Abstract
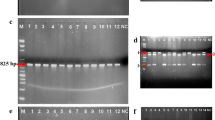
Matrix metalloproteinases (MMPs) are a group of zinc dependent enzymes that are involved in tumor cell invasion and metastasis. The role of MMP-2 and -9 genetic polymorphism in different malignancies has been the subject of numerous studies. The present research has attempted to discover any positive correlation between MMP-2 and MMP-9 SNPs and prostate cancer (PCa) in patients with a history of either diabetes or smoking habits. 112 PCa-patients and 150 unrelated healthy-controls that matched for age and sex were selected for present case–control study. MMP-2 -1575G/A and MMP-9 -1562 C/T polymorphisms detected by PCR–RFLP, serum tissue inhibitors of metalloproteinases (TIMP-1 and TIMP-2), testosterone, prostate-specific antigen (PSA), free-prostate-specific-antigen (fPSA), and fPSA/PSA levels were detected by ELISA and enzyme assay, respectively. MMP-2 and MMP-9 activities were measured by gelatin-zymography. Covariates were considered as age, status of cigarette smoking, and a possible history of diabetes mellitus (DM). The frequency of -1575 MMP-2 A/A + A/G and -1562 MMP-9 C/T + T/T genotypes were higher in PCa-patients with DM (74.3%,p = 0.003) and with smoking habits (72.5%,p = 0.005). These genotypes were associated with the increased risk of prostate cancer in smokers (3.52-folds) and in individuals with history of DM (4.34-folds). A significant positive association was found between level of TIMPs (TIMP -1 and TIMP-2) and BMI in PCa-patients and also between testosterone levels and MMP-9 activity in healthy control subjects. For the first time, this study demonstrated that activities of MMP-2 -1575G/A and MMP-9 -1562C/T variants in association with smoking and diabetes are considered significant risk factors for PCa.

Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M et al (2015) Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 136(5):E359–E386
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer J Clin 68(6):394–424
Gandaglia G, Abdollah F, Schiffmann J, Trudeau V, Shariat SF, Kim SP, Perrotte P, Montorsi F, Briganti A, Trinh QD, Karakiewicz PI, Sun M (2014) Distribution of metastatic sites in patients with prostate cancer: a population-based analysis. Prostate 74(2):210–216
Deryugina EI, Quigley JP (2006) Matrix metalloproteinases and tumor metastasis. Cancer Metastasis Rev 25(1):9–34
Batra J, Soares AS, Mehner C, Radisky ES (2013) Matrix metalloproteinase-10/TIMP-2 structure and analyses define conserved core interactions and diverse exosite interactions in MMP/TIMP complexes. PLoS ONE 8(9):e75836
Patterson ML, Atkinson SJ, Knauper V, Murphy G (2001) Specific collagenolysis by gelatinase A, MMP-2, is determined by the hemopexin domain and not the fibronectin-like domain. FEBS Lett 503(2–3):158–162
Cicco CD, Ravasi L, Zorzino L, Sandri MT, Botteri E, Verweij F et al (2008) Circulating levels of VCAM and MMP-2 may help identify patients with more aggressive prostate cancer. Curr Cancer Drug Targets 8(3):199–206
El-Chaer WK, Moraes CF, Nóbrega OT (2018) Diagnosis and prognosis of prostate cancer from circulating matrix metalloproteinases and inhibitors. J Aging Res. https://doi.org/10.1155/2018/7681039
dos Reis ST, Villanova FE, Andrade PM, Pontes J Jr, de Sousa-Canavez JM, Sañudo A et al (2010) Matrix metalloproteinase-2 polymorphism is associated with prognosis in prostate cancer. UrolOncol 28:624–627
Schveigert D, Valuckas KP, Kovalcis V, Ulys A, Chvatovic G, Didziapetriene J (2013a) Significance of MMP-9 expression and MMP-9 polymorphism in prostate cancer. Tumori 99(4):523–529
Raeeszadeh-Sarmazdeh M, Do LD, Hritz BG (2020) Metalloproteinases and their inhibitors: potential for the development of new therapeutics. Cells 9(5):1313
Munhoz F, Godoy-Santos A, Santos M (2010) MMP-3 polymorphism: genetic marker in pathological processes. Mol Med Rep 3(5):735–740
