Abstract
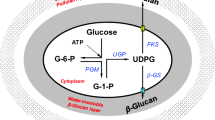
The effects of several surfactants on the biosynthesis of β-1,3-D-glucan (β-glucan) and pullulan by Aureobasidium pullulans CCTCC M 2012259 were investigated, and Triton X-100 was found to decrease biomass formation but increase β-glucan and pullulan production. The addition of 5 g/L Triton X-100 to the fermentation medium and bioconversion broth significantly increased β-glucan production by 76.6% and 69.9%, respectively, when compared to the control without surfactant addition. To reveal the physiological mechanism underlying the effect of Triton X-100 on polysaccharides production, the cell morphology and viability, membrane permeability, key enzyme activities, and intracellular levels of UDPG, NADH, and ATP were determined. The results indicated that Triton X-100 increased the activities of key enzymes involved in β-glucan and pullulan biosynthesis, improved intracellular UDPG and energy supply, and accelerated the transportation rate of precursors across the cell membrane, all of which contributed to the enhanced production of β-glucan and pullulan. Moreover, a two-stage culture strategy with combined processes of batch fermentation and bioconversion was applied, and co-production of β-glucan and pullulan in the presence of 5 g/L Triton X-100 additions was further improved. The present study not only provides insights into the effect of surfactant on β-glucan and pullulan production but also presents a feasible approach for efficient production of analogue exopolysaccharides.
Key points
• Triton X-100 increased β-glucan and pullulan production under either batch fermentation or bioconversion.
• Triton X-100 increased the permeability of cell membrane and accelerated the transportation rate of precursors across cell membrane.
• Activities of key enzymes involved in β-glucan and pullulan biosynthesis were increased in the presence of Triton X-100.
• Intracellular UDPG levels and energy supply were improved by Triton X-100 addition.







Similar content being viewed by others
References
Bulman SE, Coleman CM, Murphy JM, Medcalf N, Ryan AE, Barry F (2015) Pullulan: a new cytoadhesive for cell-mediated cartilage repair. Stem Cell Res Ther 6:34. https://doi.org/10.1186/s13287-015-0011-7
Cankılıç MY, Özaydın Ö, Güven K (2012) Effects of the addition of Triton X-100 for Xanthan production with molasses. Adv Food Sci 34(2):100–105
Chen X, Wang Q, Liu N, Liu G, Chi Z, Chi Z (2017) A glycosyltransferase gene responsible for pullulan biosynthesis in Aureobasidium melanogenum P16. Int J Biol Macromol 95:539–549. https://doi.org/10.1016/j.ijbiomac.2016.11.081
Cheng KC, Demirci A, Catchmark JM (2011) Pullulan: biosynthesis, production, and applications. Appl Microbiol Biotechnol 92(1):29–44. https://doi.org/10.1007/s00253-011-3477-y
Cui JD, Zhang YN (2012) Evaluation of metal ions and surfactants effect on cell growth and exopolysaccharide production in two-stage submerged culture of Cordyceps militaris. Appl Biochem Biotechnol 168(6):1394–1404. https://doi.org/10.1007/s12010-012-9865-7
Du B, Lin C, Bian Z, Xu B (2015) An insight into anti-inflammatory effects of fungal beta-glucans. Trends Food Sci Technol 41:49–59. https://doi.org/10.1016/j.tifs.2014.09.002
Duan X, Chi Z, Wang L, Wang X (2008) Influence of different sugars on pullulan production and activity of α-phosphoglucose mutase UDPG-pyrophosphorylase and glucosyltransferase involved in pullulan synthesis in Aureobasidium pullulans Y68. Carbohydr Polym 73:587–593. https://doi.org/10.1016/j.carbpol.2007.12.028
Duan X, Zou C, Wu J (2015) Triton X-100 enhances the solubility and secretion ratio of aggregation-prone pullulanase produced in Escherichia coli. Bioresource Tech 194:137–143. https://doi.org/10.1016/j.biortech.2015.07.024
Freitas F, Torres CAV, Reis MAM (2017) Engineering aspects of microbial exopolysaccharide production. Bioresource Tech 245:1674–1683. https://doi.org/10.1016/j.biortech.2017.05.092
