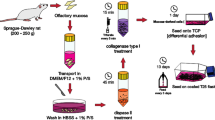
In this work, an optimal protocol was developed for obtaining adhesion culture of neural stem/progenitor cells (NSPC) of rat olfactory mucosa. During the development of the protocol, the conditions for cell culturing on adhesion substrates fibronectin and laminin in DMEM/F-12 and neurobasal media with the same culture additives were compared. Cell proliferation was maximum during culturing on both substrates in the neurobasal medium. Using the immunofluorescence method, we found that culturing on fibronectin in the neurobasal medium ensured maximum (52.22%) content of nestin-positive cells in comparison with other culturing conditions. The highest percentage of βIII-tubulin-positive cells was detected in cultures growing on fibronectin in the neurobasal medium and in DMEM/F-12 (79.11 and 83.52%, respectively). Culturing in adhesion cultures in the neurobasal medium on fibronectin allowed obtaining cultures enriched with NSPC and neurons differentiating from them in a quantity sufficient for further transplantation. The developed protocol can be recommended for obtaining NPSC from human olfactory mucosa for the treatment of spinal cord injuries.
Similar content being viewed by others
References
Shajmardanova GF, Chelyshev JuA, Lebedev SV, Savchenko EA, Viktorov IV, Karasjov AV, Chehonin VP. Post-traumatic reactions of a rat spinal cord after transplantation of human olfactory mucosa cells. Klet. Transplantol. Tkan. Inzheneriya. 2012;7(2):92-96. Russian.
Borgmann-Winter KE, Rawson NE, Wang HY, Wang H, Macdonald ML, Ozdener MH, Yee KK, Gomez G, Xu J, Bryant B, Adamek G, Mirza N, Pribitkin E, Hahn CG. Human olfactory epithelial cells generated in vitro express diverse neuronal characteristics. Neuroscience. 2009;158(2):642-653.
Carter LA, MacDonald JL, Roskams AJ. Olfactory horizontal basal cells demonstrate a conserved multipotent progenitor phenotype. J. Neurosci. 2004;24(25):5670-5683.
Chen X, Fang H, Schwob JE. Multipotency of purified, transplanted globose basal cells in olfactory epithelium. J. Comp. Neurol. 2004;469(4):457-474.
Dictus C, Tronnier V, Unterberg A, Herold-Mende C. Comparative analysis of in vitro conditions for rat adult neural progenitor cells. J. Neurosci. Methods. 2007;161(2):250-258.
Grossman SD, Rosenberg LJ, Wrathall JR. Temporal-spatial pattern of acute neuronal and glial loss after spinal cord contusion. Exp. Neurol. 2001;168(2):273-282.
Lebedev SV, Karasev AV, Chekhonin VP, Savchenko EA, Viktorov IV, Chelyshev YA, Shaimardanova GF. Study of the efficiency of transplantation of human neural stem cells to rats with spinal trauma: the use of functional load tests and BBB test. Bull. Exp. Biol. Med. 2010;149(3):377-382.
Marshall CT, Guo Z, Lu C, Klueber KM, Khalyfa A, Cooper NG, Roisen FJ. Human adult olfactory neuroepithelial derived progenitors retain telomerase activity and lack apoptotic activity. Brain Res. 2005;1045(1-2):45-56.
Muniswami DM, Kanakasabapathy I, Tharion G. Globose basal cells for spinal cord regeneration. Neural Regen. Res. 2017;12(11):1895-1904.
Murrell W, Féron F, Wetzig A, Cameron N, Splatt K, Bellette B, Bianco J, Perry C, Lee G, Mackay-Sim A. Multipotent stem cells from adult olfactory mucosa. Dev. Dyn. 2005;233(2):496-515.
Othman M, Lu C, Klueber K, Winstead W, Roisen F. Clonal analysis of adult human olfactory neurosphere forming cells. Biotech. Histochem. 2005;80(5-6):189-200.
Pearse DD, Bunge MB. Designing cell- and gene-based regeneration strategies to repair the injured spinal cord. J. Neurotrauma. 2006;23(3-4):438-452.
Reier PJ. Cellular transplantation strategies for spinal cord injury and translational neurobiology. NeuroRx. 2004;1(4):424-451.
Stepanova O, Kulikova T, Valikhov M, Poltavtseva R, Shiryaev A, Masenko V, Chekhonin V, Sukhikh G. Cardiac cells in regenerative processes in patients with heart failure due to ischaemic heart disease. Br. J. Med. Med. Res. 2016;14(7):1-11. doi: https://doi.org/10.9734/BJMMR/2016/23962
Voronova AD, Stepanova OV, Chadin AV, Reshetov IV, Chekhonin VP. The cell therapy in traumatic spinal cord injury. Vestn. Ross. Akad. Med. Nauk. 2016;71(6):420-426.
Voronova AD, Stepanova OV, Valikhov MP, Chadin AV, Semkina AS, Chekhonin VP. Neural stem/progenitor cells of human olfactory mucosa for the treatment of chronic spinal cord injuries. Bull. Exp. Biol. Med. 2020;168(4):538-541.
Viktorov IV, Savchenko EA, Chekhonin VP. Spontaneous neural differentiation of stem cells in culture of human olfactory epithelium. Bull. Exp. Biol. Med.2007;144(4):596-601.
Xiao M, Klueber KM, Guo Z, Lu C, Wang H, Roisen FJ. Human adult olfactory neural progenitors promote axotomized rubrospinal tract axonal reinnervation and locomotor recovery. Neurobiol. Dis. 2007;26(2):363-374.
Xiao M, Klueber KM, Lu C, Guo Z, Marshall CT, Wang H, Roisen FJ. Human adult olfactory neural progenitors rescue axotomized rodent rubrospinal neurons and promote functional recovery. Exp. Neurol. 2005;194(1):12-30.
Yao R, Murtaza M, Velasquez JT, Todorovic M, Rayfield A, Ekberg J, Barton M, St John J. Olfactory ensheathing cells for spinal cord injury: sniffing out the issues. Cell Transplant. 2018;27(6):879-889.
Zhang X, Klueber KM, Guo Z, Lu C, Roisen FJ. Adult human olfactory neural progenitors cultured in defined medium. Exp. Neurol. 2004;186(2):112-123.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Kletochnye Tekhnologii v Biologii i Meditsine, No. 3, pp. 202-208, September, 2020
Rights and permissions
About this article
Cite this article
Stepanova, O.V., Voronova, A.D., Fursa, G.A. et al. Preparation of Adhesion Culture of Neural Stem/Progenitor Cells of the Olfactory Mucosa for the Treatment of Spinal Cord Injuries. Bull Exp Biol Med 170, 158–163 (2020). https://doi.org/10.1007/s10517-020-05023-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-020-05023-0