Abstract
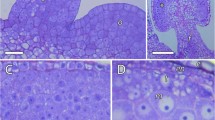
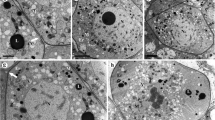
Juncaceae, a cosmopolitan family, belong to the cyperid clade of Poales together with Cyperaceae and Thurniaceae. Pollen grain of Juncaceae, as in Thurniaceae, is dispersed in a permanent tetrad, and knowledge about the ontogeny of its wall is still incipient, based on data from only one species. This study aims to analyze the formation of the pollen wall of seven Juncus species in order to characterize the timing and the ontogenetic events that lead to the cohesion of the four pollen grains in a permanent tetrad. Anthers at different developmental stages were submitted to techniques of light microscopy and transmission electron microscopy; dehiscent anthers with mature pollens were also analyzed in scanning electron microscopy. In all the species here studied, callose deposits around each microsporocyte, with dissolution prior to meiosis. Microspore wall starts at the end of the second meiotic division with formation of primexine. Exine comprises tectum, columellae, and foot layer. During cytokinesis, cell plates form the internal wall of the pollen tetrad. Mature permanent tetrad is enveloped externally by both the exine and intine and internally by the intine and the foot layer, which forms the continuous internal wall. Callose was detected in the early stages of microsporocytes, although reported to be absent in Juncaceae. Our data confirm the variation in Juncaceae cytokinesis and the occurrence of simple cohesion due to the presence of a continuous tectum along the pollen tetrad.





Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the manuscript.
References
Bhojwani SS, Bhatnagar SP (1978) The embryology of angiosperms. Vikas Publishing House PVT, New Delhi
Blackmore S, Crane PR (1988) The systematic implications of pollen and spore ontogeny. In: Humphries CJ (ed) Ontogeny and systematics. Columbia University Press, New York, pp 83–115
Caccavari MA, Galati B (1998) Pollen development in Mimosa balansae. Phytomorphology 48(4):371–381
Chamberlain CJ (1932) Methods in plants histology. The University of Chicago Press, Chicago
Coan AI, Alves MV, Scatena VL (2010) Evidence of pseudomonad pollen formation in Hypolytrum (Mapanioideae, Cyperaceae). Aust J Bot 58:663–672. https://doi.org/10.1071/BT10149
Copenhaver GP (2005) A compendium of plant species producing pollen tetrads. J N C Acad Sci 121(1):17–35
Dahlgren RMT, Clifford HT, Yeo PF (1985) The families of the monocotyledons: structure, evolution and taxonomy. Springer, Berlin
Dunbar A (1973) Pollen development in the Eleocharis palustris group (Cyperaceae). I. Ultrastructure and ontogeny. Bot Notiser 126:197–254
Feder N, O’Brien TP (1968) Plant microtechnique: some principles and new methods. Am J Bot 55(1):123–142. https://doi.org/10.1002/j.1537-2197.1968.tb06952.x
Furness CA, Rudall PJ (1999) Microsporogenesis in monocotyledons. Ann Bot 84:475–499. https://doi.org/10.1006/anbo.1999.0942
Givnish TJ, Ames M, McNeal JR, McKain MR, Steele PR, dePamphilis CW, Graham SW, Pires JC, Stevenson DW, Zomlefer WB, Briggs BG, Durvall MR, Moore MJ, Heaney JM, Soltis DE, Soltis PS, Thiele K, Leebens-Mack JH (2010) Assembling the tree of the monocotyledons: Plastome sequence phylogeny and evolution of Poales. Ann Mo Bot Gard 97(4):584–616. https://doi.org/10.3417/2010023
Godwin H, Echlin P, Chapman B (1967) The development of the pollen grain wall in Ipomoea purpurea (L.) Roth. Rev Palaeobot Palynol 3:181–195. https://doi.org/10.1016/0034-6667(67)90051-6
Harder LD, Johnson SD (2008) Function and evolution of aggregated pollen in angiosperms. Int J Plant Sci 169(1):59–78. https://doi.org/10.1086/523364
Heslop-Harrison J (1963) An ultrastructural study of pollen wall ontogeny in Silene pendula. Grana 4(1):7–24. https://doi.org/10.1080/00173136309437854
Heslop-Harrison J (1964) Cells walls, cell membranes and protoplasmic connections during meiosis and pollen development. In: Linskens HF (ed) Pollen physiology and fertilization. North-Holland Publishing Company, Amsterdam, pp 39–47
Heslop-Harrison J (1968) Pollen wall development. Science 161:230–237. https://doi.org/10.1126/science.161.3838.230
