Abstract
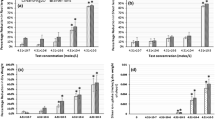
Nanoparticles are regularly released into our atmosphere, and the study of their effect on edible crop plants is of great interest today. This study accounts the enviro-friendly production of silver nanoparticles (SNPs) using Sarcococca saligna (D.Don) Muell.-Arg. leaf extract and the effect of these SNPs on growth and biochemical attributes of soybean (Glycine max L.). The nanoparticles obtained were spherical crystals with a mean size of 52.41 nm. Three soybean varieties namely Rawal‑1, NARC‑2, and Ajmari were exposed to four concentrations of SNPs (0, 25, 50 and 100 ppm) in a completely randomized design. A significant increment in seed germination (Ajmari), root length and shoot length (NARC-2) was recorded at 50 ppm SNPs while the leaf size positively increased at 25 ppm SNPs. The amount of chlorophyll, carotenoids and protein increased at 25 ppm SNPs. All other concentrations caused negative effects on these compounds. SNPs showed a positive effect on growth and biochemical attributes in G. max plant at specific concentrations (50 ppm for growth and 25 ppm for biochemical parameters). Nevertheless, to establish the most appropriate concentration, size and mode of application of SNPs for higher growth and maximum yield, a comprehensive experimentation is required.
Zusammenfassung
Nanopartikel werden regelmäßig in unsere Atmosphäre freigesetzt und die Untersuchung ihrer Wirkung auf essbare Nutzpflanzen ist heute von großem Interesse. Diese Studie beschreibt die umweltfreundliche Produktion von Silbernanopartikeln (SNPs) unter Verwendung eines Sarcococca saligna (D.Don) Muell.-Arg.-Blattextrakts und die Wirkung dieser SNPs auf das Wachstum und die biochemischen Eigenschaften von Sojabohnen (Glycine max L.). Die erhaltenen Nanopartikel waren kugelförmige Kristalle mit einer mittleren Größe von 52,41 nm. Drei Sojabohnensorten, nämlich Rawal‑1, NARC‑2 und Ajmari, wurden in einem vollständig randomisierten Design vier Konzentrationen von SNPs (0, 25, 50 und 100 ppm) ausgesetzt. Ein signifikanter Anstieg der Samenkeimung (Ajmari), der Wurzellänge und der Sprosslänge (NARC-2) wurde bei 50 ppm SNPs festgestellt, während die Blattgröße bei 25 ppm SNPs zunahm. Die Menge an Chlorophyll, Carotinoiden und Protein stieg bei 25 ppm SNPs an. Alle anderen Konzentrationen hatten negative Auswirkungen auf diese Komponenten. Die SNPs zeigten einen positiven Effekt auf das Wachstum und die biochemischen Eigenschaften in G. max-Pflanzen bei bestimmten Konzentrationen (50 ppm für Wachstum und 25 ppm für biochemische Parameter). Um die am besten geeignete Konzentration, Größe und Art der Anwendung von SNPs für ein höheres Wachstum und einen maximalen Ertrag zu ermitteln, sind jedoch umfassende Experimente erforderlich.





