Abstract
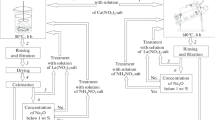
Various zeolites for preparing an active heterogeneous catalyst for the commercial alkylate production process were studied. NaX zeolites produced by Russian catalyst plants (powdered, granulated without binders, and formed with binders) were used in the experiments. The zeolites were converted to the active form by successive ion exchange with aqueous solutions of calcium, lanthanum, and ammonium salts at 140°C in a rotating autoclave. Isobutane alkylation with butylenes was performed on a flow-through laboratory installation in a fixed-catalyst-bed reactor. Comparative analysis of the physicochemical (crystal structure, acidity, mechanical strength) and catalytic (conversion of butylenes, alkylate yield, selectivity) properties of the synthesized catalysts in the CaLaHX form was performed. With respect to the set of the properties, the sample based on the NaX-ch catalyst and formed with a binder (clay) is the best catalyst. It exhibits good strength, high acidity, and high activity (conversion of butylenes 100 wt % , alkylate yields based on butylenes in the feed 99 wt % , selectivity with respect to trimethylpentanes 77.1 wt % ).



Similar content being viewed by others
Notes
Treacy, M.M.J. and Higgins, J.B., Collection of Simulated XRD Powder Patterns for Zeolites, Elsevier, 2007, 5th ed., p. 170.
REFERENCES
Vogt, E.T.C., Whiting, G.T., Chowdhury, A.D., and Weckhuysen, B.M., Adv. Catal., 2015, vol. 58, pp. 143–314. https://doi.org/10.1016/bs.acat.2015.10.001
Khadzhiev, S.N., Gerzeliev, I.M., Vedernikov, O.S., Kleymenov, A.V., Kondrashev, D.O., Oknina, N.V., Kuznetsov, S.E., Saitov, Z.A., and Baskhanova, M.N., Catal. Ind., 2017, vol. 9, pp. 198–203. https://doi.org/10.1134/S2070050417030059
Chen, Zh., Gao, F., Ren, K., Wu, Q., Luo, Y., Zhou, H., Zhang, M., and Xu, Q., RSC Adv., 2018, vol. 8, no. 7, pp. 3392–3398. https://doi.org/10.1039/C7RA12629H
Zhang, H., Xu, J., Tang, H., Yang, Z., Liu, R., and Zhang, S., Ind. Eng. Chem. Res., 2019, vol. 58, no. 22, pp. 9690–9700. https://doi.org/10.1021/acs.iecr.9b01638
Feller, A., J. Catal., 2003, vol. 216, pp. 313–323. https://doi.org/10.1016/S0021-9517(02)00068-4
Josl, R., Catal. Commun., 2004, vol. 5, no. 5, pp. 239–241. https://doi.org/10.1016/j.catcom.2004.02.005
Klingmann, R., Josl, R., Traa, Y., Gläser, R., and Weitkamp, J., Appl. Catal. A, 2005, vol. 281, nos. 1–2, pp. 215–223. https://doi.org/10.1016/j.apcata.2004.11.032
Ginosar, D.M., Thompsom, D.N., and Burch, K.C., Appl. Catal. A, 2004, vol. 262, pp. 223–321. https://doi.org/10.1016/j.apcata.2003.11.030
Guzman, A., Zuazo, I., Feller, A., Olindo, R., Sievers, C., and Lercher, J.A., Micropor. Mesopor. Mater., 2005, vol. 83, nos. 1–3, pp. 309–318. https://doi.org/10.1016/j.micromeso.2005.04.024
Sievers, C., Zuaza, I., Guzman, A., Olindo, R., Syska, Y., and Lercher, J., J. Catal., 2007, vol. 246, no. 2, pp. 315–324. https://doi.org/10.1016/j.jcat.2006.11.015
Zhuang, Y. and Ng, F.T.T., Appl. Catal. A, 2000, vol. 190, nos. 1–2, pp. 137–147. https://doi.org/10.1016/S0926-860x(99)00287-2
Costa, B.O.D. and Querini, C.A., Appl. Catal. A, 2010, vol. 385, nos. 1–2, pp. 144–152. https://doi.org/10.1016/j.apcata.2010.07.007
