Abstract
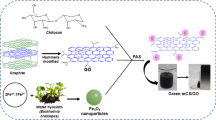
A magnetic composite was synthesized using palm kernel shell-based activated carbon, graphene oxide (GO) and iron oxide. The morphology, textural properties, magnetic properties, crystal structure and functional groups of the composite were studied. The characterization shows the successful formation of the ternary composite with a high specific surface area (280.39 m2/g) and abundant surface functional groups. The magnetic properties of the composite indicate a saturation magnetization of 33.72 emu/g, signifying that the composite can be easily separated with an external magnetic field. The performance of the composite was then investigated through the removal of an anionic azo dye, Acid Blue 113, from aqueous solution. The effect of initial pH of the solution, adsorbent dosage and adsorption contact time was studied. The composite achieved an adsorption capacity of 32.2 mg/g and 96.3% dye removal. Effective dye removal was still achieved after five cycles of adsorption-regeneration, with the dye removal of 74% after the fifth cycle. The analyses of adsorption isotherm and kinetics show that the adsorption process fits pseudo-second-order kinetic and Langmuir isotherm models, indicating a monolayer chemisorption process. The excellent adsorption performance and reusability of the composite and its magnetic separability signify its potential as an adsorbent for wastewater treatment.
Similar content being viewed by others
Abbreviations
- A1 :
-
constant for equilibrium binding [L/g]
- AB113:
-
Acid Blue 113 [−]
- ANOVA:
-
analysis of variance [−]
- b:
-
Langmuir constant [L/mg]
- B1 :
-
constant for heat of sorption [−]
- BET:
-
Brunauer-Emmett-Teller [−]
- BJH:
-
Barrett-Joyner-Halenda [−]
- C:
-
carbon [−]
- c:
-
thickness of the boundary layer constant [−]
- C-C:
-
alkene [−]
- C-N:
-
imine [−]
- C2H5OH:
-
ethanol [−]
- C6H5NaSO3 :
-
sodium benzenesulfonate [−]
- C6H5NH2 :
-
aniline [−]
- CCD:
-
central composite design [−]
- Ce :
-
AB113 dye concentration at equilibrium [mg/L]
- CH3COOH:
-
acetic acid [−]
- C0 :
-
AB113 dye concentration at initial [mg/L]
- Fe:
-
iron [−]
- Fe2O3 :
-
Hematite; Maghemite; [−]
- Fe3O4 :
-
magnetite[−]
- FeCl2·4H2O:
-
ferrous chloride tetrahydrate [−]
- FeCl3·6H2O:
-
iron (III) chloride hexahydrate [−]
- FTIR:
-
fourier transformation infrared [−]
- GO:
-
graphene oxide [−]
- H2O2 :
-
hydrogen peroxide [−]
- H2SO4 :
-
sulphuric acid [−]
- HCl:
-
hydrochloric acid [−]
- I-P:
-
intra-particle [−]
- k1 :
-
rate constant for P-F-O [min−1]
- k2 :
-
rate constant for P-S-O [g mg−1 min−1]
- KCl:
-
potassium chloride [−]
- kf :
-
Freundlich constant [mg1−n Ln g−1]
- ki :
-
intraparticle diffusion rate coefficient [mg g−1 min−0.5]
- KMnO4 :
-
potassiumpermanganate[−]
- MGOAC:
-
magnetic graphene oxide-activated carbon [−]
- N-C-S:
-
isothiocyanate [−]
- NaOH:
-
sodium hydroxide [−]
- NH4OH:
-
ammonium hydroxide [−]
- O:
-
oxygen [−]
- P-F-O:
-
pseudo-first-order [−]
- pHpzc :
-
point of zero charge [−]
- PKS-AC:
-
palm kernel shell-based activated carbon [−]
- P-S-O:
-
pseudo-second-order [−]
- qe :
-
quantity of AB113 dye adsorbed per weight of MGOAC at equilibrium [mg/g]
- Qo :
-
amount of AB113 dye adsorbed per weight of MGOAC at monolayer [mg/g]
- qt :
-
quantity of AB113 dye adsorbed per weight of MGOAC at time t [mg/g]
- R2 :
-
coefficient of determination [−]
- RL :
-
separation factor [−]
- rpm:
-
revolutions per minute [−]
- RSM:
-
response surface methodology [−]
- S:
-
sulfur [−]
- SEM/EDX:
-
scanning electron microscopy coupled with energy dispersive X-ray [−]
- t:
-
time [min]
- V:
-
sample solution volume [L]
- VSM:
-
vibrating-sample magnetometer [−]
- W:
-
dosage of MGOAC [g]
- XRD:
-
X-ray diffraction [−]
