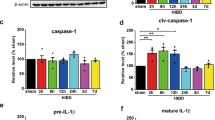
Abstract
Neonatal hypoxic-ischemic encephalopathy (HIE) is one of the main causes of neonatal disability and death. As a derivative of N-acetylserotonin, N-[2-(5-hydroxy-1H-indol-3-yl) ethyl]-2-oxopiperidine-3-carboxamide (HIOC) can easily cross the blood–brain barrier and have a long half-life in the brain. In this study, the hypothesis was verified that HIOC plays a neuroprotective role in the HIE model and its potential mechanism was evaluated. Firstly, an HIE rat model was established to deliver HIOC, revealing that it can reduce cerebral infarction volume, cerebral edema, and neuronal apoptosis. The results of immunofluorescence staining, Western blots and RT-PCR further showed that HIOC could inhibit the activation of the NLRP3 inflammasome and the expression of related proteins. Finally, the activation of the phosphatidylinositol-3-kinase (PI3K)/Akt/nuclear factor erythroid 2-related factor 2 (Nrf2) pathway by HIOC was verified in vitro and in vivo. It was discovered that HIOC could increase the nuclear translocation of Nrf2, and that this induction can be reversed by the PI3K/Akt pathway inhibitor LY294002. In general terms, the neuroprotective effect of HIOC was confirmed in the HIE model, which is related to the activation of the Pi3k/Akt/Nrf2 signal pathway and the inhibition of the NLRP3 inflammasome.





Similar content being viewed by others
Abbreviations
- Akt:
-
Protein kinase B
- DAPI:
-
4′,6-diamidino-2-phenylindole
- HIE:
-
Hypoxic-ischemic encephalopathy
- HIOC:
-
N-[2-(5-hydroxy-1H-indol-3-yl) ethyl]-2-oxopiperidine-3-carboxamide
- HO-1:
-
Heme oxygenase-1
- Iba-1:
-
Ionized calcium binding adapter molecule 1
- IL:
-
Interleukin
- NAS:
-
N-acetylserotonin
- Nrf2:
-
Nuclear factor erythroid 2-related factor 2
- PI3K:
-
Phosphatidylinositol 3-kinase
- TNF-α:
-
Tumor necrosis factor-α
- TTC:
-
2, 3, 5-triphenyltetrazolium chloride
- TUNEL:
-
Terminal deoxynucleotidyl transferase dUTP nick end labeling
References
Millar LJ, Shi L, Hoerder-Suabedissen A, Molnar Z (2017) Neonatal hypoxia ischaemia: mechanisms, models, and therapeutic challenges. Front Cell Neurosci 11:78. https://doi.org/10.3389/fncel.2017.00078
Hassell KJ, Ezzati M, Alonso-Alconada D et al (2015) New horizons for newborn brain protection: enhancing endogenous neuroprotection. Arch Dis Child Fetal Neonatal Ed 100:F541–F552
Herz J, Köster C, Reinboth BS, Dzietko M, Hansen W, Sabir H, van Velthoven C, Bendix I, Felderhoff-Müser U (2018) Interaction between hypothermia and delayed mesenchymal stem cell therapy in neonatal hypoxic-ischemic brain injury. Brain Behav Immun 70:118–130. https://doi.org/10.1016/j.bbi.2018.02.006
Rodríguez M, Valez V, Cimarra C et al (2020) Hypoxic-ischemic encephalopathy and mitochondrial dysfunction: facts, unknowns, and challenges. Antioxid Redox Signal 33:247–262
Gunn AJ, Thoresen M (2015) Animal studies of neonatal hypothermic neuroprotection have translated well in to practice. Resuscitation 97:88–90
Penny TR, Sutherland AE, Mihelakis JG et al (2019) Human umbilical cord therapy improves long-term behavioral outcomes following neonatal hypoxic ischemic brain injury. Front Physiol 10:283
