Abstract
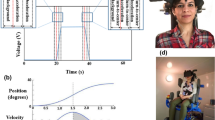
Vection is defined as an illusory self-motion sensation induced in stationary observers that can be experienced in a real/virtual world. Vection, as a result of immersion in virtual reality (VR) environments, can subsequently lead to a sense of inability to maintain postural control and cause cybersickness symptoms. The multisensory integration of visual and vestibular (balance) information plays a vital role in vection. The etiology of vection perception, as well as, the vestibular response change while experiencing vection is poorly understood. This study explores vestibular response change following vection in 20 individuals (10 females, 26.45 ± 4.40 (SD) years). Vection was induced in participants using an immersive VR roller-coaster. The vestibular response was measured simultaneously using a noninvasive method called Electrovestibulography (EVestG). The detected field potentials and the time intervals between the field potentials were extracted from the recorded EVestG signals corresponding to four segments of the VR roller-coaster trajectory namely Stationary, Up movement, Down movement, and slopes and turns (Mix). The results show that the Stationary segment is significantly different (P < 0.05) from other dynamic segments when the average field potential of the right and left ear are subtracted. Furthermore, the Stationary segment shows longer time intervals between field potentials compared to those of the other segments in the right ear. These observations suggest that the combined effect of the visually induced sensation of self-motion together with a concurrent/co-occurring stress/anxiety factor can affect the vestibular activity in an excitatory way. Increased excitatory vestibular activity implies increased feeling of imbalance and more likelihood of experiencing cybersickness by the participants.








Similar content being viewed by others
Abbreviations
- EVestG:
-
Electrovestibulography
- EOG:
-
Electrooculography
- EEG:
-
Electroencephalography
- EMG:
-
Electromyography
- VR:
-
Virtual reality
- CBF:
-
Cerebral blood flow
- LGN:
-
Lateral geniculate nucleus
- MT:
-
Middle temporal/V5 region
- mPFC:
-
Medial prefrontal cortex
References
Ashiri M, Lithgow B, Suleiman A et al (2020) Differences between physical vs. virtual evoked vestibular responses. Ann Biomed Eng. https://doi.org/10.1007/s10439-019-02446-3
Berntson GG, Norman GJ, Bechara A et al (2011) The insula and evaluative processes. Psychol Sci 22:80–86. https://doi.org/10.1177/0956797610391097
Blakley B, Dastgheib ZA, Lithgow B, Moussavi Z (2014) Preliminary report: neural firing patterns specific for Meniere’s disease. J Otolaryngol Head Neck Surg 43:52. https://doi.org/10.1186/s40463-014-0052-4
Brandt T, Bartenstein P, Janek A, Dieterich M (1998) Reciprocal inhibitory visual-vestibular interaction. Visual motion stimulation deactivates the parieto-insular vestibular cortex. Brain 121:1749–1758. https://doi.org/10.1093/brain/121.9.1749
Brown DJ, Patuzzi RB (2010) Evidence that the compound action potential (CAP) from the auditory nerve is a stationary potential generated across dura mater. Hear Res 267:12–26. https://doi.org/10.1016/j.heares.2010.03.091
Bruder GE, Alvarenga J, Abraham K et al (2016) Brain laterality, depression and anxiety disorders: new findings for emotional and verbal dichotic listening in individuals at risk for depression. Laterality 21:525–548. https://doi.org/10.1080/1357650X.2015.1105247
Burdess C (1996) The vestibulo-ocular reflex: computation in the cerebellar flocculus. University of Edinburgh, Edinburgh
Carmeli E (2015) Anxiety in the elderly can be a vestibular problem. Front Public Heal 3:216. https://doi.org/10.3389/fpubh.2015.00216
Cloherty SL, Mustari MJ, Rosa MGP, Ibbotson MR (2010) Effects of saccades on visual processing in primate MSTd. Vis Res 50:2683–2691. https://doi.org/10.1016/j.visres.2010.08.020
Cortes C, Galindo F, Galicia S et al (2013) Excitatory actions of GABA in developing chick vestibular afferents: effects on resting electrical activity. Synapse 67:374–381. https://doi.org/10.1002/syn.21646
Coubard OA, Urbanski M, Bourlon C, Gaumet M (2014) Educating the blind brain: a panorama of neural bases of vision and of training programs in organic neurovisual deficits. Front Integr Neurosci 8:1–13. https://doi.org/10.3389/fnint.2014.00089
DeAngelis GC, Angelaki DE (2012) Visual–vestibular integration for self-motion perception. CRC Press/Taylor & Francis, Boca Raton