Sievers, C., Liebert, J.S., Stratmann, M.M., Olindo, R., and Lercher, J.A., Appl. Catal. A, 2008, vol. 336, nos. 1–2, pp. 89–100. https://doi.org/10.1016/j.apcata.2007.09.039
Yoo, K.S. and Smirniotis, P.G., Catal. Lett., 2005, vol. 103, nos. 3–4, pp. 249–255. https://doi.org/10.1007/s10562-005-7161-7
Feller, A. and Lercher, J.A., Adv. Catal., 2004, vol. 48, pp. 229–295. https://doi.org/10.1016/S0360-0564(04)48003-1
Nayak, S.V., Morali, M., Ramachandran, P.A., and Dudukovic, M.P., J. Catal., 2009, vol. 226, pp. 169–181. https://doi.org/10.1016/j.jcat.2009.06.008
Sarsani, V.R. and Subramaniam, B., Green Chem., 2009, vol. 11, no. 1, pp. 102–108. https://doi.org/10.1039/b808418
Schüßler, F., Schallmoser, S., Shi, H., Haller, G.L. Ember, E., and Lercher, J.A., ACS Catal., 2014, vol. 4, no. 6, pp. 1743–1752. https://doi.org/10.1021/cs500200k
Feller, A., Guzman, A., Zuazo, I., and Lercher, J.A., J. Catal., 2004, vol. 224, no. 1, pp. 80–93. https://doi.org/10.1016/j.jcat.2004.02.019
Stöcker, M., Mostad, H., and Rørvik, T., Catal. Lett., 1994, vol. 28, nos. 2–4, pp. 203–209. https://doi.org/10.1007/BF00806049
Ro, Y., Gim, M.Y., Lee, J.W., Lee, E.J., and Song, I.K., J. Nanosci. Nanotechnol., 2018, vol. 18, no. 9, pp. 6547–6551. https://doi.org/10.1166/jnn.2018.15665
Doyle, A.M., Alismaeel, Z.T., Albayati, T.M., and Abbas, A.S., Fuel, 2017, vol. 199, pp. 394–402. https://doi.org/10.1016/j.fuel.2017.02.098
Dzubiella, J. and Hansen, J.P., J. Chem. Phys., 2005, vol. 122, no. 23, ID 234706. https://doi.org/10.1063/1.1927514
Gerzeliev, I.M., Ostroumova, V.A., Baskhanova, M.N., Saitov, Z.A., Temnikova, V.A., and Khusaimova, D.O., Petrol. Chem., 2017, vol. 57, no. 12, pp. 1182–1185. https://doi.org/10.1134/S0965544117060147
Patent RU 195954, Publ. 2020.
Patent RU 2404122, Publ. 2010.
Nazir, L.S.M., Yeong, Y.F., and Chew, T.L., Colloids Surf. A, 2020, vol. 600, ID 124803. https://doi.org/10.1016/j.colsurfa.2020.124803
Didi, Y., Said, B., Cacciaguerra, T., Nguyen, K.L., Wernert, V., Denoyel, R., Cot, D., Sebai, W., Belleville, M.P., Sanchez-Marcano, J., Fajula, F., and Galarneau, A., Micropor. Mesopor. Mater., 2019, vol. 281, pp. 57–65. https://doi.org/10.1016/j.micromeso.2019.03.003
Gerzeliev, I.M., Temnikova, V.A., Baskhanova, M.N., and Maksimov, A.L., Petrol. Chem., 2019, vol. 59, no. 11, pp. 1213–1219. https://doi.org/10.1134/S0965544119110021
Funding
The study was financially supported by the Ministry of Science and Higher Education of the Russian Federation (granting agreement no. 075-15-2019-1848, unique project identifier RFMEFI60419X0246).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
A.L. Maksimov is the Editor-in-Chief of Zhurnal Prikladnoi Khimii (Russian Journal of Applied Chemistry). The other authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Gerzeliev, I.M., Temnikova, V.A., Baskhanova, M.N. et al. Alkylation of Isobutane with Butylenes on Catalysts with Various NaX Zeolites in the CaLaHX Form. Russ J Appl Chem 93, 1578–1585 (2020). https://doi.org/10.1134/S107042722010134
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107042722010134