References
J. Martín, M. D. M. Orta, S. Medina-Carrasco, J. L. Santos, I. Aparicio and E. Alonso, Environ. Res., 164, 488 (2018).
S. N. Jain, S. R. Tamboli, D. S. Sutar, S. R. Jadhav, J. V. Marathe, A. A. Shaikh and A. A. Prajapati, J. Clean. Prod., 252, 119778 (2020).
P. Mehdizadeh, Y Orooji, O. Amiri, M. Salavati-Niasari and H. Moayedi, J. Clean. Prod., 252, 119765 (2020).
Y. W. Kim, J. H. Kim, D. H. Moon and H. J. Shin, Korean J. Chem. Eng., 36, 101 (2019).
A. Pirkarami and M. E. Olya, J. Saudi Chem. Soc., 21, S179 (2017).
A. I. Abd-Elhamid, E. A. Kamoun, A. A. El-Shanshory, H. M. A. Soliman and H. F. Aly, J. Mol. Liq., 279, 530 (2019).
A. M. Aljeboree, A. N. Alshirifi and A. F. Alkaim, Arab. J. Chem., 10, S3381 (2017).
S. Tian, S. Xu, J. Liu, C. He, Y. Xiong and P. Feng, J. Clean. Prod., 239, 117767 (2019).
A. L. D. Rosa, E. Carissimi, G. L. Dotto, H. Sander and L. A. Feris, J. Clean. Prod., 198, 1302 (2018).
M. Naushad, A.A. Alqadami, Z.A. AlOthman, I.H. Alsohaimi, M. S. Algamdi and A. M. Aldawsari, J. Mol. Liq., 293, 111442 (2019).
C. H. Nguyen and R.-S. Juang, J. Taiwan Inst. Chem. Eng., 99, 166 (2019).
N. P. Khumalo, G. D. Vilakati, S. D. Mhlanga, A. T. Kuvarega, B. B. Mamba, J. Li and D. S. Dlamini, J. Water Process Eng., 31, 100878 (2019).
G. Kyriakopoulos, I. Xiarchos and D. Doulia, Int. J. Environ. Technol. Manag., 6, 515 (2006).
Y Li, Q. Du, T. Liu, X. Peng, J. Wang, J. Sun, Y Wang, S. Wu, Z. Wang, Y. Xia and L. Xia, Chem. Eng. Res. Des., 91, 361 (2013).
R. Sahraei, Z.S. Pour and M. Ghaemy, J. Clean. Prod., 142, 2973 (2017).
P. Ranjan, J. Balakrishnan and A. D. Thakur, Mater. Today Proc., 11, 833 (2019).
P. Pal and A. Pal, J. Water Process Eng., 31, 100882 (2019).
G. Kyriakopoulos and D. Doulia, Fresenius Environ. Bull., 16, 731 (2007).
J. Roh, H.N. Umh, C.M. Yoo, S. Rengara;, B. Lee and Y. Kim, Korean J. Chem. Eng., 29, 903 (2012).
J. Pérez-Calderón, M. V. Santos and N. Zaritzky, J. Environ. Chem. Eng., 6, 6749 (2018).
W. Xiao, Z. N. Garba, S. Sun, I. Lawan, L. Wang, M. Lin and Z. Yuan, J. Clean. Prod., 253, 119989 (2020).
S. Wong, H. H. Tumari, N. Ngadi, N. B. Mohamed, O. Hassan, R. Mat and N. A. S. Amin, J. Clean. Prod., 206, 394 (2019).
A. A. Peláez-Cid, V Romero-Hernández, A. M. Herrera-González, A. Bautista-Hernández and O. Coreño-Alonso, Chinese J. Chem. Eng., 28, 613 (2019).
