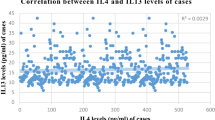
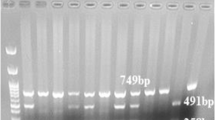
Abstract
Little is known about genetic factors and mechanisms underlying shrimp allergy. Genome-wide association studies identified HLA class-II and IL13 genes as highly plausible candidates for shrimp allergy. The present study was designed to investigate potential associations of HLA-DQ rs9275596, IL13 rs20541, and IL13 rs1800925 polymorphisms with challenge-proven shrimp allergy using the data from 532 people of West Bengal, India; selected on basis of positive skin prick test, elevated specific IgE and medical history. Risk genotypes, i.e., HLA-DQ rs9275596 CC, IL13 rs20541 AA, and IL13 rs1800925 TT, were found to be significantly associated with challenge positive shrimp allergy (P = 0.04, 0.01, and 0.03, respectively). Distribution of genotypes for HLA-DQ and IL13 polymorphisms in allergic and control subjects showed significant difference between younger (20–40 years) and older (> 40 years) age group (P = 0.006). Risk genotypes significantly associated with elevated shrimp-specific IgE. IL13 TA haplotype significantly associated with shrimp allergy and elevated specific IgE (P = 0.02). Synergistic effect of IL13 TA haplotype–HLA-DQ rs9275596 CC genotype interaction significantly elevated specific IgE (P = 0.03). The present study suggests that HLA-DQ and IL13 polymorphisms pose major risk for shrimp allergic patients in West Bengal, India and thus could be helpful for early target-specific therapeutic intervention in near future.


Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are not publicly available due to consent requirements to protect subjects’ privacy but are available from the corresponding author on reasonable request.
References
Arima K, Umeshita-Suyama R, Sakata Y et al (2002) Upregulation of IL-13 concentration in vivo by the IL13 variant associated with bronchial asthma. J Allergy Clin Immunol 109(6):980–7
Ashley SE, Tan HTT, Peters R et al (2017) Genetic variation at the Th2 immune gene IL13 is associated with IgE-mediated paediatric food allergy. Clin Exp Allergy 47(8):1032–1037
Cameron L, Webster RB, Strempel JM et al (2006) Th2 cell-selective enhancement of human IL13 transcription by IL13 -1112C>T, a polymorphism associated with allergic inflammation. J Immunol 177(12):8633–42
Carnes J, Ferrer A, Huertas AJ et al (2007) The use of raw or boiled crustacean extracts for the diagnosis of seafood allergic individuals. Ann Allergy Asthma Immunol 98(4):349–354
Chen W, Ericksen MB, Levin LS et al (2004) Functional effect of the R110Q IL13 genetic variant alone and in combination with IL4RA genetic variants. J Allergy Clin Immunol 114:553–60
Daly MJ, Rioux JD, Schaffner SF et al (2001) High-resolution haplotype structure in the human genome. Nat Genet 29:229–232
Dey D, Ghosh N, Pandey N et al (2014) A hospital-based survey on food allergy in the population of Kolkata, India. Int Arch Allergy Immunol 164:218–21
Dimitrov I, Doytchinova I (2016) Associations between milk and egg allergens and the HLA-DRB1/DQ polymorphism: a bioinformatics approach. Int Arch Allergy Immunol 169:33–39
Ebisawa M, Ito K, Fujisawa T (2017) Committee for Japanese Pediatric Guideline for Food Allergy, The Japanese Society of Pediatric Allergy and Clinical Immunology, The Japanese Society of Allergology Japanese guidelines for food allergy 2017. Allergol Int 66:248–264
Gabriel SB, Schaffner SF, Nguyen H et al (2002) The structure of haplotype blocks in the human genome. Science 296(5576):2225–9
Graves PE, Kabesch M, Halonen M et al (2000) A cluster of seven tightly linked polymorphisms in the IL-13 gene is associated with total serum IgE levels in three populations of white children. J Allergy Clin Immunol 105(3):506–13