Yu C, Pan K, Xing D, Liang G, Tan W, Zhang L et al (2002) Correlation between a single nucleotide polymorphism in the matrix metalloproteinase-2 promoter and risk of lung cancer. Cancer Res 62(22):6430–6433
Peng Z, Jia J, Gong W, Gao X, Ma P, Jin Z et al (2017) The association of matrix metalloproteinase-9 promoter polymorphisms with gastric cancer risk: a meta-analysis. Oncotarget 8(58):99024
Zhou Y, Yu C, Miao X, Wang Y, Tan W, Sun T et al (2005) Functional haplotypes in the promoter of matrix metalloproteinase-2 and lung cancer susceptibility. Carcinogenesis 26(6):1117–1121
Grieu F, Li WQ, Iacopetta B (2004) Genetic polymorphisms in the MMP-2 and MMP-9 genes and breast cancer phenotype. Breast Cancer Res Treat 88(3):197–204
Weng H, Zeng XT, Wang XH, Liu TZ, He DL (2017) Genetic association between matrix metalloproteinases gene polymorphisms and risk of prostate cancer: a meta-analysis. Front Physiol 8:975
Bahrehmand F, Vaisi-Raygani A, Kiani A, Rahimi Z, Tavilani H, Navabi S et al (2012) Matrix metalloproteinase-2 functional promoter polymorphism G1575A is associated with elevated circulatory MMP-2 levels and increased risk of cardiovascular disease in systemic lupus erythematosus patients. Lupus 21(6):616–624
Zhang K, Chen X, Zhou J, Yang C, Zhang M, Chao M et al (2017) Association between MMP2-1306 C/T polymorphism and prostate cancer susceptibility: a meta-analysis based on 3906 subjects. Oncotarget 8(27):45020
Medina-González A, Eiró-Díaz N, Fernández-Gómez JM, Ovidio-González L, Jalón-Monzón A, Casas-Nebra J, Escaf-Barmadah S (2020) Comparative analysis of the expression of metalloproteases (MMP-2, MMP-9, MMP-11 and MMP-13) and the tissue inhibitor of metalloprotease 3 (TIMP-3) between previous negative biopsies and radical prostatectomies. ActasUrolEsp 44(2):78–85
Zhang X, Miao X, Xiong P, Yu C, Tan W, Qu S et al (2004) Association of functional polymorphisms in matrix metalloproteinase-2 (MMP-2) and MMP-9 genes with risk of gastric cancer in a Chinese population. Chin J Cancer 23(11):1233–1237
Sfar S, Saad H, Mosbah F, Gabbouj S, Chouchane L (2007) TSP1 and MMP9 genetic variants in sporadic prostate cancer. Cancer Genet Cytogenet 172(1):38–44
Gann PH (2002) Risk factors for prostate cancer. Rev Urol 4(Suppl 5):S3–S10
Brawley OW (2012) Trends in Prostate Cancer in the United States. J Nat Cancer Inst 2012(45):152–156
Calle EE, Rodriguez C, Walker-Thurmond K, Thun MJ (2003) Overweight, obesity, and mortality from cancer in a prospectively studied cohort of US adults. N Engl J Med 348(17):1625–1638
McGreevy KM, Hoel BD, Lipsitz SR, Hoel DG (2007) Impact ofnutrients on insulin-like growth factor-I, insulin-like growthfactor binding protein-3 and their ratio in African American and white males. Public Health Nutr 10(1):97–105
Chen CB, Eskin M, Eurich DT, Majumdar SR, Johnson JA (2018) Metformin, Asian ethnicity and risk of prostate cancer in type 2 diabetes: a systematic review and meta-analysis. BMC Cancer 18(1):65
Crawley D, Chamberlain F, Garmo H, Rudman S, Zethelius B, Holmberg L et al (2018) A systematic review of the literature exploring the interplay between prostate cancer and type two diabetes mellitus. Ecancermedicalscience 12:802
Plaskon DF, Penson TL, Vaughan JL (2003) Stanford;cigarette smoking and risk of prostate cancer in middle-aged men. Cancer EpidemiolPrevBiomark 12(7):604–609
Lu X, Yamano Y, Takahashi H, Koda M, Fujiwara Y, Hisada A et al (2015) Associations between estrogen receptor genetic polymorphisms, smoking status, and prostate cancer risk: a case-control study in Japanese men. Environ Health Prev Med 20(5):332–337
Fowler JE Jr., Whitmore WF Jr. (1981) The response of metastatic adenocarcinoma of the prostate to exogenous testosterone. J Urol 126(3):372–375