L. P. Lingamdinne, J.-S. Choi, Y.-L. Choi, J.-K. Yang, J. R. Koduru and Y Chang, Metals (Basel)., 9, 1 (2019).
N. Singh, S. Riyajuddin, K. Ghosh, S. Mehta and A. Dan, ACS Appl. Nano Mater., 2, 7379 (2019).
Y Orooji, F. Liang, A. Razmjou, G. Liu and W Jin, Sep. Purif. Technol., 205, 273 (2018).
F. Tahmasebi, M. Alimohammadi, R. Nabizadeh, M. Khoobi, K. Karimian and A. Zarei, Korean J. Chem. Eng., 36, 894 (2019).
L. P. Lingamdinne, J. R. Koduru and R. R. Karri, J. Environ. Manage., 231, 622 (2019).
R. Nasiri and N. Arsalani, J. Clean. Prod., 190, 63 (2018).
Y. Jiang, J. L. Gong, G. M. Zeng, X. M. Ou, Y. N. Chang, C. H. Deng, J. Zhang, H. Y. Liu and S. Y. Huang, Int. J. Biol. Macromol., 82, 702 (2016).
Y Zhang, H. Li, M. Li and M. Xin, J. Mol. Struct., 1209, 127973 (2020).
G. Abdi, A. Alizadeh, J. Amirian, S. Rezaei and G. Sharma, J. Mol. Liq., 289, 111118 (2019).
H. Karimi-Maleh, M. Shafieizadeh, M. A. Taher, F. Opoku, E. M. Kiarii, P. P. Govender, S. Ranjbari, M. Rezapour and Y. Orooji, J. Mol. Liq., 298, 112040 (2020).
I. Ali, A. Arsh, X. Y. Mbianda, A. Burakov, E. Galunin, I. Burakova, E. Mkrtchyan, A. Tkachev and V. Grachev, Environ. Int., 127, 160 (2019).
G. Yao, W. Bi and H. Liu, Colloids Surf. A Physicochem. Eng. Asp., 588, 124393 (2020).
H. Hosseinzadeh and S. Ramin, Int. J. Biol. Macromol., 113, 859 (2018).
J.-Y. Chen, S.-R. Cao, C.-X. Xi, Y. Chen, X.-L. Li, L. Zhang, G.-M. Wang, Y.-L. Chen and Z.-Q. Chen, Food Chem., 239, 911 (2018).
M. Gao, Z. Wang, C. Yang, J. Ning, Z. Zhou and G. Li, Colloids Surf. A Physicochem. Eng. Asp., 566, 48 (2019).
S. Mishra, S. S. Yadav, S. Rawat, J. Singh and J. R. Koduru, J. Environ. Manage., 246, 362 (2019).
G. Asgari, A. Shabanloo, M. Salari and F. Eslami, Environ. Res., 184, 109367 (2020).
Y. Orooji, M. Ghanbari, O. Amiri and M. Salavati-Niasari, J. Hazard. Mater., 389, 122079 (2020).
F. A. Taher, F. H. Kamal, N. A. Badawy and A. E. Shrshr, Mater. Res. Bull., 97, 361 (2018).
A. I. A. Sherlala, A. A. A. Raman, M. M. Bello and A. Buthiyappan, J. Environ. Manage., 246, 547 (2019).
L. G. Bach, T. V. Tran, T. D. Nguyen, T. V. Pham and S. T. Do, Res. Chem. Intermed., 44, 1661 (2018).
L.P. Lingamdinne, K.R. Vemula, Y.Y Chang, J.K. Yang, R.R. Karri and J. R. Koduru, Chemosphere, 243, 125257 (2020).
M. O. Saeed, K. Azizli, M. H. Isa and M. J. K. Bashir, J. Water Process Eng., 8, e7 (2015).
Ş. Yılmaz, T. Şahan and A. Karabakan, Korean J. Chem. Eng., 34, 2225 (2017).