Jiang LF, Wu SJ, Kim JM (2011) Effect of different nitrogen sources on activities of UDPG-pyrophosphorylase involved in pullulan synthesis and pullulan production by Aureobasidium pullulans. Carbohydr Polym 86(2):1085–1088. https://doi.org/10.1016/j.carbpol.2011.05.016
Jiang H, Xue S, Li Y, Liu G, Chi Z, Hu Z, Chi Z (2018) Efficient transformation of sucrose into high pullulan concentrations by Aureobasidium melanogenum TN1-2 isolated from a natural honey. Food Chem 257:29–35. https://doi.org/10.1016/j.foodchem.2018.03.003
Ju X, Wang D, Zhang G, Cao D, Wei G (2015) Efficient pullulan production by bioconversion using Aureobasidium pullulans as the whole-cell catalyst. Appl Microbiol Biotechnol 99:211–220. https://doi.org/10.1007/s00253-014-6100-1
Kimura A, Arima K, Murata K (1981) Biofunctional change in yeast cell surface on treatment with triton X-100. Agric Biol Chem 45(11):2627–2629. https://doi.org/10.1271/bbb1961.45.2627
Li Y, Chi Z, Wang GY, Wang ZP, Liu GL, Lee CF, Ma ZC, Chi ZM (2015) Taxonomy of Aureobasidium spp. and biosynthesis and regulation of their extracellular polymers. Crit Rev Microbiol 41(2):228–237. https://doi.org/10.3109/1040841X.2013.826176
Li H, Li J, Jiao X, Li K, Sun Y, Zhou W, Shen Y, Qian J, Chang A, Wang J, Zhu H (2019) Characterization of the biosynthetic pathway of nucleotide sugar precursor UDP-glucose during sphingan WL gum production in Sphingomonas sp. WG J Biotechnol 302:1–9. https://doi.org/10.1016/j.jbiotec.2019.06.005
Mao ZC, Shin HD, Chen RRZ (2006) Engineering the E. coli UDP-glucose synthesis pathway for oligosaccharide synthesis. Biotechnol Prog 22(2):369–374. https://doi.org/10.1021/bp0503181
Mirbagheri M, Nahvi I, Emtiazi G, Darvishi F (2011) Enhanced production of citric acid in Yarrowia lipolytica by Triton X-100. Appl Biochem Biotechnol 165(3–4):1068–1074. https://doi.org/10.1007/s12010-011-9325-9
Moriya N, Moriya Y, Nomura H, Kusano K, Asada Y, Uchiyama H, Park EY, Okabe M (2013) Improved β-glucan yield using an Aureobasidium pullulans M-2 mutant strain in a 200-L pilot scale fermentor targeting industrial mass production. Biotechnol Bioprocess Eng 18:1083–1089. https://doi.org/10.1007/s12257-013-0516-9
Pan SK, Yao DR, Chen J, Wu SJ (2013) Influence of controlled pH on the activity of UDPG-pyrophosphorylase in Aureobasidium pullulans. Carbohydr Polym 92(1):629–632. https://doi.org/10.1016/j.carbpol.2012.08.099
Papaspyridi LM, Zerva A, Topakas E (2018) Biocatalytic synthesis of fungal β-glucans. Catalysts 8(7):274. https://doi.org/10.3390/catal8070274
Prajapati VD, Jani GK, Khanda SM (2013) Pullulan: an exopolysaccharide and its various applications. Carbohydr Polym 95:540–549. https://doi.org/10.1016/j.carbpol.2013.02.082
Prasongsuk S, Lotrakul P, Ali I, BankeereeW PH (2018) The current status of Aureobasidium pullulans in biotechnology. Folia Microbiol 63:129–140. https://doi.org/10.1007/s12223-017-0561-4
Rieder A, Ballance S, Böcker U, Knutsen S (2018) Quantification of 1,3-β-D-glucan from yeast added as a functional ingredient to bread. Carbohydr Polym 181:34–42. https://doi.org/10.1016/j.carbpol.2017.09.044
Schimoler-O’Rourke R, Renault S, Mo W, Selitrennikoff CP (2003) Neurospora crassa FKS protein binds to the (1,3) beta-glucan synthase substrate, UDP-glucose. Curr Microbiol 46:408–412. https://doi.org/10.1007/s00284-002-3884-5
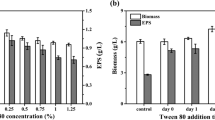
Sheng L, Zhu G, Tong Q (2013) Mechanism study of Tween 80 enhancing the pullulan production by Aureobasidium pullulans. Carbohydr Polym 97:121–123. https://doi.org/10.1016/j.carbpol.2013.04.058
Sheng L, Tang G, Su P, Zhang J, Xiao Q, Tong Q, Ma M (2016) Understanding the influence of Tween 80 on pullulan fermentation by Aureobasidium pullulans CGMCC1234. Carbohydr Polym 136:1332–1337. https://doi.org/10.1016/j.carbpol.2015.10.058
