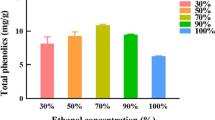
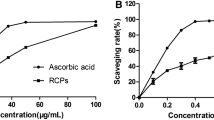
Abstract
Cinnamomum cassia Presl. (CCP) is a popular natural spice possessing various pharmacological properties. We obtained polyphenol-rich fraction (CCP-P) from CCP by bioactivity-oriented purification method and evaluated its Wnt signaling inhibition activity. Firstly, the phenolic components were identified as the main bioactive compounds with anti-colorectal cancer activity. Then, we compared the anti-colorectal cancer activity of CCP extract obtained from different solvent by cell morphology alteration and EdU assay. Ethanol extract showed higher antiproliferative activity compared to water extract on HCT116 cells, with proliferating cells reducing to 41.12 and 21.83% at 156.00 μg GAE/mL, respectively. Next, separation and enrichment of polyphenols from ethanol extract was performed on AB-8 macroporous resins under optimal conditions. Further evaluation of the CCP-P bioactivity revealed that it exerted more potent antiproliferative activity on RKO and HCT116 cells, showing higher selectivity for Wnt-dependent colorectal cancer cells (CRCs). Ten major polyphenols were identified in the CCP-P by UPLC-ESI-MS/MS. In summary, this study presents evidence that CCP-derived polyphenols are promising potential candidates as functional food ingredients against CRC.





Similar content being viewed by others
Data Availability
All data and models generated or used during the study appear in the submitted article.
Abbreviations
- BV:
-
Bed volume
- CCP:
-
Cinnamomum cassia Presl
- CCP-P:
-
Cinnamomum cassia Presl. polyphenols rich faction
- CRC:
-
Colorectal cancer
- CRCs:
-
Colorectal cancer cells
- GAE:
-
Gallic acid equivalents
- MTT:
-
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- TPC:
-
Total phenolic content
- Wnt:
-
Wingless / Integrated
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. Ca-Cancer J Clin 65(2):87–108. https://doi.org/10.3322/caac.21262
Giles RH, Van Es JH, Clevers H (2003) Caught up in a Wnt storm: Wnt signaling in cancer. Bba-Rev Cancer 1653(1):1–24. https://doi.org/10.1016/S0304-419X(03)00005-2
Anastas JN, Moon RT (2012) WNT signalling pathways as therapeutic targets in cancer. Nat Rev Cancer 13(1):11–26. https://doi.org/10.1038/nrc3419
Majewska M, Lewandowska U (2018) The chemopreventive and anticancer potential against colorectal cancer of polyphenol-rich fruit extracts. Food Rev Int 34(4):390–409. https://doi.org/10.1080/87559129.2017.1307388
Park CH, Chang JY, Hahm ER, Park S, Kim H-K, Yang CH (2005) Quercetin, a potent inhibitor against β-catenin/Tcf signaling in SW480 colon cancer cells. Biochem Bioph Res Co 328(1):227–234. https://doi.org/10.1016/j.bbrc.2004.12.151
Lin GM, Lin HY, Hsu CY, Chang ST (2016) Structural characterization and bioactivity of proanthocyanidins from indigenous cinnamon (Cinnamomum osmophloeum). J Sci Food Agric 96(14):4749–4759. https://doi.org/10.1002/jsfa.7802
Chua MT, Tung YT, Chang ST (2008) Antioxidant activities of ethanolic extracts from the twigs of Cinnamomum osmophloeum. Bioresour Technol 99(6):1918–1925. https://doi.org/10.1016/j.biortech.2007.03.020
Tung YT, Chua MT, Wang SY, Chang ST (2008) Anti-inflammation activities of essential oil and its constituents from indigenous cinnamon (Cinnamomum osmophloeum) twigs. Bioresour Technol 99(9):3908–3913. https://doi.org/10.1016/j.biortech.2007.07.050
Yeh RY, Shiu YL, Shei SC, Cheng SC, Huang SY, Lin JC, Liu CH (2009) Evaluation of the antibacterial activity of leaf and twig extracts of stout camphor tree, Cinnamomum kanehirae, and the effects on immunity and disease resistance of white shrimp, Litopenaeus vannamei. Fish Shellfish Immun 27(1):26–32. https://doi.org/10.1016/j.fsi.2008.11.008
Zhang C, Fan L, Fan S, Wang J, Yu L (2019) Cinnamomum cassia Presl: a review of its traditional uses, Phytochemistry, pharmacology and toxicology. Molecules 24(19):3473. https://doi.org/10.3390/molecules24193473
Giovinazzo G, Grieco F (2015) Functional properties of grape and wine polyphenols. Plant Foods Hum Nutr 70(4):454–462. https://doi.org/10.1007/s11130-015-0518-1
Wang W, Liu H, Wang S, Hao X, Li L (2011) A diterpenoid derivative 15-oxospiramilactone inhibits Wnt/β-catenin signaling and colon cancer cell tumorigenesis. Cell Res 21(5):730–740. https://doi.org/10.1038/cr.2011.30
Bolling BW, Chen YY, Chen CYO (2013) Contributions of phenolics and added vitamin C to the antioxidant capacity of pomegranate and grape juices: synergism and antagonism among constituents. Int J Food Sci Technol 48(12):2650–2658. https://doi.org/10.1111/ijfs.12261
