Abstract
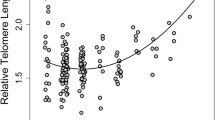
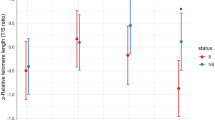
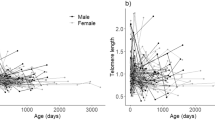
Telomere shortening has been used as an indicator of aging and is believed to accelerate under harsh environmental conditions. This can be attributed to the fact that telomere shortening has often been regarded as non-reversible and negatively impacting fitness. However, studies of laboratory mice indicate that they may be able to repair telomere loss to recover from environmental harshness, as indicated by recent studies in hibernating rodents. We studied seasonal variation in telomere dynamics in African striped mice (Rhabdomys pumilio) living in a highly seasonal environment. In our annual species, individuals born in the moist spring (high food availability) need to survive the harsh dry summer (low food availability) to be able to reproduce in the following spring. We studied the effect of the harsh dry vs. the benign moist season on telomere dynamics. We also tested if telomere length or the rate of change in telomere length over the dry season predicted the probablity of dissapearance from the population at the same time. Male, but not female, stripped mice showed age-related telomere erosion. Telomeres were longer at the beginning of the dry season compared to the rest of the year. Telomeres increased significantly in length during the moist season. Neither telomere length at the onset of the dry season nor telomere loss over the dry season predicted whether or not individuals disappeared. In conclusion, our data suggest that seasonal attrition and restoring of telomeres also occurs in non-hibernating wild rodents living in hot food restricted environments.




Similar content being viewed by others
References
Atema E, Mulder E, Van Noordwijk AJ, Verhulst S (2018) Ultra-long telomeres shorten with age in nestling great tits but are static in adults and mask attrition of short telomeres. Moler Ecol Res 19:648–658
Barrett ELB, Richardson DS (2011) Sex differences in telomeres and lifespan. Aging Cell 10:913–921. https://doi.org/10.1111/j.1474-9726.2011.00741.x
Barton K (2020) MuMIn: Multi-Model Inference. . R package version 1.43.17. https://CRAN.R-project.org/package=MuMIn. Accessed 20 May 2019
Bates D, Mächler M, Bolker B, Walker S (2014) Fitting linear mixed-effects models using lme4. arXiv 1808.00864:1406.5823
Bateson M, Eisenberg DTA, Nettle D (2019) Controlling for baseline telomere length biases estimates of the rate of telomere attrition. Royal Soc Open Sci 6:190937. https://doi.org/10.1098/rsos.19093710.6084/m9.figshare.c
Bateson M, Nettle D (2016) The telomere lengthening conundrum - it could be biology. Aging Cell 16:312–319
Bauch C, Becker PH, Verhulst S (2014) Within the genome, long telomeres are more informative than short telomeres with respect to fitness components in a long-lived seabird. Mol Ecol 23:300–310. https://doi.org/10.1111/mec.12602
Blackburn EH (1991) Structure and function of telomeres. Nature 6319:569
Blount JD, Vitikainen EIK, Stott I, Cant MA (2016) Oxidative shielding and the cost of reproduction. Biol Rev 91:483–497. https://doi.org/10.1111/brv.12179
Boonekamp JJ, Bauch C, Mulder E, Verhulst S (2017) Does oxidative stress shorten telomeres? Biol Let 13:20170164
Cawthon RM (2002) Telomere measurement by quantitative PCR. Nucleic Acids Res 30:e47
Cerchiara JA, Risques RA, Prunkard D, Smith JR, Kane OJ, Boersma PD (2017) Telomeres shorten and then lengthen before fledging in magellanic penguins (Spheniscus magellanicus). Aging 9:487–493