Gupta R, Holdford D, Bilaver L et al (2013) The economic impact of childhood food allergy in the United States. JAMA Pediatr 134(Suppl 3):S149–S150
Hoffjan S, Nicolae D, Ober C (2003) Association studies for asthma and atopic diseases: a comprehensive review of the literature. Respir Res 4(1):14
Hong X, Tsai HJ, Wang X (2009) Genetics of food allergy. Curr Opin Pediatr 21:770–776
Hong X, Hao K, Ladd-Acosta C et al (2015) Genome-wide association study identifies peanut allergy-specific loci and evidence of epigenetic mediation in US children. Nat Commun 6:6304
Kazemi S, Vaisi-Raygani A, Keramat F et al (2019) Evaluation of the relationship between IL12, IL-13 and TNF-α gene polymorphisms with the susceptibility to brucellosis: a case control study. BMC Infect Dis 19:1036
Khor SS, Morino R, Nakazono K et al (2018) Genome-wide association study of self-reported food reactions in Japanese identifies shrimp and peach specific loci in the HLA-DR/DQ gene region. Sci Rep 8(1):1069
Kodgule RR, Singh V, Dhar R et al (2014) Reference values for peak expiratory flow in Indian adult population using a European Union scale peak flow meter. J Postgrad Med 60(2):123–9
Kumar R, Kumari D, Srivastava P et al (2010) Identification of IgE-mediated food allergy and allergens in older children and adults with asthma and allergic rhinitis. Indian J Chest Dis Allied Sci 52(4):217–24
Laha A, Ghosh A, Moitra S et al (2020) Association of STAT6 rs3024974 (C/T) polymorphism with IgE mediated food sensitization among West Bengal population. India. Int Arch Allergy Immunol 181(3):200–210
Lertnawapan R, Maek-a-nantawat W (2011) Anaphylaxis and biphasic phase in Thailand: 4-year observation. Allergol Int 60:283–289
Liu X, Nickel R, Beyer K et al (2000) An IL13 coding region variant is associated with a high total serum IgE level and atopic dermatitis in the German multicenter atopy study (MAS-90). J Allergy Clin Immunol 106:167–70
Liu X, Beaty TH, Deindl P et al (2004) Associations between specific serum IgE response and 6 variants within the genes IL4, IL13, and IL4RA in German children: the German Multicenter Atopy Study. J Allergy Clin Immunol 113(3):489–495
Long X, Chen Q, Zhao J et al (2015) An IL-13 promoter polymorphism associated with Liver Fibrosis in patients with Schistosoma japonicum. PLoS ONE 10(8):e0135360
Madore AM, Vaillancourt VT, Asai Y et al (2013) HLA-DQB1*02 and DQB1*06:03P are associated with peanut allergy. Eur J Hum Genet 21(10):1181–4
Mandal J, Das M, Roy I et al (2009) Immediate hypersensitivity to common food allergens: an investigation on food sensitization in respiratory allergic patients of Calcutta, India. World Allergy Organ J 2:9–12
Martino D, Joo JE, Sexton-Oates A et al (2014) Epigenome-wide association study reveals longitudinally stable DNA methylation differences in CD4+ T cells from children with IgE-mediated food allergy. Epigenetics 9(7):998–1006
Martino DJ, Ashley S, Koplin J et al (2017) Genome-wide association study of peanut allergy reproduces association with amino acid polymorphisms in HLA-DRB1. Clin Exp Allergy 47(2):217–223
Moonesinghe H, Mackenzie H, Venter C et al (2016) Prevalence of fish and shellfish allergy: a systematic review. Ann Allergy Asthma Immunol 117(3):264–272
Muraro A, Werfel T, Hoffmann-Sommergruber K et al (2014) EAACI food allergy and anaphylaxis guidelines: diagnosis and management of food allergy. Allergy 69(8):1008–25
Ravkov EV, Pavlov IY, Martins TB et al (2013) Identification and validation of shrimp-tropomyosin specific CD4 T cell epitopes. Hum Immunol 74(12):1542–9
Rosenbaum D, Hawkins G, Zhu L et al (2014) The rare IL-13 R105Q variant, strongly enhances IL-13 protein activity (HYP6P. 268). J Immunol 192(Suppl 1):118.13.
