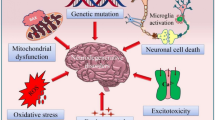
Abstract
Parkinson’s disease (PD) is characterized by progressive degeneration of dopaminergic neurons in the substantia nigra and loss of both motor and non-motor features. Several clinical and preclinical studies have provided evidence that estrogen therapy reduces the risk of PD but have limitations in terms of adverse peripheral effects. Therefore, we examined the potential beneficial effects of the brain-selective estrogen prodrug, 10β, 17β-dihydroxyestra-1,4-dien-3-one (DHED) on nigrostriatal dopaminergic neurodegeneration and behavioral abnormalities in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) mouse model of PD. Wild-type mice were treated with daily subcutaneous injections of DHED (50 and 100 µg/kg) or vehicle for four weeks. To produce PD-like symptoms, mice were injected with MPTP (18 mg/kg in saline; intraperitoneally) four times at 2-hr intervals for one day. After behavioral examination, mice were sacrificed, and the brains were isolated for neurochemical and morphological examinations. MPTP injected mice exhibited loss of dopaminergic neurons and fibers in substantia nigra and striatum respectively, along with impaired motor function at day 7 post MPTP injection. These phenotypes were associated with significantly increased oxidative stress and inflammatory responses in the striatum regions. DHED treatments significantly mitigated behavioral impairments and dopaminergic neurodegeneration induced by MPTP. We further observed that DHED treatment suppressed oxidative stress and inflammation in the striatum of MPTP treated mice when compared to vehicle treated mice. In conclusions, our findings suggest that DHED protects dopaminergic neurons from MPTP toxicity in mouse model of PD and support a beneficial effect of brain-selective estrogen in attenuating neurodegeneration and motor symptoms in PD-related neurological disorders.

Graphical Abstract






Similar content being viewed by others
Data Availability
The data generated and analyzed in this study are available from the corresponding author on reasonable request.
References
Anandhan A, Tamilselvam K, Vijayraja D, Ashokkumar N, Rajasankar S, Manivasagam T (2010) Resveratrol attenuates oxidative stress and improves behaviour in 1 -methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) challenged mice. Ann Neurosci 17:113–119. https://doi.org/10.5214/ans.0972-7531.1017304
Aquilano K, Baldelli S, Ciriolo MR (2014) Glutathione: new roles in redox signaling for an old antioxidant. Front Pharmacol 5:196. https://doi.org/10.3389/fphar.2014.00196
Breydo L, Wu JW, Uversky VN (2012) Alpha-synuclein misfolding and Parkinson’s disease. Biochim Biophys Acta 1822:261–285. https://doi.org/10.1016/j.bbadis.2011.10.002
Butler MJ, Perrini AA, Eckel LA (2020) Estradiol treatment attenuates high fat diet-induced microgliosis in ovariectomized rats. Horm Behav 120:104675. https://doi.org/10.1016/j.yhbeh.2020.104675
Camins A, Pizarro JG, Alvira D, Gutierrez-Cuesta J, de la Torre AV, Folch J, Sureda FX, Verdaguer E, Junyent F, Jordan J, Ferrer I, Pallas M (2010) Activation of ataxia telangiectasia muted under experimental models and human Parkinson’s disease. Cell Mol Life Sci 67:3865–3882. https://doi.org/10.1007/s00018-010-0408-5