Cesare AJ, Reddel RR (2010) Alternative lengthening of telomeres: models, mechanisms and implications. Nat Rev Genet 11:319–330. https://doi.org/10.1038/nrg2763
Chan SRWL, Blackburn EH (2004) Telomeres and telomerase philosophical transactions of the royal society B. Biol Sci 359:109–122. https://doi.org/10.1098/rstb.2003.1370
Cicchetti DV (1994) Guidelines, criteria, and rules of thumb for evaluating normed and standardized assessment instruments in psychology. Psychol Assess 6:284–290. https://doi.org/10.1037/1040-3590.6.4.284
Criscuolo F, Smith S, Zahn S, Heidinger BJ, Haussmann MF (2018) Experimental manipulation of telomere length: does it reveal a corner-stone role for telomerase in the natural variability of individual fitness? Philosophical transactions of the royal society B. Biol Sci 373:20160440. https://doi.org/10.1098/rstb.2016.044010.6084/m9
Criscuolo F, Sorci G, Behaim-Delarbre M, Zahn S, Faivre B, Bertile F (2018) Age-related response to an acute innate immune challenge in mice: proteomics reveals a telomere maintenance-related cost proceedings of the royal society B. Biol Sci 285:20181877. https://doi.org/10.1098/rspb.2018.1877
Daniali L et al (2013) Telomeres shorten at equivalent rates in somatic tissues of adults. Nat Commun 4:1597. https://doi.org/10.1038/ncomms2602
Dantzer B, Fletcher QE (2015) Telomeres shorten more slowly in slow-aging wild animals than in fast-aging ones. Exp Gerontol 71:38–47. https://doi.org/10.1016/j.exger.2015.08.012
De Punder K, Heim C, Przesdzing I, Wadhwa PD, Entringer S (2018) Characterization in humans of in vitro leucocyte maximal telomerase activity capacity and association with stress philosophical transactions of the royal society B. Biol Sci. https://doi.org/10.1098/rstb.2016.044110.6084/m9
Delignette-Muller M-L, Dutang C (2015) Fitdistrplus: an R package for fitting distributions. J Stat Softw 64:1–34
Eisenberg DTA (2016) Telomere length measurement validity: the coefficient of variation is invalid and cannot be used to compare quantitative polymerase chain reaction and Southern blot telomere length measurement techniques. Internat J Epidemiol. https://doi.org/10.1093/ije/dyw191
Fairlie J, Holland R, Pikington JG, Pemberton JM, Harrington L, Nussey DH (2016) Lifelong leukocyte telomere dynamics and survival in a free-living mammal. Aging Cell 15:140–148
Gil D, Alfonso-Iñiguez S, Pérez-Rodríguez L, Muriel J, Monclús R (2019) Harsh conditions during early development influence telomere length in an altricial passerine: Links with oxidative stress and corticosteroids. J Evol Biol 32:111–125. https://doi.org/10.1111/jeb.13396
Gomes NMV, Shay JW, Wright WE (2010) Telomere biology in Metazoa. FEBS Lett 584:3741–3751. https://doi.org/10.1016/j.febslet.2010.07.031
Gorbunova V, Bozzella MJ, Seluanov A (2008) Rodents for comparative aging studies: from mice to beavers. Age 30:111–119. https://doi.org/10.1007/s11357-008-9053-4
Grosbellet E et al (2015) Circadian desynchronization triggers premature cellular aging in a diurnal rodent. FASEB J 29:4794–4803. https://doi.org/10.1096/fj.14-266817
Harris SE et al (2016) Longitudinal telomere length shortening and cognitive and physical decline in later life: the Lothian Birth Cohorts 1936 and 1921. Mech Ageing Dev 154:43–48. https://doi.org/10.1016/j.mad.2016.02.004
Haussmann MF, Longenecker AS, Marchetto NM, Juliano SA, Bowden RM (2012) Embryonic exposure to corticosterone modifies the juvenile stress response, oxidative stress and telomere length proceedings of the royal society B. Biol Sci 279:1447–1456. https://doi.org/10.1098/rspb.2011.1913
