Abstract
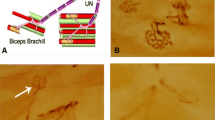
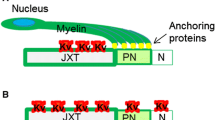
Syndecan-3 (SDC3) and Syndecan-4 (SDC4) are distributed throughout the nervous system (NS) and are favourable factors in motor neuron development. They are also essential for regulation of neurite outgrowth in the CNS. However, their roles in the reconstruction of the nodes of Ranvier after peripheral nerve injury (PNI) are still unclear. Present study used an in vivo model of end-to-side neurorrhaphy (ESN) for 1–3 months. The recovery of neuromuscular function was evaluated by grooming test. Expression and co-localization of SDC3, SDC4, and Nav1.6 channel (Nav1.6) at regenerating axons were detected by proximity ligation assay and confocal microscopy after ESN. Time-of-flight secondary ion mass spectrometry was used for imaging ions distribution on tissue. Our data showed that the re-clustering of sodium and Nav1.6 at nodal regions of the regenerating nerve corresponded to the distribution of SDC3 after ESN. Furthermore, the re-establishment of sodium and Nav1.6 correlated with the recovery of muscle power 3 months after ESN. This study suggested syndecans may involve in stabilizing Nav1.6 and further modulate the distribution of sodium at nodal regions after remyelination. The efficiency of sodium re-clustering was improved by the assistance of anionic syndecan, resulting in a better functional repair of PNI.







Similar content being viewed by others
Abbreviations
- SDC3:
-
Syndecan-3
- SDC4:
-
Syndecan-4
- CMAP:
-
Compound muscle action potential
- ESN:
-
End-to-side neurorrhaphy
- McN:
-
Musculocutaneous nerve
- UN:
-
Ulnar nerve
- TOF–SIMS:
-
Time-of-flight secondary ion mass spectrometry
- PNI:
-
Peripheral nerve injury
References
Asundi VK, Erdman R, Stahl RC, Carey DJ (2003) Matrix metalloproteinase-dependent shedding of syndecan-3, a transmembrane heparan sulfate proteoglycan Schwann cells. J Neurosci Res 73(5):593–602. https://doi.org/10.1002/jnr.10699
Bernfield M, Hinkes MT, Gallo RL (1993) Developmental expression of the syndecans: possible function and regulation. Dev Suppl 119:205–212
Bespalov MM, Sidorova YA, Tumova S, Ahonen-Bishopp A, Magalhaes AC, Kulesskiy E, Paveliev M, Rivera C, Rauvala H, Saarma M (2011) Heparan sulfate proteoglycan syndecan-3 is a novel receptor for GDNF, neurturin, and artemin. J Cell Biol 192(1):153–169. https://doi.org/10.1083/jcb.201009136
Caldwell JH, Schaller KL, Lasher RS, Peles E, Levinson SR (2000) Sodium channel Na(v)1.6 is localized at nodes of ranvier, dendrites, and synapses. Proc Natl Acad Sci U S A 97(10):5616–5620. https://doi.org/10.1073/pnas.090034797
Chattopadhyay S, Shubayev VI (2009) MMP-9 controls Schwann cell proliferation and phenotypic remodeling via IGF-1 and ErbB receptor-mediated activation of MEK/ERK pathway. Glia 57(12):1316–1325. https://doi.org/10.1002/glia.20851
Clausen TM, Pereira MA, Al Nakouzi N, Oo HZ, Agerbaek MO, Lee S, Orum-Madsen MS, Kristensen AR, El-Naggar A, Grandgenett PM, Grem JL, Hollingsworth MA, Holst PJ, Theander T, Sorensen PH, Daugaard M, Salanti A (2016) Oncofetal chondroitin sulfate glycosaminoglycans are key players in integrin signaling and tumor cell motility. Mol Cancer Res 14(12):1288–1299. https://doi.org/10.1158/1541-7786.MCR-16-0103