Chi L, Ke Y, Luo C, Gozal D, Liu R (2007) Depletion of reduced glutathione enhances motor neuron degeneration in vitro and in vivo. Neuroscience 144:991–1003. https://doi.org/10.1016/j.neuroscience.2006.09.064
Dauer W, Przedborski S (2003) Parkinson’s disease: mechanisms. and models Neuron 39:889–909. https://doi.org/10.1016/s0896-6273(03)00568-3
Dias V, Junn E, Mouradian MM (2013) The role of oxidative stress in Parkinson’s disease. J Parkinsons Dis 3:461–491. https://doi.org/10.3233/JPD-130230
Dringen R (2000) Metabolism and functions of glutathione in brain. Prog Neurobiol 62:649–671. https://doi.org/10.1016/s0301-0082(99)00060-x
Fukai T, Ushio-Fukai M (2011) Superoxide dismutases: role in redox signaling, vascular function, and diseases. Antioxid Redox Signal 15:1583–1606. https://doi.org/10.1089/ars.2011.3999
Gaschler MM, Stockwell BR (2017) Lipid peroxidation in cell death. Biochem Biophys Res Commun 482:419–425. https://doi.org/10.1016/j.bbrc.2016.10.086
Goedert M (2001) Alpha-synuclein and neurodegenerative diseases. Nat Rev Neurosci 2:492–501. https://doi.org/10.1038/35081564
Gonzalez-Hunt CP, Sanders LH (2020) DNA damage and repair in Parkinson’s disease: Recent advances and new opportunities. J Neurosci Res. https://doi.org/10.1002/jnr.24592
Guzman-Martinez L, Maccioni RB, Andrade V, Navarrete LP, Pastor MG, Ramos-Escobar N (2019) Neuroinflammation as a common feature of neurodegenerative. Disorders Front Pharmacol 10:1008. https://doi.org/10.3389/fphar.2019.01008
Hald A, Lotharius J (2005) Oxidative stress and inflammation in Parkinson’s disease: is there a causal link? Exp Neurol 193:279–290. https://doi.org/10.1016/j.expneurol.2005.01.013
Hirsch L, Jette N, Frolkis A, Steeves T, Pringsheim T (2016) The Incidence of Parkinson’s Disease: A Systematic Review and Meta-Analysis. Neuroepidemiology 46:292–300. https://doi.org/10.1159/000445751
Jackson-Lewis V, Jakowec M, Burke RE, Przedborski S (1995) Time course and morphology of dopaminergic neuronal death caused by the neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. Neurodegeneration 4:257–269. https://doi.org/10.1016/1055-8330(95)90015-2
Javed H, Thangavel R, Selvakumar GP, Dubova I, Schwartz N, Ahmed ME, Zaheer S, Kempuraj D, Iyer S, Zaheer A, Khan MM (2020) NLRP3 inflammasome and glia maturation factor coordinately regulate neuroinflammation and neuronal loss in MPTP mouse model of Parkinson’s disease. Int Immunopharmacol 83:106441. https://doi.org/10.1016/j.intimp.2020.106441
Jenner P, Olanow CW (2006) The pathogenesis of cell death in Parkinson’s disease. Neurology 66:S24-36. https://doi.org/10.1212/wnl.66.10_suppl_4.s24
Joers V, Tansey MG, Mulas G, Carta AR (2017) Microglial phenotypes in Parkinson’s disease and animal models of the disease. Prog Neurobiol 155:57–75. https://doi.org/10.1016/j.pneurobio.2016.04.006
Khan MM, Kempuraj D, Thangavel R, Zaheer A (2013) Protection of MPTP-induced neuroinflammation and neurodegeneration by. Pycnogenol Neurochem Int 62:379–388. https://doi.org/10.1016/j.neuint.2013.01.029
Khan MM, Zaheer S, Thangavel R, Patel M, Kempuraj D, Zaheer A (2015) Absence of glia maturation factor protects dopaminergic neurons and improves motor behavior in mouse model of parkinsonism. Neurochem Res 40:980–990. https://doi.org/10.1007/s11064-015-1553-x