Le K, Chibaatar Daliv E, Wu S, Qian F, Ali AI, Yu D, Guo Y (2019) SIRT1-regulated HMGB1 release is partially involved in TLR4 signal transduction: a possible anti-neuroinflammatory mechanism of resveratrol in neonatal hypoxic-ischemic brain injury. Int Immunopharmacol 75:105779. https://doi.org/10.1016/j.intimp.2019.105779
Li B, Dasgupta C, Huang L, Meng X, Zhang L (2019) MiRNA-210 induces microglial activation and regulates microglia-mediated neuroinflammation in neonatal hypoxic-ischemic encephalopathy. Cell Mol Immunol. https://doi.org/10.1038/s41423-019-0257-6
Li B, Concepcion K, Meng X, Zhang L (2017) Brain-immune interactions in perinatal hypoxic-ischemic brain injury. Prog Neurobiol 159:50–68. https://doi.org/10.1016/j.pneurobio.2017.10.006
Chen CY, Sun WZ, Kang KH, Chou HC, Tsao PN, Hsieh WS, Fu WM (2015) Hypoxic preconditioning suppresses glial activation and neuroinflammation in neonatal brain insults. Mediat Inflamm 2015:632592. https://doi.org/10.1155/2015/632592
Jo EK, Kim JK, Shin DM, Sasakawa C (2016) Molecular mechanisms regulating NLRP3 inflammasome activation. Cell Mol Immunol 13(2):148–159. https://doi.org/10.1038/cmi.2015.95
Kuwar R, Rolfe A, Di L, Xu H, He L, Jiang Y, Zhang S, Sun D (2019) A novel small molecular NLRP3 inflammasome inhibitor alleviates neuroinflammatory response following traumatic brain injury. J Neuroinflammation 16(1):81. https://doi.org/10.1186/s12974-019-1471-y
Gong Z, Pan J, Shen Q, Li M, Peng Y (2018) Mitochondrial dysfunction induces NLRP3 inflammasome activation during cerebral ischemia/reperfusion injury. J Neuroinflammation 15(1):242. https://doi.org/10.1186/s12974-018-1282-6
Chen D, Dixon BJ, Doycheva DM, Li B, Zhang Y, Hu Q, He Y, Guo Z, Nowrangi D, Flores J, Filippov V, Zhang JH, Tang J (2018) IRE1α inhibition decreased TXNIP/NLRP3 inflammasome activation through miR-17-5p after neonatal hypoxic-ischemic brain injury in rats. J Neuroinflammation 15(1):32. https://doi.org/10.1186/s12974-018-1077-9
Chen A, Xu Y, Yuan J (2018) Ginkgolide B ameliorates NLRP3 inflammasome activation after hypoxic-ischemic brain injury in the neonatal male rat. Int J Dev Neurosci 69:106–111. https://doi.org/10.1016/j.ijdevneu.2018.07.004
Yoo JM, Lee BD, Sok DE, Ma JY, Kim MR (2017) Neuroprotective action of N-acetyl serotonin in oxidative stress-induced apoptosis through the activation of both TrkB/CREB/BDNF pathway and Akt/Nrf2/antioxidant enzyme in neuronal cells. Redox Biol 11:592–599. https://doi.org/10.1016/j.redox.2016.12.034
Zhou H, Wang J, Jiang J, Stavrovskaya IG, Li M, Li W, Wu Q, Zhang X, Luo C, Zhou S, Sirianni AC, Sarkar S, Kristal BS, Friedlander RM, Wang X (2014) N-acetyl-serotonin offers neuroprotection through inhibiting mitochondrial death pathways and autophagic activation in experimental models of ischemic injury. J Neurosci 34(8):2967–2978. https://doi.org/10.1523/jneurosci.1948-13.2014
Shen J, Ghai K, Sompol P, Liu X, Cao X, Iuvone PM, Ye K (2012) N-acetyl serotonin derivatives as potent neuroprotectants for retinas. Proc Natl Acad Sci U S A 109(9):3540–3545. https://doi.org/10.1073/pnas.1119201109
Tang J, Hu Q, Chen Y, Liu F, Zheng Y, Tang J, Zhang J, Zhang JH (2015) Neuroprotective role of an N-acetyl serotonin derivative via activation of tropomyosin-related kinase receptor B after subarachnoid hemorrhage in a rat model. Neurobiol Dis 78:126–133. https://doi.org/10.1016/j.nbd.2015.01.009
