Abstract
Sorption properties of a composite based on microcrystalline cellulose and nanosized aluminum oxide film immobilized on its surface in removal of fluoride ions from water are studied. The optimal thickness of the sorbent layer at which the maximum sorption of fluoride ions occurs is 50 nm. The effects of various parameters, such as the solution pH and sorption time, on sorption are studied. The kinetic parameters of sorption and the rate-controlling step of the process are determined. The ion exchange mechanism of fluoride ion sorption onto the prepared sorbent is confirmed by IR spectroscopy.
Similar content being viewed by others
INTRODUCTION
Fluorine is one of the essential elements found in the human body, and it exhibits a therapeutic effect only in microscopic doses. Large quantities of fluorine pose a health hazard. The most common source of fluoride ions supplied to the human body is drinking water. The serious problem related to excessive fluorine content in drinking and ground water is especially pressing in many Russian regions and all over the world [1]. According to the World Health Organization, the fluoride concentration in drinking water must be ≤1.5 mg/L. Fluoride- and fluorine-bearing minerals in rocks and soil are a source of excess fluorine in groundwater, which is the main source of drinking water. Large quantities of fluorides are also released during glass production, as well as in chemical industry and metallurgy. Therefore, it is important to remove excess fluorine from drinking water.
Excess fluorine can be removed from water using various methods, such as adsorption [2–10], ion exchange [11], reverse osmosis [12], precipitation in the form of poorly soluble compounds [13, 14], electrodialysis, electrocoagulation [15], etc.
Among the aforementioned methods, absorption is considered the most efficient technique for fluorine removal due to its low cost, high performance and environmental sustainability.
It was shown in a number of studies that the efficiency of fluorine removal is strongly dependent on solution pH [16, 17]; the most efficient adsorption of fluoride ions takes place at pH 5–7.
Bahena et al. [18] studied the mechanism of adsorption onto α-Al2O3; Valdivieso et al. [19] investigated the influence of temperature on this process.
Extensive research focused on activated Al2O3 with its surface modified using lanthanum(III) and yttrium(III) compounds [20] or manganese oxide [21], via impregnation with alum [22], by applying copper oxide [23], or magnesial activation [24] has been conducted.
The methods for fluorine removal from water using adsorbents based on TiO2 [25], bismuth and aluminum titanate [26], and manganese and cerium oxides [27] have been reported in literature.
Inorganic composite materials containing natural polymers draw researchers’ attention to be potentially used as adsorbents due to a number of advantages they have. Cellulose is the most abundant renewable biopolymer on the Earth [28, 29]. It is also affordable, safe, and has a well-developed surface.
In this study, we investigated the adsorption properties of microcrystalline cellulose, with an aluminum oxide nanolayer applied onto its surface using atomic layer deposition (ALD) technology. The resulting composite material was used to study sorption of fluoride ions from water, investigate the dependence of sorption capacity on the thickness of aluminum oxide layer, contact time between the solution and the sorbent, and solution pH. The kinetic parameters of this process were also determined.
EXPERIMENTAL
All reagents used in this study were of chemically pure or pure for analysis grade: alizarin complexon (technical specifications TU 6-09-4547), lanthanum nitrate hexahydrate (technical specifications TU 6-09-4676), sodium acetate trihydrate (State Standard GOST 199), sodium hydroxide (State Standard GOST 4328), acetic acid (State Standard GOST 61), hydrochloric acid and nitric acid titration standards. Deionized water was used in the experiments. All experiments were conducted at room temperature under the standard atmospheric pressure.
Films based on microcrystalline cellulose were used as the initial matrix. Before a microcrystalline cellulose film was deposited, it had been placed in the reactor for the process to be conducted.
A model solution of fluoride ions at a certain concentration was prepared by diluting the State Standard Sample 7261-96.