Similar content being viewed by others
References
Aiyegroro OA, Okoh AI (2010) Preliminary phytochemical screening and in vitro antioxidant activities of aqueous extract of Helichrysum longifolium DC. BMC Complement Altern Med 10:21
Bansod SD, Bawaskar MS, Gade AK, Rai MK (2015) Development of shampoo, soap and ointment formulated by green synthesized silver nanoparticles functionalized with antimicrobial plants oils in veterinary dermatology: treatment and prevention strategies. IET Nanobiotechnol 9:165–171
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Chem 72:248–254
Dimkpa CO, McLean JE, Martineau N, Britt DW, Haverkamp R, Anderson AJ (2013) Silver nanoparticles disrupt wheat (Triticum aestivum L.) growth in a sand matrix. Environ Sci Technol 47:1082–1090
Dubois M, Gilles K, Hamilton JK, Rebers PA, Smith F (1951) A colorimetric method for the estimation of sugar. Nature 168:167
Giraldo JP, Landry MP, Faltermeier SM, McNicholas TP, Iverson NM, Boghossian AA, Reuel NF, Hilmer AJ, Sen F, Brew JA (2014) Plant nanobionics approach to augment photosynthesis and biochemical sensing. Nat Mater 13:400–408
Haverkamp RG, Marshall AT (2009) The mechanism of metal nanoparticle formation in plants: limits on accumulation. J Nanopart Res 11:1453–1463
Iqbal M, Raja NI, Mashwani Z, Hussain M, Ejaz M, Yasmeen F (2019) Effect of silver nanoparticles on growth of wheat under heat stress. Iran J Sci Technol Trans Sci 43(2):387–395
Khatami M, Mortazavi SM, Kishani-Farahani Z, Amini E, Heli H (2017) Biosynthesis of silver nanoparticles using pine pollen and evaluation of the antifungal efficiency. Iran J Biotechnol 1:e1436
Kudle KR, Donda MR, Alwala J, Koyyati R, Nagati V, Merugu R, Prashanthi Y, Rudra M (2012) Biofabrication of silver nanoparticles using Cuminum cyminum through microwave irradiation. Int J Nanomater Biostruct 2:65–69
Kumar B, Smita K, Cumbal L (2017) Alexis Debut Green synthesis of silver nanoparticles using Andean blackberry fruit extract. Saudi J Biol Sci 24:45–50
Latif HH, Ghareib M, Tahon MA (2017) Phytosynthesis of silver nanoparticles using leaf extracts from Ocimum basilicum and Mangifira indica and their effect on some biochemical attributes of Triticum aestivum. Gesunde Pflanz 69:39–46
Lee W, Kwak JI, An Y (2012) Effect of silver nanoparticles in crop plants Phaseolus radiatus and Sorghum bicolor: media effect on phytotoxicity. Chemosphere 86:491–499
Lei Z, Mingyu S, Xiao W, Cha L, Chunxiang Q, Liang C (2008) Antioxidant stress is promoted by nano-anatase in spinach chloroplasts under UV‑B radiation. Biol Trace Elem Res 121:69–79
Li C, Dang LM, Zhu M, Zhong H, Hintelmann H, Zhou D (2017) Effects of exposure pathways on the accumulation and phytotoxicity of silver nanoparticles in soybean and rice. Nanotoxicology 11:699–705
Lichtenthaler HK, Gamble JS (2008) Flora of the presidency of Madras. Bishan Singh Mahendra Pal Singh, Dehra Dun
Liu KS (1997) Chemistry and nutritional value of soybean components. In: Soybean: chemistry, technology, and utilization. Chapman & Hall, New York, pp 25–113 (Chapter 2)
Mahajan P, Dhoke SK, Khanna AS (2011) Effect of nano–ZnO particle suspension on growth of Mung (Vigna radiata) and Gram (Cicer arietinum) seedling using plant agar method. J Nanotechnol 1:1–7
Man W, Kwong H, Lam WWY, Xiang J, Wong T, Lam W, Wong W, Peng S, Lau T (2008) General synthesis of (salen) ruthenium (III) complexes via N‑N coupling of (salen) ruthenium (VI) nitrides. Inorg Chem 47(13):5936–5944
Mazumdar H, Ahmed GU (2011) Phytotoxicity effect of silver nanoparticles on Oryza sativa. J Chem Technol Res 3:1494–1500
Parashar V, Parashar R, Sharma B, Pandey AC (2009) Parthenium leaf extract mediated synthesis of silver nanoparticle: a novel approach for weed utilization. Dig J Nanomater Biostruct 4:45–50