Khan MM, Xiao J, Patel D, LeDoux MS (2018) DNA damage and neurodegenerative phenotypes in aged Ciz1 null. mice Neurobiol Aging 62:180–190. https://doi.org/10.1016/j.neurobiolaging.2017.10.014
Khan M, Ullah R, Rehman SU, Shah SA, Saeed K, Muhammad T, Park HY, Jo MH, Choe K, Rutten BPF, Kim MO (2019) 17beta-estradiol modulates sirt1 and halts oxidative stress-mediated cognitive impairment in a male aging mouse model. Cells 8 https://doi.org/10.3390/cells8080928
Lee YH, Cha J, Chung SJ, Yoo HS, Sohn YH, Ye BS, Lee PH (2019) Beneficial effect of estrogen on nigrostriatal dopaminergic neurons in drug-naive postmenopausal Parkinson’s disease. Sci Rep 9:10531. https://doi.org/10.1038/s41598-019-47026-6
Lin MT, Beal MF (2006) Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 443:787–795. https://doi.org/10.1038/nature05292
Mah LJ, El-Osta A, Karagiannis TC (2010) gammaH2AX: a sensitive molecular marker of DNA damage and repair. Leukemia 24:679–686. https://doi.org/10.1038/leu.2010.6
Merchenthaler I, Lane M, Sabnis G, Brodie A, Nguyen V, Prokai L, Prokai-Tatrai K (2016) Treatment with an orally bioavailable prodrug of 17beta-estradiol alleviates hot flushes without hormonal effects in the periphery. Sci Rep 6:30721. https://doi.org/10.1038/srep30721
Merchenthaler I, Lane M, Stennett C, Zhan M, Nguyen V, Prokai-Tatrai K, Prokai L (2020) Brain-selective estrogen therapy prevents androgen deprivation-associated hot flushes in a rat model. Pharmaceuticals (Basel) 13 https://doi.org/10.3390/ph13060119
Milanese C, Cerri S, Ulusoy A, Gornati SV, Plat A, Gabriels S, Blandini F, Di Monte DA, Hoeijmakers JH, Mastroberardino PG (2018) Activation of the DNA damage response in vivo in synucleinopathy models of Parkinson’s disease. Cell Death Dis 9:818. https://doi.org/10.1038/s41419-018-0848-7
Mosley RL, Benner EJ, Kadiu I, Thomas M, Boska MD, Hasan K, Laurie C, Gendelman HE (2006) Neuroinflammation, oxidative stress and the pathogenesis of parkinson’s disease. Clin Neurosci Res 6:261–281. https://doi.org/10.1016/j.cnr.2006.09.006
Ouchi Y, Yagi S, Yokokura M, Sakamoto M (2009) Neuroinflammation in the living brain of Parkinson’s disease. Parkinsonism Relat Disord 15(Suppl 3):S200-204. https://doi.org/10.1016/S1353-8020(09)70814-4
Prokai L, Nguyen V, Szarka S, Garg P, Sabnis G, Bimonte-Nelson HA, McLaughlin KJ, Talboom JS, Conrad CD, Shughrue PJ, Gould TD, Brodie A, Merchenthaler I, Koulen P, Prokai-Tatrai K (2015) The prodrug DHED selectively delivers 17beta-estradiol to the brain for treating estrogen-responsive disorders. Sci Transl Med 7:297ra113. https://doi.org/10.1126/scitranslmed.aab1290
Prokai-Tatrai K, Nguyen V, Prokai L (2018) 10beta,17alpha-Dihydroxyestra-1,4-dien-3-one: a bioprecursor prodrug preferentially producing 17alpha-estradiol in the brain for targeted neurotherapy. ACS Chem Neurosci 9:2528–2533. https://doi.org/10.1021/acschemneuro.8b00184
Przedborski S, Vila M (2003) The 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine mouse model: a tool to explore the pathogenesis of Parkinson’s disease. Ann N Y Acad Sci 991:189–198
