Abstract
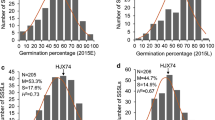
Developing new population types based on interspecific introgressions has been suggested to facilitate the discovery of novel allelic sources for traits of agronomic importance. In this study, a chromosome segment substitution line (CSSL) rice population carrying contiguous chromosomal segments of japonica Nipponbare in the genetic background of indica 9311 was developed. Meanwhile, a high-density genetic map including 655 bins covering a distance of 1480.2 Mb, with an average of 0.60 Mb between markers, was generated through a whole-genome resequencing approach. Compared with previous CSSL populations constructed from 9311 or Nipponbare, this population has a higher marker density and can effectively meet the requirements of high QTL mapping resolution. On the basis of the newly constructed high-density genetic map, a total of three major QTLs (qGP4, qGP5, and qBL4) conferring seed growth capacity after low-temperature (5 °C) treatment were detected. Among these, two QTLs shared by Nipponbare, qGP4 and qGP5, could increase germination percentage, while one QTL shared by Nipponbare, qBL4, could increase bud length after cold stress; qGP4 and qBL4 were first reported in our study. These QTLs could be highly valuable genetic factors for cold tolerance improvement in rice lines. The CSSLs carrying these QTLs not only provide an opportunity for map-based cloning of important cold tolerance QTLs but also supply useful inbred lines to improve cold tolerance in 9311. These CSSLs represent good sources of japonica tolerance QTLs (genes) for use in improving cold tolerance in indica rice directly, avoiding the genetic drag arising from incompatible epistasis effects.







Similar content being viewed by others
Data availability
Not applicable.
References
Balakrishnan D, Surapaneni M, Mesapogu S, Neelamraju S (2019) Development and use of chromosome segment substitution lines as a genetic resource for crop improvement. Theor Appl Genet 132:1–25
Bian J, Jiang L, Liu LL, Wei XJ, Xiao YH, Zhang LJ, Zhao ZG, Zhai HQ, Wan JM (2010) Construction of a new set of rice chromosome segment substitution lines and identifcation of grain weight and related traits QTLs. Breed Sci 60:305–313
Chen QQ, Mu JX, Zhou HJ, Yu SB (2007) Genetic efect of japonica alleles detected in indica candidate introgression lines. Sci Agric Sin 40:2379–2387
Fujino K, Matsuda Y (2010) Genome-wide analysis of genes targeted by qLTG3-1 controlling low-temperature germinability in rice. Plant Mol Biol 72:137–152
Fujino K, Sekeguchi H, Matsuda Y, Sugimoto K, Ono K, Yano M (2008) Molecular identification of a major quantitative trait locus, qLTG3-1, controlling low-temperature germinability in rice. Proc Natl Acad Sci U S A 105(34):12623–12628
Guo F, Huiwen Yu H, Tang Z, Jiang X, Wang L, Wang X, Xu Q, Deng X (2015) Construction of a SNP-based high-density genetic map for pummelo using RAD sequencing. Tree Genet Genome 11:2
Hori K, Sugimoto K, Nonoue Y, Ono N, Matsuba K, Yamanouchi U, Abe A, Takeuchi Y, Yano M (2010) Detection of quantitative trait loci controlling pre-harvest sprouting resistance by using backcrossed populations of japonica rice cultivars. Theor Appl Genet 120:1547–1557
Huang X, Feng Q, Qian Q, Zhao Q, Wang L, Wang A, Guan J, Fan D, Weng Q, Huang T, Dong G, Sang T, Han B (2009) High-throughput genotyping by whole-genome resequencing. Genome Res 19:1068–1076
Huang X, Kurata N, Wei X, Wang Z, Wang A, Zhao Q, Zhao Y, Liu K, Lu H, Li W, Guo Y, Lu Y, Zhou C, Fan D, Weng Q, Zhu C, Huang T, Zhang L, Wang Y, Feng L, Furuumi H, Kubo T, Miyabayashi T, Yuan X, Xu Q, Dong G, Zhan Q, Li C, Fujiyama A, Toyoda A, Lu T, Feng Q, Qian Q, Li J, Han B (2012) A map of rice genome variation reveals the origin of cultivated rice. Nature 490(7421):497–501
Iwata N, Shinada H, Kiuchi H, Sato T, Fujino K (2010) Mapping of QTLs controlling seedling establishment using a direct seeding method in rice. Breed Sci 60:353–360