Qi X, Liu J, Wu J, Bi Y, Han C, Zhang G, Lou M, Lu J, Tang J (2019) Initiating TrkB/Akt signaling cascade preserves blood–brain barrier after subarachnoid hemorrhage in rats. Cell Transplant 28(8):1002–1008. https://doi.org/10.1177/0963689719857649
Lei L, Vollmer Mary K, Ahmad Abdullah S et al (2019) Pretreatment with Korean red ginseng or dimethyl fumarate attenuates reactive gliosis and confers sustained neuroprotection against cerebral hypoxic-ischemic damage by an Nrf2-dependent mechanism. Free Radic Biol Med 131:98–114
Chen H, Burris M, Fajilan A, Spagnoli F, Tang J, Zhang JH (2011) Prolonged exposure to isoflurane ameliorates infarction severity in the rat pup model of neonatal hypoxia-ischemia. Transl Stroke Res 2(3):382–390. https://doi.org/10.1007/s12975-011-0081-5
Vannucci RC, Towfighi J, Vannucci SJ (2004) Secondary energy failure after cerebral hypoxia-ischemia in the immature rat. J Cereb Blood Flow Metab 24:1090–1097
Xu J, Feng Z, Wang X, Xiong Y, Wang L, Ye L, Zhang H (2019) hUC-MSCs exert a neuroprotective effect via anti-apoptotic mechanisms in a neonatal HIE rat model. Cell Transplant 28(12):1552–1559. https://doi.org/10.1177/0963689719874769
Miyasato Y, Yoshizawa T, Sato Y et al (2018) Sirtuin 7 deficiency ameliorates cisplatin-induced acute kidney injury through regulation of the inflammatory response. Sci Rep 8:5927
Liu T, Wang Y, Di C et al (2018) Protective effects of notoginsenoside R1 via regulation of the PI3K-Akt-mTOR/JNK pathway in neonatal cerebral hypoxic-ischemic brain injury. Neurochem Res 43:1210–1226
Lixia Y, Xue W, Chenchen C et al (2019) FGF21 promotes functional recovery after hypoxic-ischemic brain injury in neonatal rats by activating the PI3K/Akt signaling pathway via FGFR1/β-klotho. Exp Neurol 317:34–50
Yafei Z, Yuyun W, Ying L et al (2019) Intrinsic effects of gold nanoparticles on oxygen-glucose deprivation/reperfusion injury in rat cortical neurons. Neurochem Res 44:1549–1566
Luo C, Yang Q, Liu Y, Zhou S, Jiang J, Reiter RJ, Bhattacharya P, Cui Y, Yang H, Ma H, Yao J, Lawler SE, Zhang X, Fu J, Rozental R, Aly H, Johnson MD, Chiocca EA, Wang X (2019) The multiple protective roles and molecular mechanisms of melatonin and its precursor N-acetylserotonin in targeting brain injury and liver damage and in maintaining bone health. Free Radic Biol Med 130:215–233. https://doi.org/10.1016/j.freeradbiomed.2018.10.402
Li X, Wang T, Zhang D, Li H, Shen H, Ding X, Chen G (2018) Andrographolide ameliorates intracerebral hemorrhage induced secondary brain injury by inhibiting neuroinflammation induction. Neuropharmacology 141:305–315. https://doi.org/10.1016/j.neuropharm.2018.09.015
Zhang X, Zhang JH, Chen XY, Hu QH, Wang MX, Jin R, Zhang QY, Wang W, Wang R, Kang LL, Li JS, Li M, Pan Y, Huang JJ, Kong LD (2015) Reactive oxygen species-induced TXNIP drives fructose-mediated hepatic inflammation and lipid accumulation through NLRP3 inflammasome activation. Antioxid Redox Signal 22(10):848–870. https://doi.org/10.1089/ars.2014.5868
Sarkar S, Malovic E, Harishchandra DS et al (2017) Mitochondrial impairment in microglia amplifies NLRP3 inflammasome proinflammatory signaling in cell culture and animal models of Parkinson’s disease. NPJ Parkinsons Dis 3:30