Quesada A, Micevych PE (2004) Estrogen interacts with the IGF-1 system to protect nigrostriatal dopamine and maintain motoric behavior after 6-hydroxdopamine lesions. J Neurosci Res 75:107–116. https://doi.org/10.1002/jnr.10833
Ragonese P, D’Amelio M, Callari G, Salemi G, Morgante L, Savettieri G (2006) Age at menopause predicts age at onset of Parkinson’s disease. Mov Disord 21:2211–2214. https://doi.org/10.1002/mds.21127
Rajsombath MM, Nam AY, Ericsson M, Nuber S (2019) Female sex and brain-selective estrogen benefit alpha-synuclein tetramerization and the PD-like motor syndrome in 3K transgenic. Mice J Neurosci 39:7628–7640. https://doi.org/10.1523/JNEUROSCI.0313-19.2019
Rodriguez-Perez AI, Dominguez-Meijide A, Lanciego JL, Guerra MJ, Labandeira-Garcia JL (2013) Inhibition of Rho kinase mediates the neuroprotective effects of estrogen in the MPTP model of Parkinson’s disease. Neurobiol Dis 58:209–219. https://doi.org/10.1016/j.nbd.2013.06.004
Sanders LH, Timothy Greenamyre J (2013) Oxidative damage to macromolecules in human Parkinson disease and the rotenone model Free. Radic Biol Med 62:111–120. https://doi.org/10.1016/j.freeradbiomed.2013.01.003
Sawada H, Ibi M, Kihara T, Urushitani M, Akaike A, Shimohama S (1998) Estradiol protects mesencephalic dopaminergic neurons from oxidative stress-induced neuronal death J Neurosci Res 54:707–719 https://doi.org/10.1002/(SICI)1097-4547(19981201)54:5<707::AID-JNR16>3.0.CO;2-T
Seaton TA, Jenner P, Marsden CD (1996) Mitochondrial respiratory enzyme function and superoxide dismutase activity following brain glutathione depletion in the rat. Biochem Pharmacol 52:1657–1663. https://doi.org/10.1016/s0006-2952(96)00452-2
Shen D, Tian X, Zhang B, Song R (2017) Mechanistic evaluation of neuroprotective effect of estradiol on rotenone and 6-OHDA induced Parkinson’s disease. Pharmacol Rep 69:1178–1185. https://doi.org/10.1016/j.pharep.2017.06.008
Siddiqui MS, Francois M, Fenech MF, Leifert WR (2015) Persistent gammaH2AX: A promising molecular marker of DNA damage and aging. Mutat Res Rev Mutat Res 766:1–19. https://doi.org/10.1016/j.mrrev.2015.07.001
Thakkar R, Wang R, Sareddy G, Wang J, Thiruvaiyaru D, Vadlamudi R, Zhang Q, Brann D (2016) NLRP3 inflammasome activation in the brain after global cerebral ischemia and regulation by 17beta-estradiol oxid. Med Cell Longev 2016:8309031. https://doi.org/10.1155/2016/8309031
Tripanichkul W, Sripanichkulchai K, Finkelstein DI (2006) Estrogen down-regulates glial activation in male mice following 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine intoxication. Brain Res 1084:28–37. https://doi.org/10.1016/j.brainres.2006.02.029
Tripanichkul W, Sripanichkulchai K, Duce JA, Finkelstein DI (2007) 17Beta-estradiol reduces nitrotyrosine immunoreactivity and increases SOD1 and SOD2 immunoreactivity in nigral neurons in male mice following MPTP insult. Brain Res 1164:24–31. https://doi.org/10.1016/j.brainres.2007.05.076
Tsang KL, Ho SL, Lo SK (2000) Estrogen improves motor disability in parkinsonian postmenopausal women with motor fluctuations. Neurology 54:2292–2298. https://doi.org/10.1212/wnl.54.12.2292
Tschiffely AE, Schuh RA, Prokai-Tatrai K, Prokai L, Ottinger MA (2016) A comparative evaluation of treatments with 17beta-estradiol and its brain-selective prodrug in a double-transgenic mouse model of Alzheimer’s disease. Horm Behav 83:39–44. https://doi.org/10.1016/j.yhbeh.2016.05.009
