ghosh, S. Dhara, A. B. Panda, and S. Pal, RSC Adv. 3, 25340 (2013).
E. Andrzejewska-Golec, S. Ofterdinger-Daegel, I. Calis, and L. Swiatek, Plant Syst. Evol. 185, 85 (1993).
H. Ramadan, T. Coradin, S. Masse, and H. El-Rassy, Silicon 3, 63 (2011).
X. M. Liu, L. D. Quan, J. Tian, Y. Alnouti, K. Fu, G. M. Thiele, and D. Wang, Pharm. Res. 25, 2910 (2008).
I. V. Averianov, V. A. Korzhikov-Vlakh, Y. E. Moskalenko, V. E. Smirnova, and T. B. Tennikova, Mendeleev Commun. 27, 574 (2017).
E. E. Khozemy, S. M. Nasef, and G. A. Mahmoud, Adv. Polym. Technol. 37, 3252 (2018).
A. K. Bajpai and A. Giri, Carbohydr. Polym. 53, 271 (2003).
A. G. Ibrahim, A. Z. Sayed, H. A. El-Wahab, and M. M. Sayah, Am. J. Polym. Sci. Technol. 5, 55 (2019).
D. S. Seeli, S. Dhivya, N. Selvamurugan, and M. Prabaharan, Int. J. Biol. Macromol. 91, 45 (2016).
T. S. Anirudhan and A. M. Mohan, RSC Adv. 4, 12109 (2014).
R. K. Mishra, K. Ramasamy, and A. B. A. Majeed. J. Appl. Polym. Sci. 126, 98 (2012).
Y. Nakamura, N. Yoshikawa, I. Hiroki, K. Sato, K. Ohtsuki, C. C. Chang, B. L. Upham, and J. E. Trosko, Nutr. Cancer 51, 218 (2005).
Y. Huang, H. Yu, and C. Xiao, Carbohydr. Polym. 69, 774 (2007).
N. Baït, C. Derail, A. Benaboura, and B. Grasslc, Int. J. Adhes. Adhes. 96 102449 (2020).
L. I. Xinming and C. U. I. Yingde, Chin. J. Chem. Eng. 16, 640 (2008).
M. U. Ashraf, M. A. Hussain, S. Bashir, M. T. Haseeb, and Z. Hussain, J. Drug Delivery Sci. Technol. 45, 455 (2018).
E. S. Dragan, D. F. A. Loghin, A. I. Cocarta, and M. Doroftei, React. Funct. Polym. 105, 66 (2016).
M. Fischer, C. Sperling, and C. Werner, J. Mater. Sci.: Mater. Med. 21, 931 (2010).
M. Micic and E. Suljovrujic, Eur. Polym. J. 49, 3223 (2013).
K. I. Block, A. C. Koch, M. N. Mead, P. K. Tothy, R.A. Newman, and C. Gyllenhaal, Int. J. Cancer 123, 1227 (2008).
T. Uchida, S. Sakakibara, Y. Toida, N. Nagareya, N. Yasuda, and K. Matsuyama, Pharm. Pharmacol. Commun. 5, 523(1999).
I. Bravo-Osuna, C. Vauthier, A. Farabollini, G. F. Palmieri, and G. Ponchel, Biomaterials 28, 2233 (2007).
N. A. Peppas, K. B. Keys, M. Torres-Lugo, and A. M. Lowman, J. Controlled Release 62, 81 (1999).
L. L. Chaves, A. Silveri, A. C. C. Vieira, D. Ferreira, M. C. Cristiano, D. Paolino, L. D. Marzio, S. C. Lima, S. Reis, B. Sarmento, and C. Celia, Int. J. Biol. Macromol. 133, 1268 (2019).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Baljit Singh, Kumar, S., Rajneesh et al. Synthesis and Characterization of Psyllium Polysaccharide–Poly(2-hydroxypropyl methacrylate)−Poly(acrylamide) Hydrogels for Use in Sustained Drug Delivery Applications. Polym. Sci. Ser. B 62, 685–696 (2020). https://doi.org/10.1134/S1560090420330064
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1560090420330064