Ye Y, Jin T, Zhang X, Zeng Z, Ye B, Wang J, Zhong Y, Xiong X, Gu L (2019) Meisoindigo protects against focal cerebral ischemia-reperfusion injury by inhibiting NLRP3 inflammasome activation and regulating microglia/macrophage polarization via TLR4/NF-κB signaling pathway. Front Cell Neurosci 13:553. https://doi.org/10.3389/fncel.2019.00553
Hong P, Li FX, Gu RN, Fang YY, Lai LY, Wang YW, Tao T, Xu SY, You ZJ, Zhang HF (2018) Inhibition of NLRP3 inflammasome ameliorates cerebral ischemia-reperfusion injury in diabetic mice. Neural Plast 2018:9163521. https://doi.org/10.1155/2018/9163521
Yuqin Z, Liming F, Huang L et al (2018) Gualou guizhi granule protects against oxidative injury by activating Nrf2/ARE pathway in rats and PC12 cells. Neurochem Res 43:1003–1009
Wang C-Y, Xu Y, Wang X et al (2019) Dl-3-n-butylphthalide inhibits NLRP3 inflammasome and mitigates Alzheimer’s-like pathology via Nrf2-TXNIP-TrX axis. Antioxid Redox Signal 30:1411–1431
Li H, Tang Z, Chu P, Song Y, Yang Y, Sun B, Niu M, Qaed E, Shopit A, Han G, Ma X, Peng J, Hu M, Tang Z (2018) Neuroprotective effect of phosphocreatine on oxidative stress and mitochondrial dysfunction induced apoptosis in vitro and in vivo: involvement of dual PI3K/Akt and Nrf2/HO-1 pathways. Free Radic Biol Med 120:228–238. https://doi.org/10.1016/j.freeradbiomed.2018.03.014
Lee DS, Jeong GS (2016) Butein provides neuroprotective and anti-neuroinflammatory effects through Nrf2/ARE-dependent haem oxygenase 1 expression by activating the PI3K/Akt pathway. Br J Pharmacol 173(19):2894–2909. https://doi.org/10.1111/bph.13569
Lin CC, Lin WN, Cho RL, Yang CC, Yeh YC, Hsiao LD, Tseng HC, Yang CM (2020) Induction of HO-1 by mevastatin mediated via a Nox/ROS-dependent c-Src/PDGFRα/PI3K/Akt/Nrf2/ARE cascade suppresses TNF-α-induced lung inflammation. J Clin Med 9(1). https://doi.org/10.3390/jcm9010226
Zhuang Y, Wu H, Wang X, He J, He S, Yin Y (2019) Resveratrol attenuates oxidative stress-induced intestinal barrier injury through PI3K/Akt-mediated Nrf2 signaling pathway. Oxidative Med Cell Longev 2019:7591840. https://doi.org/10.1155/2019/7591840
Funding
This work was supported by grants from the Central Fund for Guiding Local Science and Technology Development (No. [2016]109) and Natural Science Foundation of Hubei Province (No. WJ2019M184).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare to have no conflict of interest.
Ethical Approval
All animal experiment programs are approved by the Animal Experimental Ethics Committee at Wuhan University and conducted in accordance with the guidance of the Animal Care and use Committee of Renmin Hospital at Wuhan University (Approval number: WDRM 20190315). Efforts were made to minimize the number of rats and their suffering.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Luo, X., Zeng, H., Fang, . et al. N-acetylserotonin Derivative Exerts a Neuroprotective Effect by Inhibiting the NLRP3 Inflammasome and Activating the PI3K/Akt/Nrf2 Pathway in the Model of Hypoxic-Ischemic Brain Damage. Neurochem Res 46, 337–348 (2021). https://doi.org/10.1007/s11064-020-03169-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-020-03169-x