G. Kyriakopoulos, D. Doulia and A. Hourdakis, Int. J. Environ. Anal. Chem., 86, 207 (2006).
S. Liu, M. Li, Y. Liu, N. Liu, X. Tan, L. Jiang, J. Wen, X. Hu and Z. Yin, J. Taiwan Inst. Chem. Eng., 102, 330 (2019).
F. Iranpour, H. Pourzamani, N. Mengelizadeh, P. Bahrami and H. Mohammadi, J. Environ. Chem. Eng., 6, 3418 (2018).
S. K. Behera, H. Meena, S. Chakraborty and B. C. Meikap, Int. J. Min. Sci. Technol., 28, 621 (2018).
A. Muhammad, A. H. A. Shah and S. Bilal, Appl. Sci., 10, 2882 (2020).
L. Li, X. L. Liu, H. Y. Geng, B. Hu, G. W. Song and Z. S. Xu, J. Mater. Chem. A, 1, 10292 (2013).
N. Wahab, M. Saeed, M. Ibrahim, A. Munir, M. Saleem, M. Zahra and A. Waseem, Front. Chem., 7, 1 (2019).
W. Wang, G. Huang, C. An, S. Zhao, X. Chen and P. Zhang, J. Clean. Prod., 172, 1986 (2018).
L.Y. Lee, S. Gan, M.S.Y. Tan, S.S. Lim, X.J. Lee and Y.F. Lam, J. Clean. Prod., 113, 194 (2016).
S. Kaur, S. Rani and R. K. Mahajan, J. Chem., 2013, 1 (2013).
M. F. Alkhatib, A. A. Mamun and I. Akbar, Int. J. Environ. Sci. Technol., 12, 1295 (2015).
M. Wang, J. Fu, Y. Zhang, Z. Chen, M. Wang, J. Zhu, W. Cui, J. Zhang and Q. Xu, J. Macromol. Sci. Part A Pure Appl. Chem., 52, 105 (2015).
S. N. Jain and P. R. Gogate, J. Environ. Chem. Eng., 5, 3384 (2017).
C. Wang, C. Feng, Y. Gao, X. Ma, Q. Wu and Z. Wang, Chem. Eng. J., 173, 92 (2011).
M. Shirzad-Siboni, A. Khataee, F. Vafaei and S. W. Joo, Korean J. Chem. Eng., 31, 1451 (2014).
R. R. Mohammed and M. F. Chong, J. Environ. Manage., 132, 237 (2014).
G. Ciobanu, M. Harja, L. Rusu, A. M. Mocanu and C. Luca, Korean J. Chem. Eng., 31, 1021 (2014).
M. Shirzad-Siboni, S. J. Jafari, O. Giahi, I. Kim, S. M. Lee and J. K. Yang, J. Ind. Eng. Chem., 20, 1432 (2014).
T. V. Tran, D. T. C. Nguyen, H. T. N. Le, O. T. K. Nguyen, V. H. Nguyen, T. T. Nguyen, L. G. Bach and T. D. Nguyen, R. Soc. Open Sci., 6, 190058 (2019).
F. Fatma, P. L. Hariani, F. Riyanti and W. Sepriani, Indones. J. Chem., 18, 222 (2018).
A. Bhattacharyya, B. Banerjee, S. Ghorai, D. Rana, I. Roy, G. Sarkar, N. R. Saha, S. De, T. K. Ghosh, S. Sadhukhan and D. Chattopadhyay, Int. J. Biol. Macromol., 116, 1037 (2018).
N. Liu, H. Wang, C. H. Weng and C. C. Hwang, Arab. J. Chem., 11, 1281 (2018).
C. Wu, C. Lin, S. Lo and T. Yasunaga, J. Colloid Interface Sci., 208, 430 (1998).
Acknowledgement
This work was supported by the Fundamental Research Grant Scheme (FRGS) provided by Ministry of Education (MOE), Malaysia with grant number FP141-2019A.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supporting Information
Additional information as noted in the text. This information is available via the Internet at http://www.springer.com/chemistry/journal/11814.
Supporting Information
Rights and permissions
About this article
Cite this article
Ying, T.Y., Raman, A.A.A., Bello, M.M. et al. Magnetic graphene oxide-biomass activated carbon composite for dye removal. Korean J. Chem. Eng. 37, 2179–2191 (2020). https://doi.org/10.1007/s11814-020-0628-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-020-0628-9