Ayaz M, Junaid M, Ullah F, Sadiq A, Subhan F, Khan MA, Ahmad W, Ali G, Imran M, Ahmad S (2016) Molecularly characterized solvent extracts and saponins from Polygonum hydropiper L. show high anti-angiogenic, anti-tumor, brine shrimp, and fibroblast NIH/3T3 cell line cytotoxicity. Front Pharmacol 7:74. https://doi.org/10.3389/fphar.2016.00074
Li J, Chase HA (2010) Development of adsorptive (non-ionic) macroporous resins and their uses in the purification of pharmacologically-active natural products from plant sources. Nat Prod Rep 27(10):1493–1510. https://doi.org/10.1039/C0NP00015A
Zhao Z, Dong L, Wu Y, Lin F (2011) Preliminary separation and purification of rutin and quercetin from Euonymus alatus (Thunb.) Siebold extracts by macroporous resins. Food Bioprod Process 89(4):266–272. https://doi.org/10.1016/j.fbp.2010.11.001
Hashidoko Y, Tahara S, Mizutani J (1986) New complex isoflavones in the roots of yellow lupin (Lupinus luteus L., cv. Barpine). Agric Biol Chem 50(7):1797–1807. https://doi.org/10.1271/bbb1961.50.1797
Li J, Jiang H, Shi R (2009) A new acylated quercetin glycoside from the leaves of Stevia rebaudiana Bertoni. Nat Prod Res 23(15):1378–1383. https://doi.org/10.1080/14786410802447294
Lommen A, Godejohann M, Venema D, Hollman P, Spraul M (2000) Application of directly coupled HPLC− NMR− MS to the identification and confirmation of quercetin glycosides and phloretin glycosides in apple peel. Anal Chem 72(8):1793–1797. https://doi.org/10.1021/ac9912303
Gaucher M, de Bernonville TD, Guyot S, Dat JF, Brisset M-N (2013) Same ammo, different weapons: enzymatic extracts from two apple genotypes with contrasted susceptibilities to fire blight (Erwinia amylovora) differentially convert phloridzin and phloretin in vitro. Plant Physiol Biochem 72:178–189. https://doi.org/10.1016/j.plaphy.2013.03.012
Imai T, Inoue S, Ohdaira N, Matsushita Y, Suzuki R, Sakurai M, de Jesus JMH, Ozaki SK, Finger Z, Fukushima K (2008) Heartwood extractives from the Amazonian trees Dipteryx odorata, Hymenaea courbaril, and Astronium lecointei and their antioxidant activities. J Wood Sci 54(6):470–475. https://doi.org/10.1007/s10086-008-0975-3
Callemien D, Collin S (2008) Use of RP-HPLC-ESI (−)-MS/MS to differentiate various proanthocyanidin isomers in lager beer extracts. J Am Soc Brew Chem 66(2):109–115. https://doi.org/10.1094/ASBCJ-2008-0215-01
Yankep E, Fomum ZT, Bisrat D, Dagne E, Hellwig V, Steglich W (1998) O-Geranylated isoflavones and a3-phenylcoumarin from Millettia griffoniana. Phytochemistry 49(8):2521–2523. https://doi.org/10.1016/S0031-9422(98)00392-6
Kamnaing P, Free SNF, Nkengfack AE, Folefoc G, Fomum ZT (1999) An isoflavan-quinone and a flavonol from Millettia laurentii. Phytochemistry 51(6):829–832. https://doi.org/10.1016/S0031-9422(99)00043-6
Kumar K, Issac A, Ninan E, Kuttan R, Maliakel B (2014) Enhanced anti-diabetic activity of polyphenol-rich de-coumarinated extracts of Cinnamomum cassia. J Funct Foods 10:54–64. https://doi.org/10.1016/j.jff.2014.05.008
Vardamides J, Azebaze A, Nkengfack A, Van Heerden F, Fomum Z, Ngando T, Conrad J, Vogler B, Kraus W (2003) Scaphopetalone and scaphopetalumate, a lignan and a triterpene ester from Scaphopetalum thonneri. Phytochemistry 62(4):647–650. https://doi.org/10.1016/S0031-9422(02)00616-7
Hope C, Planutis K, Planutiene M, Moyer MP, Johal KS, Woo J, Santoso C, Hanson JA, Holcombe RF (2008) Low concentrations of resveratrol inhibit Wnt signal throughput in colon-derived cells: implications for colon cancer prevention. Mol Nutr Food Res 52(S1):S52–S61. https://doi.org/10.1002/mnfr.200700448
Fuentes RG, Arai MA, Ishibashi M (2015) Natural compounds with Wnt signal modulating activity. Nat Prod Rep 32(12):1622–1628. https://doi.org/10.1039/c5np00074b
Funding
National Natural Science Foundation of China (No. 31671804), the National Key R&D Program of China (2018YFD0901101) and Research and Development Program in Key Areas of Guangdong Province (2019B020210002).
Author information
Authors and Affiliations
Contributions
Li Gao performed purification, cell culture and cell experiments and written the manuscript. Na Gou performed extraction and EdU assay. Erdong Yuan revised the manuscript. Jiaoyan Ren designed experiment.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 1316 kb)
Rights and permissions
About this article
Cite this article
Gao, L., Gou, N., Yuan, E. et al. Bioactivity-Oriented Purification of Polyphenols from Cinnamomum cassia Presl. with Anti-Proliferation Effects on Colorectal Cancer Cells. Plant Foods Hum Nutr 75, 561–568 (2020). https://doi.org/10.1007/s11130-020-00846-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11130-020-00846-8