Haussmann MF, Winkler DW, Huntington CE, Nisbet ICT, Vleck CM (2004) Telomerase expression is differentially regulated in birds of differing life span. Ann N Y Acad Sci 1019:186–190. https://doi.org/10.1196/annals.1297.029
Heidinger BJ, Blount JD, Boner W, Griffiths K, Metcalfe NB, Monaghan P (2012) Telomere length in early life predicts lifespan. Proc Natl Acad Sci 109:1742–1748. https://doi.org/10.1073/pnas.1113306109
Hoelzl F, Cornils JS, Smith S, Moodley Y, Ruf T (2016) Telomere dynamics in free-living edible dormice (Glis glis): the impact of hibernation and food supply. J Exper Biol 219:2469–2474. https://doi.org/10.1242/jeb.140871
Hoelzl F, Smith S, Cornils JS, Aydinonat D, Bieber C, Ruf T (2016) Telomeres are elongated in older individuals in a hibernating rodent, the edible dormouse (Glis glis). Scient Rep 6:36856. https://doi.org/10.1038/srep36856
Hothorn T, Bretz F, Westfall P, Heiberger RM (2008) Multcomp: simultaneous inference for general linear hypotheses. R Pack Vers 1:1
Huchon D, Steppan SJ, Schenk JJ (2017) Muroid rodent phylogenetics: 900-species tree reveals increasing diversification rates. PLoS ONE 12:e0183070. https://doi.org/10.1371/journal.pone.0183070
Ilmonen P, Kotrschal A, Penn DJ (2008) Telomere attrition due to infection. PLoS ONE 3:e2143. https://doi.org/10.1371/journal.pone.0002143.t001
Kotrschal A, Ilmonen P, Penn DJ (2007) Stress impacts telomere dynamics. Biol Lett 3:128–130. https://doi.org/10.1098/rsbl.2006.0594
Lê S, Josse J, Husson F (2008) FactoMineR: an R package for multivariate analysis. J Stat Softw 25:2–18
Martin-Ruiz CM, Gussekloo J, Van Heemst D, Von Zglinicki T, Westendorp RGJ (2005) Telomere length in white blood cells is not associated with morbidity or mortality in the oldest old: a population-based study. Aging Cell 4:287–290
Nettle D, Bateson M (2017) Detecting telomere elongation in longitudinal datasets: analysis of a proposal by Simons Stulp and Nakagawa. PeerJ 5:e3265. https://doi.org/10.7717/peerj.3265
Nettle D, Monaghan P, Gillespie R, Brilot B, Bedford T, Bateson M (2015) An experimental demonstration that early-life competitive disadvantage accelerates telomere loss Proceedings of the Royal Society B. Biol Sci 282:20141610–20141610. https://doi.org/10.1098/rspb.2014.1610
Nettle D, Seeker LA, Nussey D, Froy H, Bateson M (2019) Consequences of measurement error in qPCR telomere data: a simulation study. PLoS ONE 14:e0216118. https://doi.org/10.1101/491944
Nowack J, Tarmann I, Hoelzl F, Smith S, Giroud S, Ruf T (2019) Always a price to pay: hibernation at low temperatures comes with a trade-off between energy savings and telomere damage. Biol Let 15:20190466. https://doi.org/10.1098/rsbl.2019.0466
Olonikov AM (1973) A theory of marginotomy. J Theor Biol 41:181–190
Olsson M, Wapstra E, Friesen C (2018) Ectothermic telomeres: it’s time they came in from the cold philosophical transactions of the royal society B. Biol Sci 373:20160449. https://doi.org/10.1098/rstb.2016.0449
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:2003–2007
Quirici V, Guerrero CJ, Krause JS, Wingfield JC, Vásquez RA (2016) The relationship of telomere length to baseline corticosterone levels in nestlings of an altricial passerine bird in natural populations. Front Zool. https://doi.org/10.1186/s12983-016-0133-5
Reichert S, Stier A (2017) Does oxidative stress shorten telomeres in vivo? Rev Biol Lett 13:20170463. https://doi.org/10.1098/rsbl.2017.0463
Reichert S et al (2014) Increased brood size leads to persistent eroded telomeres. Front Ecol Evol. https://doi.org/10.3389/fevo.2014.00009