Tschiffely AE, Schuh RA, Prokai-Tatrai K, Ottinger MA, Prokai L (2018) An exploratory investigation of brain-selective estrogen treatment in males using a mouse model of Alzheimer’s disease. Horm Behav 98:16–21. https://doi.org/10.1016/j.yhbeh.2017.11.015
Van Den Eeden SK, Tanner CM, Bernstein AL, Fross RD, Leimpeter A, Bloch DA, Nelson LM (2003) Incidence of Parkinson’s disease: variation by age, gender, and race/ethnicity. Am J Epidemiol 157:1015–1022. https://doi.org/10.1093/aje/kwg068
Vegeto E, Belcredito S, Etteri S, Ghisletti S, Brusadelli A, Meda C, Krust A, Dupont S, Ciana P, Chambon P, Maggi A (2003) Estrogen receptor-alpha mediates the brain antiinflammatory activity of estradiol. Proc Natl Acad Sci U S A 100:9614–9619. https://doi.org/10.1073/pnas.1531957100
Wang D, Yu T, Liu Y, Yan J, Guo Y, Jing Y, Yang X, Song Y, Tian Y (2016) DNA damage preceding dopamine neuron degeneration in A53T human alpha-synuclein transgenic mice Biochem. Biophys Res Commun 481:104–110. https://doi.org/10.1016/j.bbrc.2016.11.008
Yadav SK, Pandey S, Singh B (2017) Role of estrogen and levodopa in 1-methyl-4-pheny-l-1, 2, 3, 6-tetrahydropyridine (mptp)-induced cognitive deficit in Parkinsonian ovariectomized mice model: A comparative study. J Chem Neuroanat 85:50–59. https://doi.org/10.1016/j.jchemneu.2017.07.002
Yan W, Wu J, Song B, Luo Q, Xu Y (2019) Treatment with a brain-selective prodrug of 17beta-estradiol improves cognitive function in Alzheimer’s disease mice by regulating klf5-NF-kappaB pathway Naunyn. Schmiedebergs Arch Pharmacol 392:879–886. https://doi.org/10.1007/s00210-019-01639-w
Yang Y, Kong F, Ding Q, Cai Y, Hao Y, Tang B (2020) Bruceine D elevates Nrf2 activation to restrain Parkinson’s disease in mice through suppressing oxidative stress and inflammatory response Biochem. Biophys Res Commun 526:1013–1020. https://doi.org/10.1016/j.bbrc.2020.03.097
Zhang G, Yang G, Liu J (2019) Phloretin attenuates behavior deficits and neuroinflammatory response in MPTP induced Parkinson’s disease in mice. Life Sci 232:116600. https://doi.org/10.1016/j.lfs.2019.116600
Zhu YL, Sun MF, Jia XB, Cheng K, Xu YD, Zhou ZL, Zhang PH, Qiao CM, Cui C, Chen X, Yang XS, Shen YQ (2019) Neuroprotective effects of Astilbin on MPTP-induced Parkinson’s disease mice: Glial reaction, alpha-synuclein expression and oxidative stress. Int Immunopharmacol 66:19–27. https://doi.org/10.1016/j.intimp.2018.11.004
Acknowledgements
This work was supported by the William and Ella Owens Medical Research Foundation, Department of Defense grant W81XWH-17-1-0062; NIH grant R03 NS114616 and the Division of Rehabilitation Sciences, College of Health Professions, University of Tennessee Health Science Center.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflicts of interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Thadathil, N., Xiao, J., Hori, R. et al. Brain Selective Estrogen Treatment Protects Dopaminergic Neurons and Preserves Behavioral Function in MPTP-induced Mouse Model of Parkinson’s Disease. J Neuroimmune Pharmacol 16, 667–678 (2021). https://doi.org/10.1007/s11481-020-09972-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11481-020-09972-1