Abstract
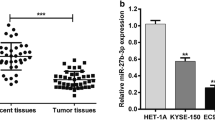
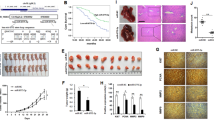
Esophageal squamous cell carcinoma (ESCC) is one of the most frequent malignancies worldwide. miR-193a-3p acts as an oncogene or tumor suppressor in different cancers. However, the functional role and regulatory mechanism of miR-193a-3p in ESCC remain to be elucidated. Our results demonstrated that miR-193a-3p expression was significantly upregulated and associated with advanced TNM stage, recurrence, and poor prognosis in ESCC patients. miR-193-3p targeted ALKBH5 and suppressed its expression. ALKBH5 inhibited miR-193a-3p expression in turn. ALKBH5 affected the primary miR-193a-3p processing by negatively regulating its m6A modification. These findings suggested a positive feedback regulation between miR-193a-3p and ALKBH5 in ESCC cells. Moreover, the functional assays indicated that the miR-193-3p-ALKBH5 feedback loop promoted the proliferation, migration and invasion ability of ESCC cells in vitro, and facilitated tumor growth and metastasis in vivo. Collectively, our current study identified a novel positive feedback regulation between miR-193a-3p and ALKBH5 in ESCC, which may be helpful to gain insight into ESCC pathogenesis and provide novel therapeutic target for ESCC.






Similar content being viewed by others
Availability of data and materials
The data and materials used in this study are available.
Abbreviations
- EC:
-
Esophageal cancer
- ESCC:
-
Esophageal squamous cell carcinoma
- miRNAs:
-
MicroRNAs
- 3′UTR:
-
3′-Untranslated region
- m6A:
-
N6-methyladenosine
- METTL3:
-
Methyltransferase-like 3
- ALKBH5:
-
AlkB homolog 5
- FTO:
-
Fat mass and obesity–associated protein
- MeRIP:
-
Methylated RNA immunoprecipitation
- qRT-PCR:
-
Quantitative Real-time PCR
References
Gupta B, Kumar N. Worldwide incidence, mortality and time trends for cancer of the oesophagus. Eur J Cancer Prev. 2017;26(2):107–18. https://doi.org/10.1097/CEJ.0000000000000249.
Huang FL, Yu SJ. Esophageal cancer: Risk factors, genetic association, and treatment. Asian J Surg. 2018;41(3):210–5. https://doi.org/10.1016/j.asjsur.2016.10.005.
Krol J, Loedige I, Filipowicz W. The widespread regulation of microRNA biogenesis, function and decay. Nat Rev Genet. 2010;11(9):597–610. https://doi.org/10.1038/nrg2843.
Hayes J, Peruzzi PP, Lawler S. MicroRNAs in cancer: biomarkers, functions and therapy. Trends Mol Med. 2014;20(8):460–9. https://doi.org/10.1016/j.molmed.2014.06.005.
Eichelmann AK, Matuszcak C, Lindner K, Haier J, Hussey DJ, Hummel R. Complex role of miR-130a-3p and miR-148a-3p balance on drug resistance and tumor biology in esophageal squamous cell carcinoma. Sci Rep. 2018;8(1):17553. https://doi.org/10.1038/s41598-018-35799-1.
Huang H, Xu Y, Guo Z, Chen X, Ji S, Xu Z. MicroRNA-133b inhibits cell proliferation and promotes apoptosis by targeting cullin 4B in esophageal squamous cell carcinoma. Exp Ther Med. 2018;15(4):3743–50. https://doi.org/10.3892/etm.2018.5906.
Park M, Yoon HJ, Kang MC, Kwon J, Lee HW. MiR-338-5p enhances the radiosensitivity of esophageal squamous cell carcinoma by inducing apoptosis through targeting survivin. Sci Rep. 2017;7(1):10932. https://doi.org/10.1038/s41598-017-10977-9.
Wang SS, Huang ZG, Wu HY, He RQ, Yang LH, Feng ZB, et al. Downregulation of miR-193a-3p is involved in the pathogenesis of hepatocellular carcinoma by targeting CCND1. PeerJ. 2020;8:e8409. https://doi.org/10.7717/peerj.8409.
Kong L, Wei Q, Hu X, Chen L, Li J. miR-193a-3p promotes radio-resistance of nasopharyngeal cancer cells by targeting SRSF2 gene and hypoxia signaling pathway. Med Sci Monit Basic Res. 2019;25:53–62. https://doi.org/10.12659/MSMBR.914572.
Yi Y, Chen J, Jiao C, Zhong J, Song Z, Yu X, et al. Upregulated miR-193a-3p as an oncogene in esophageal squamous cell carcinoma regulating cellular proliferation, migration and apoptosis. Oncol Lett. 2016;12(6):4779–84. https://doi.org/10.3892/ol.2016.5229.
Meng F, Qian L, Lv L, Ding B, Zhou G, Cheng X, et al. miR-193a-3p regulation of chemoradiation resistance in oesophageal cancer cells via the PSEN1 gene. Gene. 2016;579(2):139–45. https://doi.org/10.1016/j.gene.2015.12.060.
Roundtree IA, Evans ME, Pan T, He C. Dynamic RNA modifications in gene expression regulation. Cell. 2017;169(7):1187–200. https://doi.org/10.1016/j.cell.2017.05.045.
Ma JZ, Yang F, Zhou CC, Liu F, Yuan JH, Wang F, et al. METTL14 suppresses the metastatic potential of hepatocellular carcinoma by modulating N(6) -methyladenosine-dependent primary MicroRNA processing. Hepatology. 2017;65(2):529–43. https://doi.org/10.1002/hep.28885.