Dieterich M, Bucher SF, Seelos KC, Brandt T (1998) Horizontal or vertical optokinetic stimulation activates visual motion-sensitive, ocular motor and vestibular cortex areas with right hemispheric dominance. An fMRI study. Brain 121:1479–1495. https://doi.org/10.1093/brain/121.8.1479
Dieterich M, Bense S, Stephan T et al (2003) FMRI signal increases and decreases in cortical areas during small-field optokinetic stimulation and central fixation. Exp Brain Res 148:117–127. https://doi.org/10.1007/s00221-002-1267-6
Euston DR, Gruber AJ, McNaughton BL (2012) The role of medial prefrontal cortex in memory and decision making. Neuron 76:1057–1070
Fernández C, Goldberg JM (1976) Physiology of peripheral neurons innervating otolith organs of the squirrel monkey. III. Response dynamics. J Neurophysiol 39:996–1008
Finke RA (1986) Mental imagery and the visual system of the same neural processes in the human visual system. Sci Am 254:88–95
Galotti KM (2017) Cognitive psyghology in and out of the laboratory. Sage Publications
Gerson AD, Parra LC, Sajda P (2006) Cortically coupled computer vision for rapid image search. IEEE Trans Neural Syst Rehabil Eng 14:174–179. https://doi.org/10.1109/TNSRE.2006.875550
Goldberg JM, Fernández C (1980) Efferent vestibular system in the squirrel monkey: anatomical location and influence on afferent activity. J Neurophysiol 43:986–1025. https://doi.org/10.1152/jn.1980.43.4.986
Goldberg JM, Wilson VJ, Angelaki DE, Cullen KE, Fukushima K, Buttner-Ennever J (2012) The vestibular system: a sixth sense. Oxford University Press
Goodale MA, Milner AD (1992) Separate visual pathways for perception and action. Trends Neurosci 15:20–25. https://doi.org/10.1016/0166-2236(92)90344-8
Gurvich C, Maller JJ, Lithgow B et al (2013) Vestibular insights into cognition and psychiatry. Brain Res 1537:244–259. https://doi.org/10.1016/j.brainres.2013.08.058
Hettinger LJ, Berbaum KS, Kennedy RS et al (1990) Vection and simulator sickness. Mil Psychol 2:171–181. https://doi.org/10.1207/s15327876mp0203_4
Hoaglin DC, Iglewicz B (1987) Fine-tuning some resistant rules for outlier labeling. J Am Stat Assoc 82:1147. https://doi.org/10.2307/2289392
Huang D, Mao Y, Chen P, Li L (2014) Virtual reality training improves balance function. Neural Regen Res 9:1628. https://doi.org/10.4103/1673-5374.141795
Itō M (2012) The cerebellum: brain for an implicit self. FT Press, Upper Saddle River
Kennedy RS, Lane EN (1993) Simulator sickness questionnaire: an enhanced method for quantifying simulator sickness. Int J Aviat Psychol 33:203–220
Kim O, Pang Y, Kim JH (2019) The effectiveness of virtual reality for people with mild cognitive impairment or dementia: a meta-analysis. BMC Psychiatry 19:219. https://doi.org/10.1186/s12888-019-2180-x
Kleinschmidt A (2002) Neural correlates of visual-motion perception as object- or self-motion. Neuroimage 16:873–882. https://doi.org/10.1006/nimg.2002.1181
Krueger C, Tian L (2004) A comparison of the general linear mixed model and repeated measures ANOVA using a dataset with multiple missing data points. Biol Res Nurs 6:151–157. https://doi.org/10.1177/1099800404267682
LaViola JJ (2000) A discussion of cybersickness in virtual environments. ACM SIGCHI Bull 32:47–56. https://doi.org/10.1145/333329.333344
Liao YY, Tseng HY, Lin YJ et al (2020) Using virtual reality-based training to improve cognitive function, instrumental activities of daily living and neural efficiency in older adults with mild cognitive impairment. Eur J Phys Rehabil Med 56:47–57. https://doi.org/10.23736/S1973-9087.19.05899-4
Lithgow B (2012) A methodology for detecting field potentials from the external ear canal: NEER and EVestG. Ann Biomed Eng 408:1835–1850. https://doi.org/10.1007/s10439-012-0526-3
Lithgow BJ, Shoushtarian M (2017) Journal of the neurological sciences Parkinson’ s disease: disturbed vestibular function and levodopa. J Neurol Sci 353:49–58. https://doi.org/10.1016/j.jns.2015.03.050
Lithgow BJ, Garrett AL, Moussavi ZM et al (2015) Major depression and electrovestibulography. World J Biol Psychiatry 16(5):334–350. https://doi.org/10.3109/15622975.2015.1014410
Lithgow BJ, Moussavi Z, Gurvich C et al (2018) Bipolar disorder in the balance. Eur Arch Psychiatry Clin Neurosci. https://doi.org/10.1007/s00406-018-0935-x
Longstaff A (2005) Instant notes in neuroscience (BIOS instant notes), second edi. Taylor & Francis, Abingdon
Mallinson A (2011) Visual vestibular mismatch. Université Henri Poincaré, Doctoral dissertation