The sorbents were cellulose onto which Al2O3 films of various thicknesses were was applied using ALD technology. The principle of thin-film deposition by ALD is as follows. At an operating temperature, a substrate placed inside a vacuum chamber was alternately exposed to two reagents (precursors) existing in the vapor state. Precursors react only on the substrate surface and form a thin-film monolayer. The thickness of the deposited layers was determined from the calibration curves plotted beforehand [30]. Figure 1 shows a representative pattern obtained when scanning electron spectroscopy (SEM) is used to study the film cross-section. One can see in Fig. 1 that a dense aluminum oxide film with almost uniform thickness across its entire contour is formed.
Trimethylaluminum (Al(CH3)3; TMA) and water (H2O) were used as precursors when the aluminum oxide film was deposited; the reactor temperature was 100°C. This process has been studied well: film growth takes place due to self-controlled reactions, and aluminum containing a precursor is highly reactive [31]. The process can be conditionally divided into four stages:
(1) feeding TMA vapor into the chamber; the TMA binds to the substrate surface and loses one of its methyl groups; two methyl groups bound to aluminum atom become active surface groups;
(2) purging the chamber with a carrier gas to remove the residual precursor and reaction products from it; after purging, the surface-bound precursor remains on the substrate surface;
(3) feeding water vapor into the chamber; water molecules instantaneously react with the surface monolayer, so an oxide film is formed and methane is released as a volatile reaction product; after this stage, hydroxyl groups become active surface groups; and
(4) purging the chamber with the carrier gas to remove residual water vapor and reaction products; when the first precursor (in our case, TAM) is subsequently fed, it again reacts with the surface hydroxyl groups to start a new cycle of the ALD process; as a result, we obtain a thin film with active surface groups that participate in the next semicycle.
The ALD process can be written in the simplified form as follows:
etc., where an asterisk denotes the surface group in the substrate.
The adsorption experiments were conducted in the steady-state mode. Samples of the film made from microcrystalline cellulose modified with aluminum oxide (sample weight was ranged from 0.01 to 0.02 g) were added to the solution containing fluoride ions at a concentration of 5 mg/L and mixed for 1 h at a rate of 200 rpm. Once the equilibrium had been attained, the weight concentration of fluoride ions was measured photometrically (State Standard GOST 4386-89) on a UV-1800 Shimadzu UV-Vis spectrophotometer.
The weight percentage of aluminum oxide deposited onto cellulose was determined by atomic absorption spectroscopy (AAS). This percentage ranged from 41.40 to 66.94% depending on sorbent layer. Each experiment was run in triplicate under identical conditions; the standard deviation of measurements was ± 2%. The solutions after sorption were also investigated by AAS to study leaching of ions from the sorbent into the solution.
Next, the adsorption capacity was calculated in terms of the equivalent amount of the total sorbent weight and separately, weight of aluminum oxide as
where A is the adsorption capacity of the sorbent, mg/g; c0 is the initial concentration of fluoride ions in the solution, mg/L; c is the equilibrium concentration of fluoride ions in the solution after sorption, mg/L; m is the total sorbent weight and sorbent weight calculated as Al2O3, g; and V is the volume of the solution from which fluoride ions are being sorbed, L.
RESULTS AND DISCUSSION
The initial sample of microcrystalline cellulose film not coated with aluminum oxide does not exhibit adsorption properties as evidenced by zero adsorption capacity values during the entire experiment.
The Effect of the Al2O3 Layer Thickness on Sorption of Fluoride Ions
In order to determine the optimal conditions of fluoride ion adsorption, we found the optimal thickness of aluminum oxide layer by varying this value from 50 to 200 nm with 50 nm increments (Fig. 2). The results of the experiment show that the maximum adsorption capacity (40.3 mg/g) is reached when the thickness of aluminum oxide layer is 50 nm. This value is higher than those presented in refs. [21] (Al(OH)3 with adsorption capacity of 0.19 mg/g was used as an adsorbent), [24] (activated aluminum with adsorption capacity of 1.45 mg/g was used as an adsorbent), and [25] (granulated iron hydroxide with adsorption capacity of 7.0 mg/g was used as an adsorbent).
The adsorption capacity decreases with increasing film layer thickness possibly due to the formation of a partially crystalline phase, which impedes binding of fluoride ions.