Ji SL, Jiang L, Wang YH, Liu SJ, Liu X, Zhai HQ, Wan JM (2007) Detection and analysis of QTL for germination rate at low temperature in rice(Oryza sativa L.) using backcross inbred lines.Journal of. Nanjing Agric Univ 30(1):1–6
Jiang N, Shi S, Shi H, Khanzada H, Wassan GM, Zhu C, Peng X, Yu Q, Chen X, He X, Fu J, Hu L, Xu J, Ouyang L, Sun X, Zhou D, He H, Bian J (2017) Mapping QTL for seed germinability under low temperature using a new high-density genetic map of rice. Front Plant Sci 12(8):1223
Koseki M, Kitazawa N, Yonebayashi S, Maehara Y, Wang ZX, Minobe Y (2010) Identification and fine mapping of a major quantitative trait locus originating from wild rice, controlling cold tolerance at the seedling stage. Mol Gen Genomics 284:45–54
Li L, Mao D (2018) Deployment of cold tolerance loci from Oryza sativa ssp. Japonica cv. ‘Nipponbare’ in a high-yielding Indica rice cultivar ‘93-11’. Plant Breed 137:553–560
Li HH, Zhang LY, Wang JK (2010) Analysis and solution of some common problems in quantitative trait gene mapping research. Acta Agron Sin 36(06):918–931
Li L, Chen H, Ma D (2019) Pyramiding of rapid germination loci from Oryza Sativa cultivar ‘Xieqingzao B’and cold tolerance loci from Dongxiang wild rice to increase. Mol Breed 39:85
Liu F, Xu W, Song Q, Tan L, Liu J, Zhu Z, Fu Y, Su Z, Sun C (2013) Microarray-assisted fine-mapping of quantitative trait loci for cold tolerance in rice. Mol Plant 6(3):757–767
Liu C, Ou S, Mao B, Tang J, Wang W, Wang H, Cao S, Schläppi MR, Zhao B, Xiao G, Wang X, Chu C (2018) Early selection of bZIP73 facilitated adaptation of japonica rice to cold climates. Nat Commun 9(1)
Liu C, Schläppi MR, Mao B, Wang W, Wang A, Chu C (2019) The bZIP73 transcription factor controls rice cold tolerance at the reproductive stage. Plant Biotechnol J
Liu W, Pan X, Xiong H, Huang F, Duan Y, Li Y, Min J, Liu S, Liu L, Wei X, Zhao W, Li X (2020) QTL mapping and validation of a segment responsible for early-seedling cold tolerance in rice. Euphytica 216:7
Lou QJ, Chen L, Sun ZX, Xing YZ, Li J, Xu XY, Mei HW, Luo LJ (2007) A major QTL associated with cold tolerance at seedling stage in rice (Oryza sativa L.). Euphytica 158:87–94
Ma Y, Dai XY, Xu YY, Luo W, Zheng XM, Zeng DL, Pan YJ, Lin XL, Liu HH, Zhang DJ, Xiao J, Guo XY, Xu SJ, Niu YD, Jin JB, Zhang H, Xu X, Li LG, Wang W, Qian Q, Ge S, Chong K (2015) COLD1 confers chilling tolerance in rice. Cell 160:1–13
Mackill DJ, Lei X (1997) Genetic variation for traits related to temperate adaptation of rice cultivars. Crop Sci 37:1340–1346
Mao D, Xin Y, Tan Y, Hu X, Bai J, Liu Z, Yu Y, Li L, Peng C, Fan T, Zhu Y, Guo Y, Wang S, Lu D, Xing Y, Yuan L, Chen C (2019) Natural variation in the HAN1 gene confers chilling tolerance in rice and allowed adaptation to a temperate climate. Proc Natl Acad Sci 116(9):3494–3501
McCouch SR (2008) Gene nomenclature system for rice. Rice 1:72–84
Murray M, Thompson W (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8(19):4321–4325
Peng X, He H, Zhu G, Jiang L, Zhu C, Yu Q, He J, Shen X, Yan S, Bian J (2017) Identifcation and analyses of chromosome segments affecting heterosis using chromosome-segment substitution lines in rice. Crop Sci 57(4):1836–1843
Qiao W, Qi L, Cheng Z, Su L, Li J, Sun Y, Ren J, Zheng X, Yang Q (2016) Development and characterization of chromosome segment substitution lines derived from Oryza rufipogon in the genetic background of O. sativa spp. indica cultivar 9311. BMC Genomics 17:580
Saito K, Hayano-Saito Y, Kuroki M, Sato Y (2010) Map-based cloning of the rice cold tolerance gene Ctb1.Plant. Sci 179(1–2):97–102
Sasaki T, Burr B (2000) International rice genome sequencing project: the effort to completely sequence the rice genome. Curr Opin Plant Biol 3:138–141
Shen YJ, Jiang H, Jin JP, Zhang ZB, Xi B, He YY, Wang G, Wang C, Qian L, Li X, Yu QB, Liu HJ, Chen DH, Gao JH, Huang H, Shi TL, Yang ZN (2004) Development of genome-wide DNA polymorphism database for map-based cloning of rice genes. Plant Physiol 135(3):1198–1205
Udin SN, Soejadi (1991) Predrying and soaking seeds can be effective methods to break the dormancy of IR64 seeds. Seed Sci Technol 19(2)