Marlinsky VV (1995) The effect of somatosensory stimulation on second-order and efferent vestibular neurons in the decerebrate decerebellate guinea-pig. Neuroscience 69:661–669. https://doi.org/10.1016/0306-4522(95)00231-7
Öhman A (2005) The role of the amygdala in human fear: automatic detection of threat. Psychoneuroendocrinology 30:953–958. https://doi.org/10.1016/j.psyneuen.2005.03.019
Oman CM (1989) Sensory conflict in motion sickness: an observer theory approach. NASA, Ames Res Center, Spat Displays Spat Instruments 1–16
Palmisano S, Allison RS, Schira MM, Barry RJ (2015) Future challenges for vection research: definitions, functional significance, measures, and neural bases. Front Psychol 6:193
Phan KL, Wager T, Taylor SF, Liberzon I (2002) Functional neuroanatomy of emotion: a meta-analysis of emotion activation studies in PET and fMRI. Neuroimage 16:331–348. https://doi.org/10.1006/NIMG.2002.1087
Pisella L, Alahyane N, Blangero A et al (2011) Right-hemispheric dominance for visual remapping in humans. Philos Trans R Soc B Biol Sci 366:572–585. https://doi.org/10.1098/rstb.2010.0258
Prakriti Trivedi DNB (2017) Effect of left and right hemisphere of brain in both eye open and close state on minimum power values and frequency of alpha wave activity. Int J Sci Eng Technol Res 6:2278–7798. https://doi.org/10.3389/fnins.2016.00584
Reschke MF, Good EF, Clément GR (2017) Neurovestibular symptoms in astronauts immediately after space shuttle and international space station missions. OTO Open 1:2473974X1773876. https://doi.org/10.1177/2473974x17738767
Saman Y, Bamiou DE, Gleeson M, Dutia MB (2012a) Interactions between stress and vestibular compensation—a review. Front Neurol. https://doi.org/10.3389/fneur.2012.00116
Saman Y, Bamiou DE, Gleeson M, Dutia MB (2012b) Interactions between stress and vestibular compensation—a review. Front Neurol 3:116. https://doi.org/10.3389/fneur.2012.00116
Sharpe J, Wong AM (2005) Anatomy and physiology of ocular motor systems. Walsh Hoyt’s Clin Neuro-Ophthalmol 16:809–885
Shin LM, Liberzon I (2010) The neurocircuitry of fear, stress, and anxiety disorders. Neuropsychopharmacology 35:169–191. https://doi.org/10.1038/npp.2009.83
Shinder ME, Taube JS (2010) Differentiating ascending vestibular pathways to the cortex involved in spatial cognition. J Vestib Res Equilib Orientat 20:3–23. https://doi.org/10.3233/VES-2010-0344
Simes RJ (1986) An improved Bonferroni procedure for multiple tests of significance. Biometrika 73:751–754
Smart LJ, Stoffregen TA, Bardy BG (2002) Visually induced motion sickness predicted by postural instability. Hum Factors 44:451–465
Srinivasan V (1989) The pineal gland: its physiological and pharmacological role. Ind J Physiol Pharmac 33(4)
Suleiman A, Lithgow B, Mansouri B, Moussavi Z (2018) Investigating the validity and reliability of Electrovestibulography (EVestG) for detecting post-concussion syndrome (PCS) with and without comorbid depression. Sci Rep 8:14495. https://doi.org/10.1038/s41598-018-32808-1
Szirmai A (2011) Anxiety in vestibular disorders. Anxiety Relat Disord 1:191
Teather RJ (2008) Comparing 2D and 3D direct manipulation interfaces. York University
Thorpe S, Fize D, Marlot C (1996) Speed of processing in the human visual system. Nature 381:520–522. https://doi.org/10.1038/381520a0
Väljamäe A, Smart LJ, Keshavarz B et al (2015) Vection and visually induced motion sickness: how are they related? Front Psychol. https://doi.org/10.3389/fpsyg.2015.00472
Van Essen D, Lewis J, Drury H (2001) Mapping visual cortex in monkeys and humans using surface-based atlases. Vis Res 41:1359–1378
White BJ, Kan JY, Levy R et al (2017) Superior colliculus encodes visual saliency before the primary visual cortex. Proc Natl Acad Sci U S A 114:9451–9456. https://doi.org/10.1073/pnas.1701003114
Yamanaka T (1994) Excitatory effects of glucocorticoids on neuronal activity in the medial vestibular nucleus–mediation by glucocorticoid receptor on the membrane. Nihon Jibiinkoka Gakkai Kaiho 97:855–867
Zajonc TP, Roland PS (2005) Vertigo and motion sickness. Part I: vestibuiar anatomy and physiology. ENT J 84:581–584
Acknowledgements
This study was partly supported by the Natural science and engineering research council (NSERC) of Canada as well as Mitacs through the Mitacs Accelerate program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflicts of interest and disclose no affiliations with or involvement in any organization or entity that could potentially bias the subject matter or materials discussed in this manuscript. Figure 2c is the author (MA) who has agreed to its publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
