Abstract
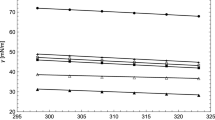
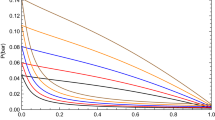
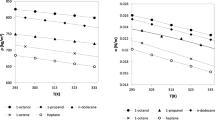
This work has been dedicated to modeling the vapor–liquid equilibria and surface tension of carboxylic acids + water mixtures at different temperatures. The Peng–Robinson equation of state with modified Huron–Vidal + Wilson mixing rule correctly models the vapor pressure and vapor mole fraction of these mixtures. The modified Huron–Vidal mixing rule was better than quadratic mixing rule to model the phase equilibria properties for the mixtures studied in this work. The parachor method and linear gradient theory were used to model the surface tension of these mixtures. Finally, a new symmetric parameter dependent on the liquid molar fraction and temperature was necessary to correctly adjust the surface tension of the carboxylic acids + water mixtures using linear gradient theory. The surface tension results obtained in this work are better than those published so far.
















Similar content being viewed by others
Abbreviations
- \({\mathbb {P}}\) :
-
Parachor parameter of the pure fluid
- a :
-
Cohesion parameter in PR-EOS
- A, B :
-
Coefficients of the influence parameter
- AAD :
-
Statistical deviation
- b :
-
Covolume parameter in PR-EOS
- \(c_1\) :
-
Adjustable parameter in cohesive parameter
- \(f_0\) :
-
Helmholtz energy density
- G :
-
Gibbs free energy
- \(k_{12}\) :
-
Binary interaction parameter of quadratic mixing rule
- \(l_{12}\), \(m_{12}\), \(s_{12}\), \(s_{21}\) :
-
Coefficients of the correlation for the symmetric parameter
- m :
-
Parameter of the thermal cohesive function
- n :
-
Number of points
- \(n_c\) :
-
Number of components
- P :
-
Absolute pressure
- R :
-
Universal gas constant
- T :
-
Absolute temperature
- u :
-
MHV function
- x :
-
Liquid mole fraction
- y :
-
Vapor mole fraction
- z :
-
Position in the interface
- \(\alpha (T)\) :
-
Thermal cohesive function
- \(\beta _{12}\) :
-
Symmetric parameter
- \(\gamma \) :
-
Coefficient activity
- \(\kappa \) :
-
Influence parameter
- \(\lambda _{12}\) :
-
Adjustable parameter of the parachor method
- \(\Lambda _{12}\), \(\Lambda _{21}\) :
-
Wilson parameters
- \(\mu \) :
-
Chemical potential
- \(\Omega \) :
-
Grand thermodynamic potential
- \(\omega \) :
-
Acentric factor
- \(\rho \) :
-
Molar density
- \(\sigma \) :
-
Surface tension
- \(\xi \) :
-
MHV function
- c :
-
Critical condition
- i, j, s :
-
Species
- 0:
-
Equilibrium condition
- E :
-
Excess
- exp :
-
Experimental
- L :
-
Liquid phase
- theo :
-
Theoretical
- V :
-
Vapor phase
References
L.A. Román-Ramírez, G.A. Leeke, Int. J. Thermophys. 41, 1–27 (2020)
K. Granados, J. Gracia-Fadrique, A. Amigo, R. Bravo, J. Chem. Eng. Data 51, 1356–1360 (2006)