Couchman JR (2003) Syndecans: proteoglycan regulators of cell-surface microdomains? Nat Rev Mol Cell Biol 4(12):926–937. https://doi.org/10.1038/nrm1257
Coulson-Thomas VJ (2016) The role of heparan sulphate in development: the ectodermal story. Int J Exp Pathol 97(3):213–229. https://doi.org/10.1111/iep.12180
Dufour A, Zucker S, Sampson NS, Kuscu C, Cao J (2010) Role of matrix metalloproteinase-9 dimers in cell migration: design of inhibitory peptides. J Biol Chem 285(46):35944–35956. https://doi.org/10.1074/jbc.M109.091769
Edwards TJ, Hammarlund M (2014) Syndecan promotes axon regeneration by stabilizing growth cone migration. Cell Rep 8(1):272–283. https://doi.org/10.1016/j.celrep.2014.06.008
Elfenbein A, Simons M (2013) Syndecan-4 signaling at a glance. J Cell Sci 126(Pt 17):3799–3804. https://doi.org/10.1242/jcs.124636
Ferguson TA, Muir D (2000) MMP-2 and MMP-9 increase the neurite-promoting potential of schwann cell basal laminae and are upregulated in degenerated nerve. Mol Cell Neurosci 16(2):157–167. https://doi.org/10.1006/mcne.2000.0859
Fukuda T, Kusuhara H, Nakagoshi T, Isogai N, Sueyoshi Y (2018) A basic fibroblast growth factor slow-release system combined to a biodegradable nerve conduit improves endothelial cell and Schwann cell proliferation: A preliminary study in a rat model. Microsurgery 38(8):899–906. https://doi.org/10.1002/micr.30387
Gao X, Wang Y, Chen J, Peng J (2013) The role of peripheral nerve ECM components in the tissue engineering nerve construction. Rev Neurosci 24(4):443–453. https://doi.org/10.1515/revneuro-2013-0022
Garber M, Grabherr MG, Guttman M, Trapnell C (2011) Computational methods for transcriptome annotation and quantification using RNA-seq. Nat Methods 8(6):469–477. https://doi.org/10.1038/nmeth.1613
Gardiner DM (2017) Regulation of regeneration by heparan sulfate proteoglycans in the extracellular matrix. Regen Eng Transl Med 3(3):192–198. https://doi.org/10.1007/s40883-017-0037-8
Goutebroze L, Carnaud M, Denisenko N, Boutterin MC, Girault JA (2003) Syndecan-3 and syndecan-4 are enriched in Schwann cell perinodal processes. BMC Neurosci 4:29. https://doi.org/10.1186/1471-2202-4-29
Henry MA, Freking AR, Johnson LR, Levinson SR (2007) Sodium channel Nav1.6 accumulates at the site of infraorbital nerve injury. BMC Neurosci. 8:56. https://doi.org/10.1186/1471-2202-8-56
Hsueh YP, Sheng M (1999) Regulated expression and subcellular localization of syndecan heparan sulfate proteoglycans and the syndecan-binding protein CASK/LIN-2 during rat brain development. J Neurosci 19(17):7415–7425
Jordanova A, Thomas FP, Guergueltcheva V, Tournev I, Gondim FA, Ishpekova B, De Vriendt E, Jacobs A, Litvinenko I, Ivanova N, Buzhov B, De Jonghe P, Kremensky I, Timmerman V (2003) Dominant intermediate Charcot-Marie-Tooth type C maps to chromosome 1p34-p35. Am J Hum Genet 73(6):1423–1430. https://doi.org/10.1086/379792
Katz E, Stoler O, Scheller A, Khrapunsky Y, Goebbels S, Kirchhoff F, Gutnick MJ, Wolf F, Fleidervish IA (2018) Role of sodium channel subtype in action potential generation by neocortical pyramidal neurons. Proc Natl Acad Sci U S A 115(30):E7184–E7192. https://doi.org/10.1073/pnas.1720493115