Heslop-Harrison J (1971) The pollen wall: structure and development. In: Heslop-Harrison J (ed) Pollen: development and physiology. Butterworth-Heinemann, London, pp 75–98. https://doi.org/10.1016/B978-0-408-70149-5.50013-0
Hesse M (1981) Pollenkitt and viscin threads: their role in cementing pollen grains. Grana 20(3):145–152. https://doi.org/10.1080/00173138109427656
Izhar S, Frankel R (1971) Mechanism of male sterility in Petunia: the relationship between pH, callase activity in the anthers, and the breakdown of the microsporogenesis. Theor Appl Genet 41(3):104–108. https://doi.org/10.1007/BF00277751
Johansen DA (1940) Plant microtechnique. McGraw-Hill, New York
Karnovsky MJ (1965) A formaldehyde-glutaraldehyde fixative of high osmolality for use in electron microscopy. J Cell Biol 27(2):137A
Kirschner J et al (2002a) Juncaceae 1: Rostkovia to Luzula, Species Plantarum: Flora of the World Part 6. Australian Biological Resources Study, Canberra
Kirschner J et al (2002b) Juncaceae 2: Juncus subg. Juncus. Species Plantarum: Flora of the World Part 7. Australian Biological Resources Study, Canberra
Kirschner J et al (2002c) Juncaceae 3: Juncus subg. Agathryon. Species Plantarum: Flora of the World 8. Australian Biological Resources Study, Canberra
Knox RB (1984) The pollen grain. In: Johri BM (ed) Embryology of angiosperms. Springer-Verlag, Berlin, pp 197–271. https://doi.org/10.1007/978-3-642-69302-1_5
Knox RB, Friederich E (1974) Tetrad pollen grain development and sterility in Leschenaultia formosa (Goodeniaceae). New Phytol 73:251–258. https://doi.org/10.1111/j.1469-8137.1974.tb04622.x
Knox RB, McConchie CA (1986) Structure and function of compound pollen. In: Blackmore S, Ferguson IK (eds) Pollen and spores: form and function. Linnean Society Symposium Series, number 12, Academic Press, London, pp 265–282
Lambert AM (1969) Observations sur la première mitose pollinique de Luzules. C R Séances Acad Sci, Sér D, Sci Nat 269:153–156
Lambert AM (1970) Formation et différenciation des grains de pollen chez les Luzules (Structures et Ultrastructures). Bull Acad Soc Lorraines Sci 9(1):147–149
Linder HP, Rudall PJ (2005) Evolutionary history of Poales. Annu Rev Ecol Evol Syst 36:107–124. https://doi.org/10.1146/annurev.ecolsys.36.102403.135635
Lora J, Testillano PS, Risueño MC, Hormaza JI, Herrero M (2009) Pollen development in Annona cherimola Mill. (Annonaceae). Implications for the evolution of aggregated pollen. BMC Plant Biol 9:129. https://doi.org/10.1186/1471-2229-9-129
Maheshwari P (1950) An introduction to the embryology of angiosperms. McGraw-Hill, New York
Meyer NR, Yaroshevskaya AS (1976) The phylogenetic significance of the development of pollen grain walls in Liliaceae, Juncaceae e Cyperaceae. In: Ferguson IK, Muller J (eds) The evolutionary significance of the exine. Linnean Society Symposium Series, number 1, Academic Press, London, pp 91–100
Nagaraj M, Nijalingappa BHM (1972) Sporogeneses and development of gametophytes in Hypolytrum latifolium L.C. Rich. Curr Sci India 41(7):260–261
Pacini E (1990) Tapetum and microspore function. In: Blackmore S, Knox RB (eds) Microspores: evolution and ontogeny. Academic Press, London, pp 213–237
Reynolds ES (1963) The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol 17(1):208–212. https://doi.org/10.1083/jcb.17.1.208
Rhee SY, Somerville CR (1998) Tetrad pollen formation in quartet mutants of Arabidopsis thaliana is associated with persistence of pectic polysaccharides of the pollen mother cell wall. Plant J 15(1):79–88. https://doi.org/10.1046/j.1365-313X.1998.00183.x
Rhee SY, Osborne E, Poindexter PD, Somerville CR (2003) Microspore separation in the quartet 3 mutants of Arabidopsis is impaired by a defect in a developmentally regulated polygalacturonase required for pollen mother cell wall degradation. Plant Physiol 133:1170–1180. https://doi.org/10.1104/pp.103.028266
Rocha DM, Vanzela ALL, Mariath JEA (2018) Comparative study of microgametogenesis in members of Cyperaceae and Juncaceae: a shift from permanent pollen tetrads to pseudomonads. Bot J Linn Soc 188(1):59–73. https://doi.org/10.1093/botlinnean/boy041
Sampson FB (1977) Pollen tetrads of Hedycarya arborea J. R. et G. Forst (Monimiaceae). Grana 16(2):61–73. https://doi.org/10.1080/00173134.1977.11864640