Han J, Wang JZ, Yang X, Yu H, Zhou R, Lu HC, et al. METTL3 promote tumor proliferation of bladder cancer by accelerating pri-miR221/222 maturation in m6A-dependent manner. Mol Cancer. 2019;18(1):110. https://doi.org/10.1186/s12943-019-1036-9.
Chen Y, Zhao Y, Chen J, Peng C, Zhang Y, Tong R, et al. ALKBH5 suppresses malignancy of hepatocellular carcinoma via m(6)A-guided epigenetic inhibition of LYPD1. Mol Cancer. 2020;19(1):123. https://doi.org/10.1186/s12943-020-01239-w.
Li J, Xie H, Ying Y, Chen H, Yan H, He L, et al. YTHDF2 mediates the mRNA degradation of the tumor suppressors to induce AKT phosphorylation in N6-methyladenosine-dependent way in prostate cancer. Mol Cancer. 2020;19(1):152. https://doi.org/10.1186/s12943-020-01267-6.
Yang X, Zhang S, He C, Xue P, Zhang L, He Z, et al. METTL14 suppresses proliferation and metastasis of colorectal cancer by down-regulating oncogenic long non-coding RNA XIST. Mol Cancer. 2020;19(1):46. https://doi.org/10.1186/s12943-020-1146-4.
He L, Li H, Wu A, Peng Y, Shu G, Yin G. Functions of N6-methyladenosine and its role in cancer. Mol Cancer. 2019;18(1):176. https://doi.org/10.1186/s12943-019-1109-9.
Lin M, Zhang Z, Gao M, Yu H, Sheng H, Huang J. MicroRNA-193a-3p suppresses the colorectal cancer cell proliferation and progression through downregulating the PLAU expression. Cancer Manag Res. 2019;11:5353–63. https://doi.org/10.2147/CMAR.S208233.
Zheng G, Dahl JA, Niu Y, Fedorcsak P, Huang CM, Li CJ, et al. ALKBH5 is a mammalian RNA demethylase that impacts RNA metabolism and mouse fertility. Mol Cell. 2013;49(1):18–29. https://doi.org/10.1016/j.molcel.2012.10.015.
Zhang S, Zhao BS, Zhou A, Lin K, Zheng S, Lu Z, et al. m(6)A demethylase ALKBH5 maintains tumorigenicity of glioblastoma stem-like cells by sustaining FOXM1 expression and cell proliferation program. Cancer Cell. 2017;31(4):591–606. https://doi.org/10.1016/j.ccell.2017.02.013.
Jin D, Guo J, Wu Y, Yang L, Wang X, Du J, et al. m(6)A demethylase ALKBH5 inhibits tumor growth and metastasis by reducing YTHDFs-mediated YAP expression and inhibiting miR-107/LATS2-mediated YAP activity in NSCLC. Mol Cancer. 2020;19(1):40. https://doi.org/10.1186/s12943-020-01161-1.
Nagaki Y, Motoyama S, Yamaguchi T, Hoshizaki M, Sato Y, Sato T, et al. m(6) A demethylase ALKBH5 promotes proliferation of esophageal squamous cell carcinoma associated with poor prognosis. Genes Cells. 2020. https://doi.org/10.1111/gtc.12792.
Acknowledgements
The study was supported by Natural Science Fund of Luoyang Central Hospital Affiliated to Zhengzhou University.
Funding
The study was supported by Natural Science Fund of Luoyang Central Hospital Affiliated to Zhengzhou University.
Author information
Authors and Affiliations
Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no competing financial interests.
Ethical approval
The Ethics Committee of the Luoyang Central Hospital Affiliated to Zhengzhou University approved the study protocols and the use of these tissue samples (approval number: 201908–06) in accordance with the Declaration of Helsinki. The animal studies were approved by the Institutional Animal Care and Use Committee of the Luoyang Central Hospital Affiliated to Zhengzhou University.
Informed consent
The informed consent was obtained from all patients in this research. The protocol of this study was approved by the Ethics Committee of Luoyang Central Hospital Affiliated to Zhengzhou University and carried out in accordance with the World Medical Association Declaration of Helsinki.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
13577_2020_458_MOESM1_ESM.tif
Supplemental Figure 1 FTO does not affect miR-193a-3p expression. A. KYSE-150 and ECA109 cells were transfected with negative control or FTO siRNAs, and the FTO mRNA expression was detected by qRT-PCR. B. KYSE-150 and ECA109 cells were transfected with negative control or FTO siRNAs, and the miR-193a-3p expression was detected by qRT-PCR. **p<0.01. (TIF 1102 KB)
13577_2020_458_MOESM2_ESM.tif
Supplemental Figure The knockdown efficacy of siRNA against METTL3, METTL14 or WTAP in ESCC cells was determined by qRT-PCR (TIF 1198 KB)
13577_2020_458_MOESM3_ESM.jpg
Supplemental Figure 3 The correlation between ALKBH5 and prognosis of Esophageal carcinoma patients from TCGA database was analyzed GEPIA online tool (JPG 55 KB)
Rights and permissions
About this article
Cite this article
Xue, J., Xiao, P., Yu, X. et al. A positive feedback loop between AlkB homolog 5 and miR-193a-3p promotes growth and metastasis in esophageal squamous cell carcinoma. Human Cell 34, 502–514 (2021). https://doi.org/10.1007/s13577-020-00458-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13577-020-00458-z