Abstract
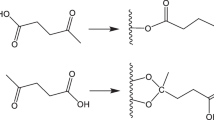
This paper reports on a flowable lignocellulosic thermoplastic prepared from forestry biomass by solvent-free acetylation. The non-solvent approach relies upon a functionalizing agent derived from benzethonium chloride (hyamine) and sulfuric acid, which was chose for its similar wetting attributes to an ionic liquid for the lignocellulose but was much less expensive to use. Besides acetylation, this functionalizing agent became chemically bonded to the lignocellulose by the sulfate group formed in situ, as demonstrated by 13C NMR, infrared and elemental analysis. This attached species appeared to contribute strongly to the flowable nature of the product. The modified material showed good melt flowability by compression molding, as demonstrated in this study by the production of semi-transparent films and was characterized by differential scanning calorimetry and dynamic mechanical analysis. An experimental investigation of reaction parameters was included in the study, exploring the mechanism by which the cationic functionalizing agent modified the structure of lignocellulose.
Graphic Abstract











Similar content being viewed by others
References
Alila S, Boufi S, Belgacem MN, Beneventi D (2005) Adsorption of a cationic surfactant onto cellulosic fibers I. Surface charge effects. Langmuir 21:8106–8113. https://doi.org/10.1021/la050367n
Ass BAP, Frollini E, Heinze T (2004) Studies on the homogeneous acetylation of cellulose in the novel solvent dimethyl sulfoxide/tetrabutylammonium fluoride trihydrate. Macromol Biosci 4:1008–1013. https://doi.org/10.1002/mabi.200400088
Beck S, Bouchard J (2014) Auto-catalyzed acidic desulfation of cellulose nanocrystals. Nord Pulp Pap Res J 29:6–14. https://doi.org/10.3183/npprj-2014-29-01-p006-014
Böke H, Akkurt S, Özdemir S et al (2004) Quantification of CaCO3-CaSO3·0.5H2O-CaSO4·2H2O mixtures by FTIR analysis and its ANN model. Mater Lett 58:723–726. https://doi.org/10.1016/j.matlet.2003.07.008
Boufi S, Chakchouk M, Aloulou F (2004) Adsorption of octadecyltrimethylammonium chloride and adsolubilization on to cellulosic fibers. Colloid Polym Sci 282:699–707. https://doi.org/10.1007/s00396-003-1000-4
Boulven M, Quintard G, Cottaz A et al (2019) Homogeneous acylation of cellulose diacetate: towards bioplastics with tuneable thermal and water transport properties. Carbohydr Polym 206:674–684. https://doi.org/10.1016/j.carbpol.2018.11.030
Chauvelon G, Buléon A, Thibault J-F, Saulnier L (2003) Preparation of sulfoacetate derivatives of cellulose by direct esterification. Carbohydr Res 338:743–750. https://doi.org/10.1016/S0008-6215(03)00008-9
Chen G, Zhang B, Zhao J, Chen H (2013) Improved process for the production of cellulose sulfate using sulfuric acid/ethanol solution. Carbohydr Polym 95:332–337. https://doi.org/10.1016/j.carbpol.2013.03.003
Chen J, Tang C, Yue Y et al (2017) Highly translucent all wood plastics via heterogeneous esterification in ionic liquid/dimethyl sulfoxide. Ind Crops Prod 108:286–294. https://doi.org/10.1016/j.indcrop.2017.06.054
Chen L, Wang Q, Hirth K et al (2015) Tailoring the yield and characteristics of wood cellulose nanocrystals (CNC) using concentrated acid hydrolysis. Cellulose 22:1753–1762. https://doi.org/10.1007/s10570-015-0615-1
Chen M-J, Li R-M, Zhang X-Q et al (2017) Homogeneous transesterification of sugar cane bagasse toward sustainable plastics. ACS Sustain Chem Eng 5:360–366. https://doi.org/10.1021/acssuschemeng.6b01735