Effect of Solution pH on Fluoride Adsorption
The effect of solution pH on adsorption of fluoride ions was studied by conducting the experiment at different pH values (1.5–8). Figure 3 shows the adsorption of fluoride ions onto the biopolymer as a function of solution pH. Adsorption capacity increases with pH. Fluoride adsorption onto the sorbent is maximum at pH 6.8. Adsorption capacity declines as pH is further increased.
As shown in [32], the effect of pH can also be explained by examining the point of zero charge of the adsorbent (pHpzc). The surface of the adsorbent under study is positively charged at pH < 2.3 and becomes negatively charged at pH > 2.3. Therefore, adsorption is unfavorable at pH < 2.3 due to repulsive electrostatic interactions between ions and positively charged functional groups. The maximum adsorption of fluorine takes place at pH values above pHpzc when the adsorbent surface has a high negative charge.
Sorbent stability during sorption was studied by AAS; stability of the resulting material was investigated. Aluminum concentration in the solutions was measured after sorption at different modes. The results are summarized in Table 1. One can see that the solutions under study contain no aluminum ions, so it can be inferred that the resulting material is sufficiently stable over time under sorption conditions
The Kinetics of Sorption
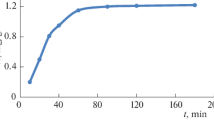
By studying the sorption kinetics one can determine the rate of equilibrium attainment, the maximum sorbent operating capacity for a solution with certain composition, and the mechanism of interaction between fluoride ions and the sorbent during sorption. The findings indicate that sorption of fluoride ions onto a sample coated with a 50 nm thick aluminum oxide layer occurs more vigorously than onto a sample coated with a 100 nm thick sorption layer (Fig. 4).
Figure 4 demonstrates that the adsorption capacity significantly rises during 1 h; after 60 min, it is slightly changed and attains the equilibrium state.
It is known from the literature that the mechanism of sorption of ions is complex and multistage, so it is difficult to consider all stages of the process [33]. Therefore, the rate-controlling step of sorption and the contribution of diffusion processes have been determined for the kinetic process using the linearity criterion. For external diffusion processes, the dependence can be written as –ln(1 – F) = f(τ), and the kinetic curve of this process is fitted by Eq. (2):
where y is a certain constant for these conditions and F is the equilibrium attainment degree.
The dependence –ln(1 – F) = f(τ) (Fig. 5) was obtained to confirm the effect of external diffusion on sorption rate.
For the sample coated with a 100 nm Al2O3 layer (Fig. 5, curve 2), the function –ln(1–F) = f(τ) is linear not over its entire range. The resulting dependence indicates that sorption is characterized by a mixed diffusion-controlled mechanism (i.e., sorbate diffusion from the solution to the sorbent surface and sorbate diffusion inside the sorbent grain.)
The situation is different for the sample coated with a 50 nm Al2O3 layer (Fig. 5, curve 1). Here, –ln(1 – F) = f(τ) is a linear function over its entire range, thus indicating that sorption onto the sorbent proceeds via the external diffusion mechanism.
Linear dependence is observed for the process where internal diffusion is the rate-controlling step: Гτ = f(τ1/2); the kinetic curve is described by Eq. (3):
where Гτ is the amount of fluoride ions per unit weight of the sorbent, mmol/g; Kd is the rate constant of internal diffusion, mmol g–1 min–0.5; and A is the value proportional to thickness of the film surrounding the sorbent grain (an intercept cut off by continuation of the linear function Гτ = f(τ) on the Y axis).
The linearity of the Гτ = f(τ) function shown in Fig. 6 proves that internal diffusion is the rate-controlling step of sorption.
These dependences were used to determine the kinetic parameters (the rate constants of internal diffusion Kd) characterizing the internal diffusion of fluoride ions in the sorbent with aluminum oxide layer being 50 and 100 nm thick: Kd (50 nm) = 0.317 L mol–1 s–1; Kd (100 nm) = 0.179 L mol–1 s–1.