E. Álvarez, G. Vázquez, M. Sánchez-Vilas, B. Sanjurjo, J.M. Navaza, J. Chem. Eng. Data 42, 957–960 (1997)
J.C. Eriksson, Adv. Chem. Phys., 145–174, 1964
F.B. Sprow, J.M. Prausnitz, Trans. Faraday Soc. 62, 1105–1111 (1966)
J.A. Hugill, A.J. Van Welsenes, Fluid Phase Equilib. 29, 383–390 (1986)
J. Hekayati, S. Raeissi, J. Mol. Liq. 231, 451–461 (2017)
X. Liang, M.L. Michelsen, G.M. Kontogeorgis, Fluid Phase Equilib. 428, 153–163 (2016)
J.D. van der Waals, Zeit Phys Chem. 13, 675–725 (1894)
J.W. Cahn, J.E. Hilliard, J. Chem. Phys. 28, 258–267 (1958)
A. Mejía, J.C. Pàmies, D. Duque, H. Segura, L.F. Vega, J. Chem. Phys. 123, 034505 (2005)
A. Hernández, Chem. Phys., 110747, 2020
A. Hernández, S. Khosharay, Int. J. Thermophys. 41, 1–22 (2020)
Y.-X. Zuo, E.H. Stenby, J. Chem. Eng. Japan 29, 159–165 (1996)
K.A.G. Schmidt, G.K. Folas, B. Kvamme, Fluid Phase Equilib. 261, 230–237 (2007)
A. Hernández, M. Cartes, A. Mejía, Fuel 229, 105–115 (2018)
A. Hernández, Int. J. Thermophys. 41, 1–18 (2020)
D.Y. Peng, D.B. Robinson, Ind. Eng. Chem. Fundam. 15, 59–64 (1976)
A. Anderko, Fluid Phase Equilib. 61, 145–225 (1990)
M.L. Michelsen, Fluid Phase Equilib. 60, 47–58 (1990)
G.M. Wilson, J. Am. Chem. Soc. 86, 127–130 (1964)
D.B. Macleod, Trans. Faraday Soc. 19, 38–41 (1923)
O.R. Quayle, Chem. Rev. 53, 439–589 (1953)
A. Mejía, H. Segura, L. Vega, J. Wisniak, Fluid Phase Equilib. 227, 225–238 (2005)
Y.-X. Zuo, E.H. Stenby, Can. J. Chem. Eng. 75, 1130–1137 (1997)
Y.-X. Zuo, E.H. Stenby, J. Colloid Interface Sci. 182, 126–132 (1996)
Y.-X. Zuo, E.H. Stenby, SPE J. 3, 134–145 (1998)
J. Wisniak, A. Apelblat, H. Segura, Chem. Eng. Sci. 53, 743–751 (1998)
H.C. Van Ness, Classical thermodynamics of non-electrolyte solutions (Elsevier, Amsterdam, 2015)
C. Miqueu, B. Mendiboure, A. Graciaa, J. Lachaise, Fluid Phase Equilib. 207, 225–246 (2003)
A.N. Campbell, A.J.R. Campbell, Trans. Faraday Soc. 30, 1109–1114 (1934)
T. Sommer, J. Trejbal, D. Kopecky, J. Chem. Eng. Data 61, 3398–3405 (2016)
E.H.M. Wright, B.A. Akhtar, J. Chem. Soc. B, 151–157, 1970
S. Miyamoto, S. Nakamura, Y. Iwai, Y. Arai, J. Chem. Eng. Data 46, 1225–1230 (2001)
L.A. Román-Ramírez, G.A. Leeke, J. Chem. Eng. Data 61, 2078–2082 (2016)
L.A. Román-Ramírez, F. García-Sánchez, R.C.D. Santos, G.A. Leeke, Fluid Phase Equilib. 388, 151–159 (2015)
V.V. Udovenko, L.P. Aleksandrova, Zhurnal Fizicheskoi Khimii 34, 1366–1372 (1960)
Acknowledgements
A.H. acknowledges the economic support given by the UCSC.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hernández, A. Modeling Vapor–Liquid Equilibria and Surface Tension of Carboxylic Acids + Water Mixtures Using Peng–Robinson Equation of State and Gradient Theory. Int J Thermophys 42, 13 (2021). https://doi.org/10.1007/s10765-020-02763-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10765-020-02763-z