Chen Z, Zhang J, Xiao P et al (2018) Novel thermoplastic cellulose esters containing bulky moieties and soft segments. ACS Sustain Chem Eng 6:4931–4939. https://doi.org/10.1021/acssuschemeng.7b04466
Dong XM, Gray DG (1997) Effect of counterions on ordered phase formation in suspensions of charged rodlike cellulose crystallites. Langmuir 13:2404–2409. https://doi.org/10.1021/la960724h
Edgar KJ, Buchanan CM, Debenham JS et al (2001) Advances in cellulose ester performance and application. Prog Polym Sci 26:1605–1688. https://doi.org/10.1016/S0079-6700(01)00027-2
Gan T, Zhang Y, Chen Y et al (2018) Reactivity of main components and substituent distribution in esterified sugarcane bagasse prepared by effective solid phase reaction. Carbohydr Polym 181:633–641. https://doi.org/10.1016/j.carbpol.2017.11.102
Hirose D, Kusuma SBW, Ina D et al (2019) Direct one-step synthesis of a formally fully bio-based polymer from cellulose and cinnamon flavor. Green Chem 21:4927–4931. https://doi.org/10.1039/c9gc01333d
Huang S, O’Donnell KP, Keen JM et al (2016) A new extrudable form of hypromellose: AFFINISOLTM HPMC HME. AAPS PharmSciTech 17:106–119. https://doi.org/10.1208/s12249-015-0395-9
Huang W, Wang Y, Zhang L, Chen L (2016) Rapid dissolution of spruce cellulose in H2SO4 aqueous solution at low temperature. Cellulose 23:3463–3473. https://doi.org/10.1007/s10570-016-1047-2
Hummel A (2004) 3.2 Industrial processes. Macromol Symp 208:61–80. https://doi.org/10.1002/masy.200450406
Jordan JH, Easson MW, Condon BD (2019) Alkali hydrolysis of sulfated cellulose nanocrystals: optimization of reaction conditions and tailored surface charge. Nanomaterials 9:1232. https://doi.org/10.3390/nano9091232
Kono H, Oka C, Kishimoto R, Fujita S (2017) NMR characterization of cellulose acetate: mole fraction of monomers in cellulose acetate determined from carbonyl carbon resonances. Carbohydr Polym 170:23–32. https://doi.org/10.1016/j.carbpol.2017.04.061
Kotelnikova N, Bykhovtsova Y, Mikhailidi A et al (2014) Solubility of lignocellulose in N, N-dimethylacetamide/lithium chloride. WAXS,13C CP/MAS NMR, FTIR and SEM studies of samples regenerated from the solutions. Cellul Chem Technol 48:643–651
Kuo C, Bogan R (1997) Process for the manufacture of cellulose acetate. US Patent 5608050
Larsson PA, Wågberg L (2016) Towards natural-fibre-based thermoplastic films produced by conventional papermaking. Green Chem 18:3324–3333. https://doi.org/10.1039/c5gc03068d
Li J, Thompson M, Lawton DJW (2019) Improved chemical reactivity of lignocellulose from high solids content micro-fibrillation by twin-screw extrusion. J Polym Environ 27:643–651. https://doi.org/10.1007/s10924-019-01377-3
Malm CJ, Tanghe LJ, Laird BC (1946) Preparation of cellulose acetate - action of sulfuric acid. Ind Eng Chem 38:77–82. https://doi.org/10.1021/ie50433a033
Mao J, Heck B, Reiter G, Laborie M-P (2015) Cellulose nanocrystals’ production in near theoretical yields by 1-butyl-3-methylimidazolium hydrogen sulfate ([Bmim]HSO4) mediated hydrolysis. Carbohydr Polym 117:443–451. https://doi.org/10.1016/j.carbpol.2014.10.001
Nge TT, Takata E, Takahashi S, Yamada T (2016) Isolation and thermal characterization of softwood-derived lignin with thermal flow properties. ACS Sustain Chem Eng 4:2861–2868. https://doi.org/10.1021/acssuschemeng.6b00447
Salajková M, Berglund LA, Zhou Q (2012) Hydrophobic cellulose nanocrystals modified with quaternary ammonium salts. J Mater Chem 22:19798. https://doi.org/10.1039/c2jm34355j