Parveen A, Rao S (2015) Effect of nano silver on seed germination and seedling growth in Pennisetum glaucum. J Cluster Sci 26:693–701
Pessarakli M (2014) Handbook of plant and crop physiology. CRCPress, USA
Pourmorad F, Hosseinimehr SJ, Shahabimajd N (2006) Antioxidant activity, phenol and flavonoid contents of some selected Iranian medicinal plants. Afr J Biotechnol 5:1142–1145
Prakash P, Gnanaprakasam P, Emmanuel R, Arokiyaraj S, Saravanan M (2013) Green synthesis of silver nanoparticles from leaf extract of Mimusops elengi L. for enhanced antibacterial activity against multi drug resistant clinical isolates. Colloids Surf B Biointerfaces 108:255–259
Quadros ME, Marr LC (2010) Environmental and human health risks of aerosolized silver nanoparticles. J Air Waste Manag 60:770–781
Rathore SS, Chaudhary DR, Boricha GN, Ghosh A, Bhatt BP, Zodape ST, Patoli JS (2009) Effect of seaweed extract on the growth, yield and nutrient uptake of soybean (Glycine max) under rainfed conditions. S Afr J Bot 75:351–355
Sakihama Y, Cohen MF, Grace SC, Yamasaki H (2002) Plant phenolic antioxidant and prooxidant activities: phenolics-induced oxidative damage mediated by metals in plants. Toxicology 177:67–80
Salama HMH (2012) Effects of silver nanoparticles in some crop plants, common bean (Phaseolus vulgaris L.) and corn (Zea mays L.). Int Res J Biotechnol 3:190–197
Shankar SS, Rai A, Ahmad A, Sastry M (2004) Rapid synthesis of Au, Ag, and bimetallic Au core-Ag shell nanoparticles using Neem (Azadirachta indica) leaf broth. J Colloid Interface Sci 275:496–502
Sharma P, Bhatt D, Zaidi MG, Saradhi PP, Khanna PK, Arora S (2012) Silver nanoparticle-mediated enhancement in growth and antioxidant status of Brassica juncea. Appl Biochem Biotechnol 167:2225–2233
Sharma VK, Yngard RA, Lin Y (2009) Silver nanoparticles: green synthesis and their antimicrobial activities. Adv Colloid Interface Sci 145:83–96
Shelar GB, Chavan AM (2015) Myco-synthesis of silver nanoparticles from Trichoderma harzianum and its impact on germination status of oil seed. Biolife 3:109–113
Stampoulis D, Sinha SK, White JC (2009) Assay-dependent phytotoxicity of nanoparticles to plants. Environ Sci Technol 43:9473–9479
Vannini C, Domingo G, Onelli E, Prinsi B, Marsoni M, Espen L, Bracale M (2013) Morphological and proteomic responses of Eruca sativa exposed to silver nanoparticles or silver nitrate. Plos One 8:e68752
Wijnhoven SWP, Peijnenburg WJGM, Herberts CA, Hagens WI, Oomen AG, Heugens EHW, Roszek B, Bisschops J, Gosens I, Van de Meent D et al (2009) Nanosilver—a review of available data and knowledge gaps in human and environmental risk assessment. Nanotechnology 3:109–138
Zilberberg L, Mitlin S, Shankar H, Asscher M (2015) Buffer layer assisted growth of Ag nanoparticles in titania thin films. J Phys Chem C 119:28979–28991
Zou J, Xu T, Hou B, Wu D, Sun Y (2007) Controlled growth of silver nanoparticles in a hydrothermal process. Chin Particuol 5:206–212
Acknowledgements
We are thankful to IST Islamabad for providing scanning electron microscopy and x‑ray diffraction analysis and QAU Islamabad for UV-vis spectroscopy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
H. Sharif, A. Mehmood, A. Ulfat, K.S. Ahmad, I. Hussain and R.T. Khan declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Sharif, H., Mehmood, A., Ulfat, A. et al. Environmentally Sustainable Production of Silver Nanoparticles and Their Effect on Glycine max L. Seedlings. Gesunde Pflanzen 73, 95–103 (2021). https://doi.org/10.1007/s10343-020-00532-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10343-020-00532-4