Kim Y, Remacle AG, Chernov AV, Liu H, Shubayev I, Lai C, Dolkas J, Shiryaev SA, Golubkov VS, Mizisin AP, Strongin AY, Shubayev VI (2012) The MMP-9/TIMP-1 axis controls the status of differentiation and function of myelin-forming Schwann cells in nerve regeneration. PLoS ONE 7(3):e33664. https://doi.org/10.1371/journal.pone.0033664
Leterrier C, Brachet A, Fache MP, Dargent B (2010) Voltage-gated sodium channel organization in neurons: protein interactions and trafficking pathways. Neurosci Lett 486(2):92–100. https://doi.org/10.1016/j.neulet.2010.08.079
Liao WC, Wang YJ, Huang MC, Tseng GF (2013) Methylcobalamin facilitates collateral sprouting of donor axons and innervation of recipient muscle in end-to-side neurorrhaphy in rats. PLoS ONE 8(9):e76302. https://doi.org/10.1371/journal.pone.0076302
Liu CH, Chang HM, Tseng TJ, Lan CT, Chen LY, Youn SC, Lee JJ, Mai FD, Chou JF, Liao WC (2016) Redistribution of Cav21 channels and calcium ions in nerve terminals following end-to-side neurorrhaphy: ionic imaging analysis by TOF-SIMS. Histochem Cell Biol 146(5):599–608. https://doi.org/10.1007/s00418-016-1470-3
Liu CH, Chang HM, Wu TH, Chen LY, Yang YS, Tseng TJ, Liao WC (2017) Rearrangement of potassium ions and Kv1.1/Kv1.2 potassium channels in regenerating axons following end-to-end neurorrhaphy: ionic images from TOF-SIMS. Histochem Cell Biol 148(4):407–416. https://doi.org/10.1007/s00418-017-1570-8
Mackenzie F, Ruhrberg C (2012) Diverse roles for VEGF-A in the nervous system. Development 139(8):1371–1380. https://doi.org/10.1242/dev.072348
Manon-Jensen T, Itoh Y, Couchman JR (2010) Proteoglycans in health and disease: the multiple roles of syndecan shedding. FEBS J 277(19):3876–3889. https://doi.org/10.1111/j.1742-4658.2010.07798.x
Marchionini DM, Lehrmann E, Chu Y, He B, Sortwell CE, Becker KG, Freed WJ, Kordower JH, Collier TJ (2007) Role of heparin binding growth factors in nigrostriatal dopamine system development and Parkinson’s disease. Brain Res 1147:77–88. https://doi.org/10.1016/j.brainres.2007.02.028
Marquardt LM, Ee X, Iyer N, Hunter D, Mackinnon SE, Wood MD, Sakiyama-Elbert SE (2015) Finely tuned temporal and spatial delivery of GDNF promotes enhanced nerve regeneration in a long nerve defect model. Tissue Eng Part A 21(23–24):2852–2864. https://doi.org/10.1089/ten.TEA.2015.0311
Matthews HK, Marchant L, Carmona-Fontaine C, Kuriyama S, Larrain J, Holt MR, Parsons M, Mayor R (2008) Directional migration of neural crest cells in vivo is regulated by Syndecan-4/Rac1 and non-canonical Wnt signaling/RhoA. Development 135(10):1771–1780. https://doi.org/10.1242/dev.017350
Meneghetti MC, Hughes AJ, Rudd TR, Nader HB, Powell AK, Yates EA, Lima MA (2015) Heparan sulfate and heparin interactions with proteins. J R Soc Interface 12(110):0589. https://doi.org/10.1098/rsif.2015.0589
Mitsou I, Multhaupt HAB, Couchman JR (2017) Proteoglycans, ion channels and cell-matrix adhesion. Biochem J 474(12):1965–1979. https://doi.org/10.1042/BCJ20160747
Oberlin C, Beal D, Leechavengvongs S, Salon A, Dauge MC, Sarcy JJ (1994) Nerve transfer to biceps muscle using a part of ulnar nerve for C5–C6 avulsion of the brachial plexus: anatomical study and report of four cases. J Hand Surg Am 19(2):232–237. https://doi.org/10.1016/0363-5023(94)90011-6