Adsorption was conducted in the temperature range between 10 and 37°C. One can see from the curves shown in Fig. 7 that the adsorption rate is highest during the first 20 min, and the system approaches the equilibrium in ~ 60 min.
Figure 7 shows fitting the resulting data in the coordinates from Eq. (4):
Since the kinetics of chemical reactions describe well the reactions that have not proceeded entirely to completion, the region where the reaction had been completed by 70–80% was used to analyze and plot the logarithmic curve.
One can see in Fig. 7 that the logarithmic curves are linear, which proves that the adsorption of fluoride ions is a first-order reaction [34].
The IR spectroscopy data verify that fluoride ion adsorption onto the sorbent under study occurs via the ion-exchange mechanism. After sorption, the IR spectra of the samples feature absorption bands at 735 and 740 cm–1, corresponding to the asymmetric stretching vibrations of the Al–F (Fig. 8).
CONCLUSIONS
Sorption of fluoride ions onto a cellulose-based material with an aluminum oxide nanolayer applied onto it using the atomic layer deposition technology has been studied. Adsorption of fluoride ions is maximum at the minimum sorbent layer thickness of 50 nm. To study the adsorption capacity of the sorbent with respect to fluoride ions, we have studied the sorption as a function of solution pH. The maximum adsorption capacity is observed at pH 7. The fluoride ion adsorption isotherms have been recorded; the rate-controlling step of sorption has been determined by calculating the kinetic parameters of the process.
REFERENCES
M. Inaniyan and T. Raychoudhury, Int. J. Environ. Sci. Technol. 16, 7545 (2019). https://doi.org/10.1007/s13762-018-2097-9
C. Ren, Z. Yu, B. L. Phillips, et al., J. Colloid Interface Sci. 557, 357 (2019). https://doi.org/10.1016/j.jcis.2019.09.039
P. Mondal and M. K. Purkait, Chemosp 235, 391 (2019). https://doi.org/http:1016/j.chemosphere.2019.06.189
J. Liu, P. Zhao, Xu. Yue, and X. Ji, Bioinorg. Chem. Appl. 2019, 1 (2019). https://doi.org/10.1155/2019/5840205
L. Wei, F. Zietzschmann, L. C. Rietveld, and D. Halem, Chemosphere 243, 125 307 (2020). https://doi.org/10.1016/j.chemosphere.2019.125307
S. Raghav, M. Nair, and D. Kumar, Appl. Surf. Sci. 498, 143 785 (2019). https://doi.org/10.1016/j.apsusc.2019.143785
R. Kumar and S. Mondal, Lecture Notes Civil Eng. 57, 417 (2020). https://doi.org/10.1007/978-981-15-0990-2_34
C. Yang, L. Guan, J. Wang, et al., Environ. Technol. 40, 3668 (2019). https://doi.org/10.1080/09593330.2018.1484523
N. B. Singh, Y. K. Srivastava, S. P. Shukla, and J. B. Markandeya, J. Inst. Eng.(India): A 100, 667 (2019). https://doi.org/10.1007/s40030-019-00387-7
P. Miretzky and A. F. Cirelli, J. Fluorine Chem. 132, 231 (2011). https://doi.org/10.1016/j.jfluchem.2011.02.001
S. Meenakshi and N. Viswanathan, J. Colloid Interface Sci. 308, 438 (2007). https://doi.org/10.1016/j.jcis.2006.12.032
L. A. Richards, B. S. Richards, and A. I. Schafer, J. Membr. Sci. 369, 188 (2011). https://doi.org/10.1016/j.memsci.2010.11.069
I. Labastida, M. A. Armienta, M. Beltran, et al., J. Geochem. Explor. 183, 206 (2017). https://doi.org/10.1016/j.gexplo.2016.12.001
