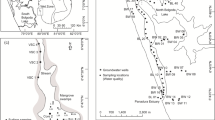
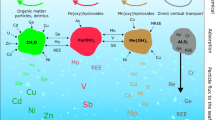
Abstract
Extensive field hydrogeochemical data and thermodynamic simulations demonstrate that the composition of lacustrine waters is controlled not only by evaporation but also by interaction between the waters and rocks. This interaction begins in the catchment areas of the lakes and continues in these lakes themselves, because the waters (regardless of their salinity) do not occur in equilibrium with many of the endogenic minerals. These processes are the most intense in soda lakes, as follows from the highest pH of their waters (9.0–10.7). These values provide evidence that these waters most intensely interact with primary aluminosilicates. The two processes responsible for sulfur behavior in these lakes are sulfate reduction and sulfide oxidation. Different geochemical environments produced in lakes of different type are favorable for the selective enrichment of some elements, which are mobile in these environments and whose concentrations can significantly increase.










Similar content being viewed by others
REFERENCES
V. A. Alekseev, M. B. Bukaty, V. P. Zverev, O. V. Chudaev, and S. L. Shvartsev, Geological Evolution and Self-Organization of the Water—Rock System. Volume 1. Water—Rock Syste, in the Earth’s Crust: Interaction, Kinetics, Equilibrium, and Modeling, Ed. by S. L. Shvartsev (SO RAN, Novosibirsk, 2005) [in Russian].
S. V. Borzenko, Extended Abstract of Doctoral Dissertation in Geology and Mineralogy (Inst. Prir. Res. Ekol., Criol., Sib. Otd. Ross. Akad. Nauk, Chita 2018).
S. V. Borzenko and Shvartsev S. L. “Chemical composition of salt lakes in East Transbaikalia (Russia),” Appl. Geochem.103, 72–84 (2019). https://doi.org/10.1016/j.apgeochem.2019.02.014
S. V. Borzenko and L. V. Zamana, “Reduced forms of sulfur in the brine of saline–soda Lake Doroninskoe, Eastern Transbaikal region,” Geochem. Int. 49 (3), 253–261 (2011).
M. B. Bukaty, “Development of software for petroleum hydrogeology,” Razved. Okhr. Nedr, No. 2, 37–39 (1997).
J. Chen, F. Wang, G. Wan, D. Tang, D. D. Zang, R. Hunag, J. Li, and T. Xiao, “Sulphate reduction and sulfur cycling in lake sediments,” Acta Geol. Sinica 82 (5), 975–981 (2008).
A. Droubi, “Geochimie des sels et des solutions concentrees par evaporation,” Modele Thermodynamique de Simulation. Application aux Sols Sales du Tchad, Sci.Ceol., Mem. 46, (1976).
A. Droubi, C. Cheverry, B. Fritz, and Y. Tardy, “Geochimie des eaux et des sels dans les sols des polders du lac Tchad: application d’un model thermodynamique de simulation de l’evaporation,” Chem. Geol. 17 (3), 165–177 (1976).
A. I. Dzens-Litovsky, Kara-Bogaz-Gol (Nedra, Leningrad, 1967) [in Russian].
V. K. Filippov and M. V. Charykova, “Die Anwendung der Pitzer–Gleichungen fur die Ber–echnung der Phasengleichgewichte in quaternaren system Na+, \({\text{NH}}_{4}^{ + }\) Cl, \({\text{SO}}_{4}^{{2 - }}\) H2O,” Z. Phys. Chem. 270(1), 49–56 (1989).
V. K. Filippov and L. M. Cheremnykh, “Calculation of thermodynamic functions of the Na2SO4–K2SO4–H2O system at 25°C,” Zh. Prikl. Khim. 56 (7), 1475–1479 (1983).
R. M. Garrels, Solutions, Minerals, and Equilibria (Harper & Row, New York, 1965)
J. P. Greenberg and N. Moller, “The prediction of mineral solubilities in natural waters: A chemical equilibrium model the Na–K–Ca–Cl–SO4–H2O system to high concentration from 0 to 250°C,” Geochim. Cosmochim. Acta 53 (10), 2503–2518 (1989).
V. A. Grinenko and L. N Grinenko, Sulfur Isotope Geochemistry (Nauka, Moscow, 1974) [in Russian].
M. Gueddari, “Geochimie et thermodynamique deseri dena eri denals. Etude du Lac Natron en Tanzanie et du Chott el eri den Tunisie,“ Sci. Ceol., Mem. (76), 143 (1984).
M. Gueddari, C. Monnin, D. Perret, B. Fritz, and Y. Tardy, “Geochemistry of brines of the Chott el Jerid in Southern Tunisia application of Pitzer’s equations,” Chem. Geol. 39 (1), 165–178 (1983).
L. A. Hardie and H. P. Eugster, “Evaporation of seawater: calculated mineral sequences,” Science 208, 498–500 (1980).
A. V. Ivanov, Hydrochemistry of Rivers and Lakes in Zone of Extremely Continental Climate (DVGU. Vladivostok, 1977) [in Russian].
B. F. Jones, H. P. Eugster, and S. L. Rettig, “Hydrochemistry of the Lake Magadi basin, Kenya,” Geochim. Cosmochim. Acta 41 (1), 53–72 (1977).
W. D. Keller, The Principles of Chemical Weathering (Lucas, Columbia, 1955).
M. N. Kolpakova, S. L. Shvartsev, S. V. Borzenko, V. P. Isupov, and S. S. Shatskaya, “Geochemical features of Kulunda plain lakes (Altay region, Russia),” IOP Conf. Ser.: Earth and Environmental Science 33 (1), no. 012007 (2016).