Singh A, van Hamme JD, Ward OP (2007) Surfactants in microbiology and biotechnology: part 2. Application aspects. Biotechnol Adv 25(1):99–121. https://doi.org/10.1016/j.biotechadv.2006.10.004
Singh RS, Kaur N, Rana V, Kennedy JF (2017) Pullulan: a novel molecule for biomedical applications. Carbohydr Polym 171:102–121. https://doi.org/10.1016/j.carbpol.2017.04.089
Singh RS, Kaur N, Kennedy JF (2019) Pullulan production from agro-industrial waste and its applications in food industry: a review. Carbohydr Polym 217:46–57. https://doi.org/10.1016/j.carbpol.2019.04.050
Sugumarana KR, Ponnusami V (2017) Review on production, downstream processing and characterization of microbial pullulan. Carbohydr Polym 173:573–591. https://doi.org/10.1016/j.carbpol.2017.06.022
Taguchi R, Sakano Y, Kikuchi Y, Sakuma M, Kobayashi T (1973) Synthesis of pullulan by acetone-dried cells and cell-free enzyme from Pullularia pullulans, and the participation of lipid intermediate. Agric Biol Chem 37(7):1635–1641. https://doi.org/10.1080/00021369.1973.10860882
Uchiyama H, Iwai A, Dohra H, Ohnishi T, Kato T, Park EY (2018) The effects of gene disruption of Kre6-like proteins on the phenotype of β-glucan-producing Aureobasidium pullulans. Appl Microbiol Biotechnol 102:4467–4475. https://doi.org/10.1007/s00253-018-8947-z
van Hamme JD, Singh A, Ward OP (2006) Physiological aspects. Part 1 in a series of papers devoted to surfactants in microbiology and biotechnology. Biotechnol Adv 24(6):604–620. https://doi.org/10.1016/j.biotechadv.2006.08.001
Wang D, Bian J, Wei G, Jiang M, Dong M (2016a) Simultaneously enhanced production and molecular weight of pullulan using a two-stage agitation speed control strategy. J Chem Technol Biotechnol 91:467–475. https://doi.org/10.1002/jctb.4600
Wang D, Ju X, Zhang G, Wang D, Wei G (2016b) Copper sulfate improves pullulan production by bioconversion using whole cells of Aureobasidium pullulans as the catalyst. Carbohydr Polym 150:209–215. https://doi.org/10.1016/j.carbpol.2016.05.035
Wang D, Ni T, Ju X, Wei G (2018) Sodium chloride improves pullulan production by Aureobasidium pullulans but reduces the molecular weight of pullulan. Appl Microbiol Biotechnol 102:8921–8930. https://doi.org/10.1007/s00253-018-9292-y
Wang D, Zhu C, Zhang G, Wang C, Wei G (2020) Enhanced β-glucan and pullulan production by Aureobasidium pullulans with zinc sulfate supplementation. Appl Microbiol Biotechnol 104:1751–1760. https://doi.org/10.1007/s00253-019-10326-y
Yin H, Gao C, Ye K, Zhao T, Sun A, Qiao C (2019) Evaluation of surfactant effect on β-poly(L-malic acid) production by Aureobasidium pullulans. Biotechnol Biotec Eq 33(1):954–966. https://doi.org/10.1080/13102818.2019.1631718
Yu X, Wang Y, Wei G, Dong Y (2012) Media optimization for elevated molecular weight and mass production of pigment-free pullulan. Carbohydr Polym 89:928–934. https://doi.org/10.1016/j.carbpol.2012.04.038
Zhu F, Du B, Xu B (2016) A critical review on production and industrial applications of beta-glucans. Food Hydrocoll 52:275–288. https://doi.org/10.1016/j.foodhyd.2015.07.003
Funding
This work was supported by the Natural Science Foundation of Jiangsu Province (BK20181440), the National Natural Science Foundation of China (21776189), and a Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Contributions
DHW and GYW conceived and designed research. GLW, AUD, and YSQ conducted experiments. CLW and DHW analyzed the data. GLW and AUD wrote the manuscript. CLW and GYW reviewed and edited the manuscript. DHW and GYW administrated project and acquired funding. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(PDF 488 kb)
Rights and permissions
About this article
Cite this article
Wang, GL., Din, A.U., Qiu, YS. et al. Triton X-100 improves co-production of β-1,3-D-glucan and pullulan by Aureobasidium pullulans. Appl Microbiol Biotechnol 104, 10685–10696 (2020). https://doi.org/10.1007/s00253-020-10992-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10992-3