Shimizu M, Saito T, Isogai A (2014) Bulky quaternary alkylammonium counterions enhance the nanodispersibility of 2,2,6,6-tetramethylpiperidine-1-oxyl-oxidized cellulose in diverse solvents. Biomacromol 15:1904–1909. https://doi.org/10.1021/bm500384d
Tanaka S, Iwata T, Iji M (2017) Long/short chain mixed cellulose esters: effects of long acyl chain structures on mechanical and thermal properties. ACS Sustain Chem Eng 5:1485–1493. https://doi.org/10.1021/acssuschemeng.6b02066
Tedeschi G, Guzman-Puyol S, Paul UC et al (2018) Thermoplastic cellulose acetate oleate films with high barrier properties and ductile behaviour. Chem Eng J 348:840–849. https://doi.org/10.1016/j.cej.2018.05.031
Teramoto Y (2015) Functional thermoplastic materials from derivatives of cellulose and related structural polysaccharides. Molecules 20:5487–5527. https://doi.org/10.3390/molecules20045487
Teramoto Y, Nishio Y (2003) Cellulose diacetate-graft-poly(lactic acid)s: synthesis of wide-ranging compositions and their thermal and mechanical properties. Polymer (Guildf) 44:2701–2709. https://doi.org/10.1016/S0032-3861(03)00190-3
Toyama K, Soyama M, Tanaka S, Iji M (2015) Development of cardanol-bonded cellulose thermoplastics: high productivity achieved in two-step heterogeneous process. Cellulose 22:1625–1639. https://doi.org/10.1007/s10570-015-0601-7
Wang J, Boy R, Nguyen NA et al (2017) Controlled assembly of lignocellulosic biomass components and properties of reformed materials. ACS Sustain Chem Eng 5:8044–8052. https://doi.org/10.1021/acssuschemeng.7b01639
Wen X, Wang H, Wei Y et al (2017) Preparation and characterization of cellulose laurate ester by catalyzed transesterification. Carbohydr Polym 168:247–254. https://doi.org/10.1016/j.carbpol.2017.03.074
Yan C, Zhang J, Lv Y et al (2009) Thermoplastic cellulose-graft-poly(L-lactide) copolymers homogeneously synthesized in an ionic liquid with 4-dimethylaminopyridine catalyst. Biomacromol 10:2013–2018. https://doi.org/10.1021/bm900447u
Zare A, Ghobadpoor A, Safdari T (2020) Preparation, characterization and utilization of a novel dicationic molten salt as catalyst for the synthesis of bis(6-amino-1,3-dimethyluracil-5-yl)methanes. Res Chem Intermed 46:1319–1327. https://doi.org/10.1007/s11164-019-04036-3
Zhen L, Zhang G, Huang K et al (2016) Modification of rice straw for good thermoplasticity via graft copolymerization of ε-caprolactone onto acetylated rice straw using ultrasonic-microwave coassisted technology. ACS Sustain Chem Eng 4:957–964. https://doi.org/10.1021/acssuschemeng.5b01039
Acknowledgments
This research was funded under the “Clean Manufacturing and Nano-engineering of Sustainable Materials” Ontario Research Fund (ORF) Grant (#07-41) led by Dr. Sain at the University of Toronto. The authors are also grateful to Sonja Hadzidedic at Xerox Research Center of Canada, and Dr. Zhicheng Pan at McMaster University for their technical help on the nitrogen and sulfur elemental analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors associated with McMaster University declare no conflict of interest. David J. W. Lawton is an employee of the Xerox Research Centre of Canada, a division of Xerox Canada.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, J., Zhang, H., Sacripante, G.G. et al. Solvent-free modification of lignocellulosic wood pulp into a melt-flowable thermoplastic. Cellulose 28, 1055–1069 (2021). https://doi.org/10.1007/s10570-020-03589-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-020-03589-6