S. R. Krainov, B. N. Ryzhenko, and V. M. Shvets, Geochemistry of Groundwaters. Theoretical, Applied, and Ecological Aspects (Nauka, Moscow, 2004) [in Russian].
V. V. Kurilenko, Modern Basins of Evaporite Sedimentation (St. Petersburg, St. Petersb. Gos. Univ., 1997) [in Russian].
N. S. Kurnakova, and V. I. Nikolaeva, “Solar evaporation of seawater and lacustrine brines,” Izv. Inst. Fiz.-Khim. Anal. Akad. Nauk SSSR, No. 10, 333–366 (1938).
G. A. Leonova, V. A. Bobrov, A. A. Bogush, G. N. Anoshin, and V. A. Bychinskii, “Geochemical characteristics of the modern state of salt lakes in Altai Krai,” Geochem. Int. 45 (10), 1025–1039 (2007).
S. Matsuo, Y. Kuroda, and T. Suzuoki, “Mantle water based on the hydrogen isotope ratios of hydrouse silicates in the mantel,” Geol. Surv. Open–file Rep. 701, 278–280 (1978).
M. A. McCaffrey, B. Lazar, and H. D. Holland, “The evaporation path of seawater and the coprecipitation of Br– and K+ with halite,” J. Sedim. Petrol. 57, 928–937 (1987).
Yu. V. Novikov, K. O. Lastochkina, and Z. N. Boldina, Methods of Study of Water Quality in Basins (Meditsina, Moscow, 1990) [in Russian].
E. V. Pinneker, Main Hypotheses of the Formation of Composition of Concentrated Brines. Principles of Hydrogeology. Hydrogeochemistry (Nauka, Novosibirsk, 1982), pp. 202–206 [in Russian].
K. S. Pitser, “A thermodynamic model for aqueous solutions of liquid-like density,” Thermodynamc Mdeling of Geological Materials: Minerals, Fluids, and Melts, Rev. Mineral., Ed. by I. S. E.Carmichael and H. P. Eugster, 17, 97—142 (1987).
S. L. Shvartsev, Hydrogeochemistry of Supergene Zone (Nedra, Moscow, 1978) [in Russian].
S. L. Shvartsev, Hydrogeochemistry of Supergene Zone (Nedra, Moscow, 1998) [in Russian].
S. L. Shvartsev, M. N. Kolpakova, V. P. Isupov, A. G. Vladimirov, and S. Ariunbileg, “Geochemistry and chemical evolution of saline lakes of western Mongolia,” Geochem. Int. 52(5), 388–403 (2014).
E. V. Sklyarov, O. A. Sklyarova, Yu. V. Men’shagin, and M. A. Danilova, “Mineralized lakes of the Transbaikalia and Northeastern Mongolia: specific features of occurrence and ore-generating potential,” Geograph. Prir. Resour. 32 (4), 323–332 (2011).
P. Sonnenfeld, Brines and Evaporites (Academic Press, New York, 1984)
State Geological Map of the Russian Federation. Scale 1 : 1000000 (Third Generation). Aldan—Transbaikalian Series. Sheet M–50 – Borzya. Explanatory Notes, Ed. by E. A. Shivokhin et al., (VSEGEI, St. Petersburg, 2010) [in Russian].
N. M. Strakhov, Principles of Lithogenesis Theory (TLI, Moscow, 1962) [in Russian].
V. D. Strakhovenko, B. L. Shcherbov, I. N. Malikova, and Yu. S. Vosel’, “The regularities of disgtribution of radionuclides and rare-earth elements in bottom sediments of Siberial lakes,” Russ. Geol. Geophys. 51 (11), 1167–1178 (2010).
Textbook on the Chemical Analysis of Continental Surface Waters, Ed. by A. D. Semenov (Gidrometeoizdat, Leningrad, 1977) [in Russian].
J. H. Van’t Hoff, Zur Bildung der Ozeanischen Salzablagerungen, Erstes Heft, Zweites Heft (Vieweg und Sohn, Braunschweig, 1905, 1909).
N. A. Vlasov, Mineral Waters of Southern East Siberia. Volume 1. Hydrogeology of Mineral Waters and their National Economic Significance, Ed. by V. G. Tkachuk and N. I. Tolstikhina (AN SSSR, Moscow—Leningrad, 1961) [in Russian].
J. K. Warren, “Evaporite sedimentology,” J. Sediment. 29 (5), 548–556 (1989).
A. G. Zakharyuk, Extended Abstract of Candidate’s Dissertation in Biology (Inst. Obshch. Eksp. Biol., Ulan-Ude, 2010)
L. V. Zamana and S. V. Borzenko, “Hydrochemical mode of salinized lakes of southeastern Transbaikalia,” Geograf. Prir. Resour. 31 (4), 370–376 (2010).
M. Zheng, Saline Lakes and Salt Basin Deposits in China (Science Press, Beijing, 2014).
I. K. Zherebtsova and N. N. Volkova, “Experimental study of trace-elememnt behavior during natural solar evaporation of Black Sea water and the Sasyk—Sivash Lake brine,” Geokhimiya, No. 7, 832–845 (1966).
Funding
This study was carried out under a government-financed research project and was partly supported by the Russian Foundation for Basic Research, project no. 18-05-00104.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by E. Kurdyukov
Rights and permissions
About this article
Cite this article
Borzenko, S.V. Principal Parameters Controlling Water Composition in Saline and Brackish Lakes in Eastern Transbaikalia. Geochem. Int. 58, 1356–1373 (2020). https://doi.org/10.1134/S0016702920090037
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0016702920090037