S. Lahnid, M. Tahaikt, K. Elaroui, et al., Desalination 230, 213 (2008). https://doi.org/10.1016/j.desal.2007.11.027
A. Nagaraj, K. K. Sadasivuni, and M. Rajan, Carbohydr. Res. 176, 402 (2017). https://doi.org/10.1016/j.carbpol.2017.08.089
H. Farrah, J. Slavek, and W. F. Pickering, J. Soil Res. 25, 55 (1987). https://doi.org/10.1071/SR9870055
Y. Ku and H.-M. Chiou, Water Air Soil Pollut. 133, 349 (2002). https://doi.org/10.1023/A:1012929900113
J. L. R. Bahena, A. R. Cabrera, A. L. Valdivieso, and R. H. Urbina, Sep. Sci. Technol. 37, 1973 (2002). https://doi.org/10.1081/SS-120003055
A. L. Valdivieso, BahenaJ. L. Reyes, S. Song, and UrbinaR. Herrera, J. Colloid Interface Sci. 298, 1 (2006). https://doi.org/10.1016/j.jcis.2005.11.060
S. A. Wasay, S. Tokunaga, and S. W. Park, Sep. Sci. Technol. 31, 1501 (1996). https://doi.org/10.1080/01496399608001409
S. M. Maliyekkal, A. K. Sharma, and L. Philip, Water Res. 40, 3497 (2006). https://doi.org/10.1016/j.watres.2006.08.007
S. S. Tripathy, J.-L. Bersillon, and K. Gopal, Sep. Purif. Technol. 50, 310 (2006). https://doi.org/10.1016/j.seppur.2005.11.036
A. Bansiwal, P. Pillewan, R. B. Biniwale, and S. S. Rayalu, Microporous Mesoporous Mater. 129, 54 (2010). https://doi.org/10.1016/j.micromeso.2009.08.032
S. M. Maliyekkal, S. Shukla, L. Philip, and I. M. Nambi, Chem. Eng. J. 140, 183 (2008). https://doi.org/10.1016/j.cej.2007.09.049
T. Wajima, Y. Umeta, S. Narita, and K. Sugawara, Desalination 249, 323 (2009). https://doi.org/10.1016/j.desal.2009.06.038
M. Karthikeyan and K. P. Elango, J. Environ. Sci. (China) 21, 1513 (2009). https://doi.org/10.1016/S1001-0742(08)62448-1
S. Deng, H. Liu, W. Zhou, et al., J. Hazard. Mater. 186, 1016 (2011). https://doi.org/10.1016/j.jhazmat.2010.12.024
Y. Ku and H.-M. Chiou, Water Air Soil Pollut. 133, 349 (2002). https://doi.org/10.1023/A:1012929900113
C. Sundaram, N. Viswanathan, and S. Meenakshi, J. Hazard. Mater. 163, 618 (2009). https://doi.org/10.1016/j.jhazmat.2008.07.009
K. N. Nishchev, M. I. Novopoltsev, N. A. Ruzavina, et al., Proceedings of the 14th International Baltic Conference on Atomic Layer Deposition, BALD 2016, p. 24. https://doi.org/10.1109/BALD.2016.7886524
V. Miikkulainen, M. Leskelä, M. Ritala, and R. L. Puurunen, J. Appl. Phys., 021 301 (2013). https://doi.org/10.1063/1.4757907
A. Toyoda and T. Taira, IEEE Trans. Semicond. Manuf. 13, 1109 (2000). https://doi.org/10.1109/66.857940
R. Alosmanov, Sorb. Khromatograf. Prots. 10, 427 (2010).
H. N. Tran, S.-J. You, A. Hosseini-Bandegharaei, et al., Chemosphere, 10 (2017). https://doi.org/10.1016/j.watres.2017.04.014
Funding
This work was supported by the Ministry of Science and Higher Education of the Russian Federation (project no. 0729-2020-0039).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by D. Terpilovskaya
Rights and permissions
About this article
Cite this article
Dolganov, A.V., Balandina, A.V., Chugunov, D.B. et al. Sorption of Fluoride Ions onto Cellulose and Aluminum Oxide Composites. Russ. J. Inorg. Chem. 65, 1770–1775 (2020). https://doi.org/10.1134/S0036023620110030
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023620110030