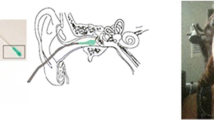
Appendix 1: Transmission of optical information to the vestibular periphery
Human eyes are intricate apparatus that include a number of different structures working together to provide us the visual representation of the surrounding environment. The retina is the light-sensitive component of the eye containing layers of photoreceptors and different cells (amacrine, bipolar, horizontal). In the retina, light photons are converted to electrical signals (following chemical changes in the pigment of the photoreceptors) that are then carried to the optic nerves (axons of ganglion cells). Passing through the optic nerves, these electrical signals reach the optic tract where four major projections diverge; they are the lateral geniculate nucleus (LGN) in the thalamus, superior colliculus, pretectum of the midbrain, and suprachiasmatic nucleus of the hypothalamus (Fig. 1). From these regions, visual information projects to the vestibular nuclei through a number of different neural pathways (Fig. 1). These pathways are different in terms of visual processing time and function (e.g., responsible for elementary and complex visual function). The retino-cortical pathway, through the LGN, projects to the visual cortex wherein delicate visual processing such as the perception of emotions is performed. Based on the results of PET, fMRI and lesion studies (Van Essen et al. 2001), Brodmann’s areas 17–21, 37, 39, and 7 of the visual cortex have been reported to contribute to the sensation of self-motion. These areas send fibers to the cerebellum and more particularly the vestibulo-cerebellum (the flocculus and that part of the vermis connected to it) which have a bi-directional connection to the vestibular nuclei (Burdess 1996). The vestibulocerebellar region receives inputs from the vestibular nuclei and the primary vestibular afferents and project efferents to the vestibular nuclei (Burdess 1996). The processing time through retino-cortical pathway is about 100–150 ms which is longer than that of the other three subcortical visual pathways: the retino-colliculus, the retino-tectal, and the retino hypothalamic pathways (Thorpe et al. 1996; Gerson et al. 2006). Short-latency retino-colliculus pathways involved in primary visual processing tasks, e.g., motion detection and eye movements, has direct connection to the vestibular nuclei, which in turn, projects fibers to extraocular muscles through the abducens, trochlear, and oculomotor nuclei (Fernández and Goldberg 1976); from these regions, three types of voluntary eye movements including smooth pursuit (i.e., tracking a moving object), saccade (i.e., rapid gaze shift toward the object of interest) and vergence (simultaneous movement of both eyes in opposite directions to attain binocular vision) can be controlled (Ito 2012). Processing of the visual information occurs approximately within 80 ms through the retino-collicular pathway (White et al. 2017). The third major projection from the optic tract that sends neurons to the vestibular nuclei is the pretectal area, which is involved in pupillary eye reflex (PLR) and accommodation. Other projections from pretectal areas are to the thalamus, subthalamus, superior colliculus and vestibulo-cerebellum. The last major split originating from the optic tract is the suprachiasmatic nucleus, involved in the regulation of neuronal and hormonal activities, which sends fibers to the hypothalamic nuclei and pineal gland and is responsible for controlling circadian rhythms, reproduction, and human mood disorders (Srinivasan 1989). All these regions, directly or indirectly, have some connections with the vestibular nuclei. Vestibular nuclei send fibers via the efferent vestibular system toward the vestibular periphery, where motion-sensitive sensors (hair cells of semicircular canals and otolith organ) exist.
Appendix 2: SSQ results

Rights and permissions
About this article
Cite this article
Ashiri, M., Lithgow, B., Suleiman, A. et al. Quantitative measures of the visually evoked sensation of body movement in space (Vection) using Electrovestibulography (EVestG). Virtual Reality 25, 731–744 (2021). https://doi.org/10.1007/s10055-020-00488-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10055-020-00488-w