Ruethers T, Taki AC, Johnston EB et al (2018) Seafood allergy: a comprehensive review of fish and shellfish allergens. Mol Immunol 100:28–57
Sampson HA, van Wijk RG, Bindslev-Jensen C et al (2012) Standardizing double-blind, placebo-controlled oral food challenges: American academy of allergy, asthma & immunology-European academy of allergy and clinical immunology PRACTALL consensus report. J Allergy Clin Immunol 130(6):1260–74
Sicherer SH, Sampson HA (2010) Food allergy. J Allergy Clin Immunol 125(2 Suppl 2):S116-125
Smit DV, Cameron PA, Rainer TH (2005) Anaphylaxis presentations to an emergency department in Hong Kong: incidence and predictors of biphasic reactions. J Emerg Med 28:381–388
Techapornroong M, Akrawinthawong K, Cheungpasitporn W et al (2010) Anaphylaxis: a ten years inpatient retrospective study. Asian Pac J Allergy Immunol 28:262–269
Tejedor-Alonso MA, Moro-Moro M, Mugica-Garcia MV (2015) Epidemiology of anaphylaxis: contributions from the last 10 years. J Investig Allergol Clin Immunol 25(3):163–75
Thong BY, Cheng YK, Leong KP et al (2005) Anaphylaxis in adults referred to a clinical immunology/allergy centre in Singapore. Singapore Med J 46:529–534
Tsai HJ, Kumar R, Pongracic J et al (2009) Familial aggregation of food allergy and sensitization to food allergens: a family-based study. Clin Exp Allergy 39(1):101–9
Vladich FD, Brazille SM, Stern D et al (2005) IL-13 R130Q, a common variant associated with allergy and asthma, enhances effector mechanisms essential for human allergic inflammation. J Clin Invest 115:747–754
Woo CK, Bahna SL (2011) Not all shellfish “allergy” is allergy! Clin Transl Allergy 1:3
Yadav A, Govindasamy GK, Naidu R (2012) Polymorphic variants of interleukin-13 R130Q, interleukin-4 T589C, interleukin-4RA I50V, and interleukin-4RA Q576R in allergic rhinitis: a pilot study. Allergy Rhinol (Providence) 3(1):e35-40
Zotova V, Clarke AE, Chan ES et al (2019) Low resolution rates of seafood allergy. J Allergy Clin Immunol Pract 7(2):690–692
Acknowledgements
The authors convey thanks to the patients who voluntarily participated in our study. We are also grateful to the lab professionals of the Allergy and Asthma Research Center, Kolkata, for their technical help. We are also thankful to the Principal, Barasat Govt. College for providing infrastructure facilities for this study.
Funding
Was provided by Council of Scientific and Industrial Research, Govt. of India (Sanction no. 08/606(0004)/2017-EMR-I dated 14/11/2017) and Department of Science and Technology, Govt. of West Bengal (Sanction No. 1170(Sanc.)/ST/P/S&T/1G-4/2016, dated 02.03.16).
Author information
Authors and Affiliations
Contributions
All the authors have made substantial contributions. Arghya Laha contributed to data compilation and wrote first draft of the manuscript. Amlan Ghosh designed the genotyping protocol for HLA-DQ rs9275596 and helps in LD analysis. Saibal Moitra diagnosed the disease, provided the sample, and supervised the oral food challenge. Himani Biswas helped in data collection. Srijit Bhattacharya did the statistical analysis. Nimai Chandra Saha, Goutam Kumar Saha, and Sanjoy Podder conceived the study design. Sanjoy Podder supervised the work, critically revised the manuscript, and made the final draft. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
The study protocol was approved by the Clinical Research Ethics Committee, Allergy and Asthma Research Center, West Bengal, India (CREC-AARC Ref: 004/17). All the procedures performed in the present study involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Written informed consent was obtained from all included subjects and patient anonymity was preserved using methods approved by the Ethics Committee.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Laha, A., Ghosh, A., Moitra, S. et al. Association of HLA-DQ and IL13 gene variants with challenge-proven shrimp allergy in West Bengal, India. Immunogenetics 72, 489–498 (2020). https://doi.org/10.1007/s00251-020-01185-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-020-01185-3