Abstract
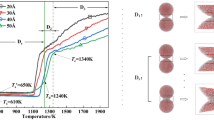
We employed ReaxFF molecular dynamics (MD) to simulate the physicochemical properties of HMX nanoparticle at low temperatures. As the temperature increases, its physical and chemical properties change accompanying some transition, which is not reported by any previous studies. The Poisson Scoring Model was used to count phase transition. It is found that the number of the β- to δ-HMX conformational transition at 458 K is higher than that at other temperatures. The types of decomposition products of the HMX nanoparticle at low temperatures are the same as those by pervious high-temperature experiments and theoretical simulations. The decomposition of the HMX nanoparticle is triggered by the C–H bond dissociation. Its decomposition includes two steps. The one is the decay of the HMX molecules, which is called the direct influence of the solid phase; the other one is the second reaction of small intermediate in less condensed phase. Moreover, the temperature will deeply affect the duration of each stage.










Similar content being viewed by others
References
Asay B, Henson B, Smilowitz L, Dickson PM (2003) On the difference in impact sensitivity of beta and delta HMX. J Energ Mater 21:223–235
Behrens R, Bulusu S (1995) Thermal decomposition of hmx: low temperature reaction kinetics and their use for assessing response in abnormal thermal environments and implications for long-term aging. MRS Proc 418:119
Brill TB, Brush PJ, Kinloch PGA (1992) Energetic materials: condensed phase chemistry of explosives and propellants at high temperature: hmx, rdx and bamo [and discussion]. Philos Trans Phys Sci Eng 339:377–385
Burnham AK, Weese RK, Weeks BL (2004) A distributed activation energy model of thermodynamically inhibited nucleation and growth reactions and its application to the β−δ phase transition of HMX. J Phys Chem B 108:19432–19441
Cady HH, Smith LC, Laboratory LAS, Commission USAE (1962) Studies on the Polymorphs of HMX. Los Alamos Scientific Laboratory of the University of California, California
Cady HH, Larson AC, Cromer DT (1963) The crystal structure of α-HMX and a refinement of the structure of β-HMX. Acta Cryst 16:617–623
Chen L, Ma X, Huang YM, Wu JY, Chang XM (2011a) Multi-point temperature measuring cook-off test and numerical simulation of explosive. Acta Armamentarii 32:1230–1236
Chen J, Long Y, Liu Y, Nie F, Sun J (2011b) The first-principle study on the equation of state of HMX under high pressure. Sci China Phys Mech Astron 54:831–835
Cobbledick RE, Small RW (1974) The crystal structure of the δ-form of 1,3,5,7-tetranitro-1,3,5,7-tetraazacyclooctane (δ-HMX). Acta Cryst 30:1918–1922
Cui HL, Ji GF, Chen XR, Zhu WH, Zhao F, Wen Y, Wei DQ (2010) First-principles study of high-pressure behavior of solid β-HMX. J Phys Chem A 114:1082–1092
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Zakrzewski VG, Montgomery JA, Stratmann RE, Burant JC, Dapprich S, Millam JM, Daniels AD, Kudin KN, Strain MC, Farkas O, Tomasi J, Barone V, Cossi M, Cammi R, Mennucci B, Pomelli C, Adamo C, Clifford S, Ochterski J, Petersson GA, Ayala PY, Cui Q, Morokuma K, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Cioslowski J, Ortiz JV, Baboul AG, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Gomperts R, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Gonzalez C, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Andres JL, Gonzalez C, Head-Gordon M, Replogle ES, Pople JA (2009) Gaussian 09. Gaussian, Inc., Wallingford
Fukui K (1981) The path of chemical reactions - the IRC approach. Acc Chem Res 14:363–368
Glascoe EA, Zang JM, Burnham K (2009) Pressure-dependent decomposition kinetics of the energetic material HMX up to 3.6 GPa. J Phys Chem A 113:13548–13555
Goetz Z, Brlll TB (1979) Laser Raman spectra of .alpha.-, .beta.-, .gamma.-, and .delta.-octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine and their temperature dependence. J Phys Chem 83:340–346
Henson BF, Asay BW, Sander RK, Son SF, Robinson JM, Dickson PM (1999) Dynamic measurement of the HMXβ-δphase transition by second harmonic generation. Phys Rev Lett 82:1213–1216
Henson BF, Smilowitz L, Asay BW, Dickson PM (2002) The β–δ phase transition in the energetic nitramine octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine: thermodynamics. J Chem Phys 117:3780–3788
Herrmann M, Engel W, Eisenreich N (1993) Thermal analysis of the phases of HMX using X-ray diffraction. Zeitschrift Für Kristallographie Crystal Mater 204:121–128
Ji GF, Xiao HM, Dong HS (2002) High level calculations on structure and properties of crystalline β-HMX. Acta Chim Sin 60:194–199
Karpowicz RJ, Gelfand LS, Brill TB (1983) Application of solid-phase transition kinetics to the properties of HMX. AIAA J 21:310–312
Kuklja MM, Tsyshevsky RV, Sharia O (2017) Elucidation of high sensitivity of δ-HMX: New insight from first principles simulations. Am Inst Phys 1793:070007
Lewis JP (2003) Energetics of intermolecular HONO formation in condensed-phase octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine (HMX). Chem Phys Lett 371:588–593