Park PW (2018) Isolation and functional analysis of syndecans. Methods Cell Biol 143:317–333. https://doi.org/10.1016/bs.mcb.2017.08.019
Prince RN, Schreiter ER, Zou P, Wiley HS, Ting AY, Lee RT, Lauffenburger DA (2010) The heparin-binding domain of HB-EGF mediates localization to sites of cell-cell contact and prevents HB-EGF proteolytic release. J Cell Sci 123(Pt 13):2308–2318. https://doi.org/10.1242/jcs.058321
Quintes S, Goebbels S, Saher G, Schwab MH, Nave KA (2010) Neuron-glia signaling and the protection of axon function by Schwann cells. J Peripher Nerv Syst 15(1):10–16. https://doi.org/10.1111/j.1529-8027.2010.00247.x
Sarrazin S, Lamanna WC, Esko JD (2011) Heparan sulfate proteoglycans. Cold Spring Harb Perspect Biol. https://doi.org/10.1101/cshperspect.a004952
Schlosshauer B, Muller E, Schroder B, Planck H, Muller HW (2003) Rat Schwann cells in bioresorbable nerve guides to promote and accelerate axonal regeneration. Brain Res 963(1–2):321–326. https://doi.org/10.1016/s0006-8993(02)03930-6
Susuki K, Chang KJ, Zollinger DR, Liu Y, Ogawa Y, Eshed-Eisenbach Y, Dours-Zimmermann MT, Oses-Prieto JA, Burlingame AL, Seidenbecher CI, Zimmermann DR, Oohashi T, Peles E, Rasband MN (2013) Three mechanisms assemble central nervous system nodes of Ranvier. Neuron 78(3):469–482. https://doi.org/10.1016/j.neuron.2013.03.005
Wang CC, Yen JH, Cheng YC, Lin CY, Hsieh CT, Gau RJ, Chiou SJ, Chang HY (2017) Polygala tenuifolia extract inhibits lipid accumulation in 3T3-L1 adipocytes and high-fat diet-induced obese mouse model and affects hepatic transcriptome and gut microbiota profiles. Food Nutr Res 61(1):1379861. https://doi.org/10.1080/16546628.2017.1379861
Webber C, Zochodne D (2010) The nerve regenerative microenvironment: early behavior and partnership of axons and Schwann cells. Exp Neurol 223(1):51–59. https://doi.org/10.1016/j.expneurol.2009.05.037
Acknowledgements
This study was supported by research grants to Dr. Wen-Chieh Liao and Dr. Chiung-Hui Liu from the Ministry of Science and Technology, Taiwan, (MOST-108-2320-B-040-013-, 109-2320-B-040-009-, and 109-2320-B-040-007-MY3). Cryostatting (CM3050S, Leica 149 Microsystems) was performed in the Instrument Center of Chung Shan Medical University, which was supported by Ministry of Science and Technology, Ministry of Education, and Chung Shan Medical University.
Author information
Authors and Affiliations
Contributions
Chiung-hui Liu and Yu-chen Kuo conceived of the study, performed image analysis; and participated in the redaction of the manuscript. Che-Wu Wan and Chao-Chun Hsug carried out immunobloots on nerve tissue. Ying-Jui Ho and Yun-Chi Chiang performed immunocytochemistry experiments on repaired nerves. Fu‑Der Mai and Wei-Jhih Lin participated in the TOF–SIMS analysis. Wen-Chieh, Liao participated in the design and coordination of the study, and drafted the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, CH., Kuo, YC., Wang, CY. et al. Syndecan-3 contributes to the regulation of the microenvironment at the node of Ranvier following end-to‑side neurorrhaphy: sodium image analysis. Histochem Cell Biol 155, 355–367 (2021). https://doi.org/10.1007/s00418-020-01936-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-020-01936-z