Rimbach R, Blanc S, Zahariev A, Gatta M, Pillay N, Schradin C (2018) Seasonal variation in energy expenditure in a rodent inhabiting a winter-rainfall desert. J Comp Physiol B. https://doi.org/10.1007/s00360-018-1168-z
Rimbach R, Jäger J, Pillay N, Schradin C (2018) Food availability is the main driver of seasonal changes in resting metabolic rate in african striped mice (Rhabdomys pumilio). Physiol Biochem Zool 91:826–833. https://doi.org/10.1086/696828
Rimbach R, Pillay N, Schradin C (2016) Both thyroid hormone levels and resting metabolic rate decrease in African striped mice when food availability decreases. J Exper Biol. https://doi.org/10.1242/jeb.151449
Salomons HM, Mulder GA, van de Zande L, Haussmann MF, Linskens MHK, Verhulst S (2009) Telomere shortening and survival in free-living corvids proceedings of the royal society B. Biol Sci 276:3157–3165. https://doi.org/10.1098/rspb.2009.0517
Schradin C (2008a) Differences in prolactin levels between three alternative male reproductive tactics in striped mice (Rhabdomys pumilio). Proce Royal Soc London 275:1047–1052
Schradin C (2008b) Seasonal changes in testosterone and corticosterone levels in four social categories of a desert dwelling sociable rodent. Horm Behav 53:573–579
Schradin C (2008c) Seasonal changes in testosterone and corticosterone levels in four social classes of a desert dwelling sociable rodent. Horm Behav 53:573–579. https://doi.org/10.1016/j.yhbeh.2008.01.003
Schradin C, König B, Pillay N (2010) Reproductive competition favours solitary living while ecological constraints impose group-living in African striped mice. J Anim Ecol 79:515–521
Schradin C et al (2012) Social flexibility and social evolution in mammals: a case study of the African striped mouse (Rhabdomys pumilio). Mol Ecol 21:541–553. https://doi.org/10.1111/j.1365-294X.2011.05256.x
Schradin C, Pillay N (2005) Demography of the striped mouse (Rhabdomys pumilio) in the succulent karoo: a unique population in an extreme environment. Mammalian Biol 70:84–92
Schradin C, Pillay N, Bertelsmeier C (2019) Social flexibility and environmental unpredictability in African striped mice. Behav Ecol Sociobiol 73:94
Schradin C, Raynaud J, Arrivé M, Blanc S (2014) Leptin levels in free ranging striped mice (Rhabdomys pumilio) increase when food decreases: the ecological leptin hypothesis. Gen Comp Endocrinol 206:139–145. https://doi.org/10.1016/j.ygcen.2014.06.024
Seeker LA et al (2018) Longitudinal changes in telomere length and associated genetic parameters in dairy cattle analysed using random regression models. PLoS ONE 13:e0192864. https://doi.org/10.1371/journal.pone.0192864
Seluanov A et al (2007) Telomerase activity coevolves with body mass, not lifespan. Aging Cell 6:45–52
Simons MJP (2015) Questioning causal involvement of telomeres in aging. Ageing Res Rev. https://doi.org/10.1016/j.arr.2015.08.002
Simons MJP, Stulp G, Nakagawa S (2014) A statistical approach to distinguish telomere elongation from error in longitudinal datasets. Biogerontology 15:99–103. https://doi.org/10.1007/s10522-013-9471-2
Smith S, Turbill C, Penn DJ (2011) Chasing telomeres, not red herrings, in evolutionary ecology. Heredity 107:372–373. https://doi.org/10.1038/hdy.2011.14
Spurgin LG et al (2018) Spatio-temporal variation in lifelong telomere dynamics in a long-term ecological study. J Anim Ecol 87:187–198. https://doi.org/10.1111/1365-2656.12741
Steenstrup T, JvB H, Kark JD, Christensen K, Aviv A (2013) The telomere lengthening conundrum–artifact or biology? Nucleic Acids Res 41:e131–e131. https://doi.org/10.1093/nar/gkt370