Liu Z, Zhu W, Ji G, Song K, Xiao H (2017) Decomposition mechanisms of α-octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine nanoparticles at high temperatures. J Phys Chem C 121:7728–7740
Lu LY, Wei DQ, Chen XR, Lian D, Ji GF, Zhang QM, Gong ZZ (2008) The first principle studies of the structural and vibrational properties of solid β-HMX under compression. Mol Phys 106:2569–2580
Nlemantsverdrlet JW, Kraan AMVD, Dilk WLV, Bean SVD (1980) Behavior of metallic iron catalysts during Fischer-Tropsch synthesis studied with Mössbauer spectroscopy, x-ray diffraction, carbon content determination, and reaction kinetic measurements. J Phys Chem 84:3363–3370
Plimpton S (1995) Fast parallel algorithms for short-range molecular dynamics. J Comput Phys 117:1–19
Pravica M, Galley M, Kim E, Weck P, Liu Z (2010) A far- and mid-infrared study of HMX (octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine) under high pressure. Chem Phys Lett 500:28–34
Sharia O, Kuklja MM (2011) Modeling thermal decomposition mechanisms in gaseous and crystalline molecular materials: application to β-HMX. J Phys Chem B 115:12677–12686
Sharia O, Kuklja MM (2012a) Rapid materials degradation induced by surfaces and voids:ab initiomodeling of β-octatetramethylene tetranitramine. J Am Chem Soc 134:11815–11820
Sharia O, Kuklja MM (2012b) Surface-enhanced decomposition kinetics of molecular materials illustrated with cyclotetramethylene-tetranitramine. J Phys Chem C 116:11077–11081
Smilowitz L, Henson BF, Asay BW, Dickson PM (2002) The β–δ phase transition in the energetic nitramine-octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine: kinetics. J Chem Phys 117:3789–3798
Smilowitz L, Henson BF, Greenfield M, Sas A, Asay BW, Dickson PM (2004) On the nucleation mechanism of the β-δ phase transition in the energetic nitramine octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine. J Chem Phys 121:5550–5552
Stevens LL, Eckhardt CJ (2005) The elastic constants and related properties of β-HMX determined by Brillouin scattering. J Chem Phys 122:174701
Stukowski A (2010) Visualization and analysis of atomistic simulation data with OVITO–the open visualization tool. Model Simul Mater Sci Eng 18:015012
Su R, Long Y, Jiang SL, He J, Chen J (2012) Elastic properties of β-HMX under extra pressure: a first principles study. Acta Phys Sin 61:206201–206201
Tappan AS, Renlund AM, Gieske JH, Miller JC (1999) Office of Scientific & Technical Information Technical Reports
Weeks BL, Ruddle CM, Zaug JM, Cook DJ (2002) Monitoring high-temperature solid–solid phase transitions of HMX with atomic force microscopy. Ultramicroscopy 93:19–23
Weese RK, Maienschein JL, Perrino CT (2003) Kinetics of the β → δ solid–solid phase transition of HMX, octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine. Thermochim Acta 401:1–7
Wemhoff AP, Burnham AK, Nichols AL (2007) Application of global kinetic models to HMX β−δ transition and cookoff processes. J Phys Chem A 111:1575–1584
Wu Z, Kalia RK, Nakano A, Vashishta P (2011) Vibrational and thermodynamic properties of β-HMX: A first-principles investigation. J Chem Phys 134:204509
Wu Q, Xiong G, Zhu W, Xiao H (2015) How does low temperature coupled with different pressures affect initiation mechanisms and subsequent decompositions in nitramine explosive HMX? Phys Chem Chem Phys 17:22823–22831
Xiang D, Zhu W (2018) Adiabatic and constant volume decomposition process of condensed phase δ-1,3,5,7-tetranitro-1,3,5,7-tetrazocane at high temperatures: quantum molecular dynamics simulations. J Mol Graph Model 85:68–74
Xue C, Sun J, Kang B, Liu Y, Liu X, Song G, Xue Q (2010) The β‐δ-Phase Transition and Thermal Expansion of Octahydro‐1,3,5,7-Tetranitro‐1,3,5,7‐Tetrazocine. Propellants Explos Pyrotech 35:333–338
Ye CC, An Q, Zhang WQ, Goddard WA (2019) Initial decomposition of HMX energetic material from quantum molecular dynamics and the molecular structure transition of β-HMX to δ-HMX. J Phys Chem C 123:9231–9236
Yoo CS, Cynn H (1999) Equation of state, phase transition, decomposition of β-HMX (octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine) at high pressures. J Chem Phys 111:10229–10235
Zaug JM, Farber DL, Saw CK, Weeks BL (2002) Kinetics of solid phase reactions at high pressure and temperature United States. Lawrence Livermore National Laboratory, California
Zerilli FJ, Kuklja MM (2010) Ab initio equation of state of the organic molecular crystal: β-octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine. J Phys Chem A 114:5372–5376
Zhao XS, Hintsa EJ, Lee YT (1988) Infrared multiphoton dissociation of RDX in a molecular beam. J Chem Phys 88:801–810
Funding
This research was supported by the National Natural Science Foundation of China (Grant No. 21773119) and Science Challenging Program (No. TZ2016001).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhu, S., Zhu, W. β → δ phase transition and initial decomposition of HMX nanoparticle from reactive molecular dynamics simulations. J Nanopart Res 22, 362 (2020). https://doi.org/10.1007/s11051-020-05099-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-020-05099-9