Van OJ (2006) JoinMap 4 software for the calculation of genetic linkage maps in experimental populations. Kyazma BV, Wageningen
Wang Z, Wang F, Zhou R, Wang J, Zhang H (2011) Identification of quantitative trait loci for cold tolerance during the germination and seedling stages in rice (Oryza sativa L.). Euphytica 181:405–413
Wang X, Fang G, Li Y, Ding M, Gong H, Li Y (2013) Differential antioxidant responses to cold stress in cell suspension cultures of two subspecies of rice. Plant Cell Tissue Organ Cult 113:353–361
Xiao N, Gao Y, Qian H, Gao Q, Wu Y, Zhang D, Zhang X, Yu L, Li Y, Pan C, Liu G, Zhou C, Jiang M, Huang N, Dai Z, Liang C, Chen Z, Chen J, Li A (2018) Identification of genes related to cold tolerance and a functional allele that confers cold tolerance. Plant Physiol 177(3):1108–1123
Xie L, Tan Z, Zhou Y, Xu R, Feng L, Xing Y, Qi X (2014) Identification and fine mapping of quantitative trait loci for seed vigor in germination and seedling establishment in rice. J Integr Plant Biol 56:749–759
Xu J, Zhao Q, Du P, Xu C, Wang B, Feng Q, Liu Q, Tang S, Gu M, Han B, Liang G (2010) Developing high throughput genotyped chromosome segment substitution lines based on population whole-genome re-sequencing in rice (Oryza sativa L.). BMC Genomics 11:656
Yu CY, Wan JM, Zhai HQ, Wang CM, Jiang L, Xiao YH, Liu YQ (2005) Study on heterosis of intersubspecies between indica and japonica rice (Oryza sativa L.) using chromosome segment substitution lines. Chin Sci Bull 50:131–136
Zhang ZH, Li S, Wei L, Wei C, Zhu YG (2005) A major QTL conferring cold tolerance at the early seedling stage using recombinant inbred lines of rice (Oryza sativa L.). Plant Sci 168:527–534
Zhang H, Zhao Q, Sun ZZ, Zhang CQ, Feng Q, Tang SZ, Liang GH, Gu MH, Han B, Liu QQ (2011) Development and high-throughput genotyping of substitution lines carrying the chromosome segments of indica 9311 in the background of japonica Nipponbare. J Genet Genomics 38:603–611
Zhang Z, Li J, Pan Y, Li J, Zhou L, Shi H, Zeng Y, Guo H, Yang S, Zheng W, Yu J, Sun X, Li G, Ding Y, Ma L, Shen S, Dai L, Zhang H, Yang S, Guo Y, Li Z (2017) Natural variation in CTB4a enhances rice adaptation to cold habitats. Nat Commun 8(1):14788
Zhao L, Zhou H, Lu L, Liu L, Li X, Lin Y, Yu S (2009) Identification of quantitative trait loci controlling rice mature seed culturability using chromosomal segment substitution lines. Plant Cell Rep 28:247–256
Zhao J, Zhang S, Dong J, Yang T, Mao X, Liu Q, Wang X, Liu B (2017) A novel functional gene associated with cold tolerance at the seedling stage in rice.Plant. Biotechnol J 15(9):1141–1148
Zhao J, Wang S, Qin J, Sun C, Liu F (2019) The lipid transfer protein OsLTPL159 is involved in cold tolerance at the early seedling stage in rice.Plant Biotechnol J 18 (3):756–769
Zhu WY, Lin J, Yang DW, Zhao L, Zhang YD, Zhu Z, Chen T, Wang CL (2009) Development of chromosome segment substitution lines derived from backcross between two sequenced rice cultivars, indica recipient 93-11 and japonica donor Nipponbare. Plant Mol Biol Rep 27:126–131
Acknowledgments
We thank the anonymous referees for their critical comments on this manuscript.
Funding
This study was supported by grants 20192ACBL20017 and 20192BCB23010 from Project of Science and Technology Department of Jiangxi Province, grant (JXYJG-2018-062) from Jiangxi Provincial Department of Education.
Author information
Authors and Affiliations
Contributions
JB and HH designed the experiments. GW, HD, MY, YC, DZ, JT, JY, XL, ST, PW, XZ, CL, CL, YW, and QC performed the experiments and analyzed the results. GW, HD, and MY wrote the manuscript. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(XLSX 67 kb)
Rights and permissions
About this article
Cite this article
Wu, G., Deng, H., Yu, M. et al. Genetic analysis of rice seed recovery under low-temperature conditions using a new CSSL population with a high-density genetic map in rice. Mol Breeding 40, 109 (2020). https://doi.org/10.1007/s11032-020-01189-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11032-020-01189-7