Stier A et al (2014) Starting with a handicap: phenotypic differences between early- and late-born king penguin chicks and their survival correlates. Funct Ecol 28:601–611. https://doi.org/10.1111/1365-2435.12204
Tian X et al (2018) Evolution of telomere maintenance and tumour suppressor mechanisms across mammals philosophical transactions of the royal society B. Biol Sci 373:20160443. https://doi.org/10.1098/rstb.2016.044310.6084/m9
Tissier ML, Williams TD, Criscuolo F (2014) Maternal effects underlie ageing costs of growth in the zebra finch (Taeniopygia guttata). PLoS ONE 9:e97705. https://doi.org/10.1371/journal.pone.0097705
Turbill C, Ruf T, Smith S, Bieber C (2013) Seasonal variation in telomere length of a hibernating rodent. Biol Let 9:20121095–20121095. https://doi.org/10.1098/rsbl.2012.1095
Turbill C, Smith S, Deimel C, Ruf T (2012) Daily torpor is associated with telomere length change over winter in Djungarian hamsters. Biol Let 8:304–307. https://doi.org/10.1098/rsbl.2011.0758
Vera E, Bernardes de Jesus B, Foronda M, Flores Juana M, Blasco Maria A (2012) The rate of increase of short telomeres predicts longevity in mammals. Cell Rep 2:732–737. https://doi.org/10.1016/j.celrep.2012.08.023
Verhulst S, Aviv A, Benetos A, Berenson GS, Kark JD (2013) Do leukocyte telomere length dynamics depend on baseline telomere length? An analysis that corrects for ‘regression to the mean.’ Eur J Epidemiol 28:859–866. https://doi.org/10.1007/s10654-013-9845-4
Viblanc VA et al (2018) Maternal oxidative stress and reproduction: Testing the constraint, cost and shielding hypotheses in a wild mammal. Funct Ecol 32:722–735. https://doi.org/10.1111/1365-2435.13032
Vitikainen EIK et al (2016) Evidence of oxidative shielding of offspring in a wild Mammal. Front Ecol Evol. https://doi.org/10.3389/fevo.2016.00058
Vuarin P, Pillay N, Schradin C (2019) Elevated basal corticosterone levels increase disappearance risk of light but not heavy individuals in a long-term monitored rodent population. Horm Behav 113:95–102
Whittemore K, Vera E, Martínez-Nevado E, Sanpera C, Blasco MA (2019) Telomere shortening rate predicts species life span. Proc Natl Acad Sci 116:15122–15127. https://doi.org/10.1073/pnas.1902452116
Wolak ME, Fairbairn DJ, Paulsen YR (2012) Guidelines for estimating repeatability. Methods Ecol Evol 3:129–137. https://doi.org/10.1111/j.2041-210X.2011.00125.x
Young AJ (2018) The role of telomeres in the mechanisms and evolution of life-history trade-offs and ageing philosophical transactions of the royal society B. Biol Sci 373:20160452. https://doi.org/10.1098/rstb.2016.0452
Acknowledgements
This study was supported by the Succulent Karoo Research Station, the University of the Witwatersrand, and the CNRS. We are grateful to Goegap Nature Reserve and for the help of several field managers and assistants. Data are available online at figshare https://doi.org/10.6084/m9.figshare.13160405.v1.
Author information
Authors and Affiliations
Contributions
CS, NP and FC originally formulated the idea of the manuscript, FC and CS run the telomere analysis, FC and CS wrote the manuscript, NP contributed to writing, FC and CS made tables and prepared figures.
Corresponding authors
Additional information
Communicated by Pawel Koteja.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Criscuolo, F., Pillay, N., Zahn, S. et al. Seasonal variation in telomere dynamics in African striped mice. Oecologia 194, 609–620 (2020). https://doi.org/10.1007/s00442-020-04801-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-020-04801-x