Selling OH (1947) Studies in Hawaiian pollen statistics. II. The pollen of the Hawaiian phanerogams. Special Publication, Bernice Pauahi Bishop Museum, Bishop Museum, Honolulu, 38:1–430
Simpson D (1995) Relationships within Cyperales. In: Rudall PJ, Cribb PJ, Cutler DF, Humphries CJ (eds) Monocotyledons: systematics and evolution. Royal Botanic Gardens, Kew, pp 497–509
Spurr AR (1969) A low-viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res 26:31–43
Stanley RG, Linskens HF (1974) Pollen: biology, biochemistry, management. Springer-Verlag, Berlin
Takahashi H (1979) Pollen development in Chimaphila japonica Miq. (Pyrolaceae). Sci Rep Tôhoku Univ, Ser 4. Biol 37:263–272
Takahashi H, Sohma K (1980) Pollen development in Pyrola japonica Klenze. Sci Rep Tôhoku Univ, Ser 4. Biol 38:57–71
Takahashi H, Sohma K (1984) Development of pollen tetrad in Typha latifolia L. Pollen Spores 26(1):5–18
Thiers B (2020) [continuously updated] Index Herbariorum: a global directory of public herbaria and associated staff. New York Botanical Garden's Virtual Herbarium. http://sweetgum.nybg.org/science/ih/. Accessed on 04 Oct 2020
Trias-Blasi A, Baker WJ, Haigh AL, Simpson DA, Weber O, Wilkin P (2015) A genus-level phylogenetic linear sequence of monocots. Taxon 64(3):552–581. https://doi.org/10.12705/643.9
Tsou C-H, Fu Y-L (2002) Tetrad pollen formation in Annona (Annonaceae): proexine formation and binding mechanism. Am J Bot 89(5):734–747. https://doi.org/10.3732/ajb.89.5.734
Vijayaraghavan MR, Shukla AK (1977) Absence of callose around the microspore tetrad and poorly developed exine in Pergularia daemia. Ann Bot 41(5):923–926. https://doi.org/10.1093/oxfordjournals.aob.a085388
Walker JW, Doyle JA (1975) The bases of angiosperm phylogeny: palynology. Ann Mo Bot Gard 62:664–723
Watson ML (1958) Staining of tissue sections for electron microscopy with heavy metals. J Biophys Biochem Cytol 4(4):475–478. https://doi.org/10.1083/jcb.4.4.475
Worrall D, Hird DL, Hodge R, Paul W, Draper J, Scott R (1992) Premature dissolution of the microsporocyte callose wall causes male sterility in transgenic tobacco. Plant Cell 4:759–771. https://doi.org/10.1105/tpc.4.7.759
Wulff HD (1939) Die pollenentwicklung der Juncaceen nebst einer auswertung der embryologischen befunde hinsichtlich einer verwandtschaft zwischen den Juncaceen und Cyperaceen. Jahrb Wiss Botanik 87:533–556
Yeung EC (1987) Mechanisms of pollen aggregation into pollinia in Epidendrum ibaguense (Orchidaceae). Grana 26(1):47–52. https://doi.org/10.1080/00173138709428903
Yue L, Kuang Y, Liao J (2017) Ontogeny of permanent tetrads in Gardenia jasminoides (Rubiaceae) provides insight into pollen evolution. Rev Palaeobot Palynol 247:120–132. https://doi.org/10.1016/j.revpalbo.2017.09.004
Acknowledgments
The authors thank Prof. Dr. Elliot Watanabe Kitajima for the permission to use the facilities of Laboratório de Microscopia Eletrônica aplicada à Pesquisa Agropecuária (NAP/MEPA - ESALQ/USP, Piracicaba, São Paulo, Brazil) and Renato Barbosa Salaroli for the technical assistance with the TEM.
Funding
This work was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq – Ph.D. Scholarship process number 141848/2017-0 to F.P.L. and Research Grant process numbers 307515/2015-0 and 309504/2018-0 to A.I.C.) and by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES – PNPD – Finance Code 001 to A.O.).
Author information
Authors and Affiliations
Contributions
The present study is part of the Ph.D. Thesis of the first author. All authors contributed to its conception and design. Material preparation and data collection were performed by Fernanda Passarini Lopes, and analysis was performed by all authors. All authors contributed and approved the final version of the present manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors give their consent for information to be published in Protoplasma.
Code availability
Not applicable.
Additional information
Handling Editor: Benedikt Kost
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Passarini Lopes, F., Oriani, A. & Coan, A.I. Development of the permanent tetrad wall in Juncus L. (Juncaceae, Poales). Protoplasma 258, 495–506 (2021). https://doi.org/10.1007/s00709-020-01583-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-020-01583-0