Schweizer MT, Antonarakis ES, Wang H, Ajiboye AS, Spitz A, Cao H et al (2015) Effect of bipolar androgen therapy for asymptomatic men with castration-resistant prostate cancer: results from a pilot clinical study. SciTransl Med 7(269):269ra2
dos Reis ST et al (2010) Matrix metalloproteinase-2 polymorphism is associated with prognosis in prostate cancer. UrolOncol 28(6):624–627
Schveigert D et al (2013) Significance of MMP-9 expression and MMP-9 polymorphism in prostate cancer. Tumori J 99(4):523–529
Bultitude MF (2012) Campbell-walsh urology tenth edition. BjuInt 109(3):E10
Barakzai MA (2019) Prostatic adenocarcinoma: A grading from Gleason to the new grade-group system: a historical and critical review. Asian Pac J Cancer P: APJCP 20(3):661
World Health Organization (2006) Definition and diagnosis of diabetes mellitus and intermediate hyperglycaemia: report of a WHO/IDF consultation. World Health Organisation, Geneva
Saedi M, Vaisi-Raygani A, Khaghani S, Shariftabrizi A, Rezaie M, Pasalar P, Rahimi Z, Pourmotabbed T (2012) Matrix metalloproteinase-9 functional promoter polymorphism 1562C>T increased risk of early-onset coronary artery disease. MolBiol Rep 39(1):555–562
Vaisi-Raygani A, Rahimi Z, Entezami H, Kharrazi H, Bahrhemand F, Tavilani H et al (2008) Butyrylcholinesterase K variants increase the risk of coronary artery disease in the population of western Iran. Scand J Clin Lab Inv 68(2):123–129
Bahrehmand F, Vaisi-Raygani A, Kiani A, Rahimi Z, Tavilani H, Ardalan M, Vaisi-Raygani H, Shakiba E, Pourmotabbed T (2015) Matrix metalloproteinase 9 polymorphisms and systemic lupus erythematosus: correlation with systemic inflammatory markers and oxidative stress. Lupus 24(6):597–605
Masuhara K, Nakai T, Yamaguchi K, Yamasaki S, Sasaguri Y (2002) Significant increases in serum and plasma concentrations of matrix metalloproteinases 3 and 9 in patients with rapidly destructive osteoarthritis of the hip. Arthritis Rheum 46(10):2625–2631
Xie T, Dong B, Yan Y, Hu G, Xu Y (2016) Association between MMP-2 expression and prostate cancer: a meta-analysis. Biomed Rep 4(2):241–245
Schveigert D, Valuckas KP, Kovalcis V, Ulys A, Chvatovic G, Didziapetriene J (2013b) Significance of MMP-9 expression and MMP-9 polymorphism in prostate cancer. Tumori J 99(4):523–529
Murray NP, Reyes E, Tapia P, Badínez L, Orellana N (2012) Differential expression of matrix metalloproteinase-2 expression in disseminated tumor cells and micrometastasis in bone marrow of patients with nonmetastatic and metastatic prostate cancer: theoretical considerations and clinical implications—an immunocytochemical study. Bone Marrow Res. https://doi.org/10.1155/2012/259351
Maral S, Acar M, Balcik OS, Uctepe E, Hatipoglu OF, Akdeniz D et al (2015) Matrix metalloproteinases 2 and 9 polymorphism in patients with myeloproliferative diseases: a STROBE-compliant observational study. Medicine (Baltimore) 94(1):e732
Dong Z, Nemeth JA, Cher ML, Palmer KC, Bright RC, Fridman R (2001) Differential regulation of matrix metalloproteinase-9, tissue inhibitor of metalloproteinase 1 (TIMP 1) and TIMP 2 expression in cocultures of prostate cancer and stromal cells. Int J Cancer 93(4):507–515
Wood M, Fudge K, Mohler J, Frost A, Garcia F, Wang M et al (1997) In situ hybridization studies of metalloproteinases 2 and 9 and TIMP-1 and TIMP-2 expression in human prostate cancer. ClinExp Metastasis 15(3):246–258
Dos Reis ST, Pontes J Jr, Villanova FE, de Andrade Borra PM, Antunes AA, Dall’oglio MF et al (2009) Genetic polymorphisms of matrix metalloproteinases: susceptibility and prognostic implications for prostate cancer. J Urol 181(5):2320–2325
Srivastava P, Lone TA, Kapoor R, Mittal RD (2012) Association of Promoter Polymorphisms in MMP2 and TIMP2 with Prostate Cancer Susceptibility in North India. Arch Med Res 43(2):117–124
Acknowledgements
This study was performed in partial fulfillment of requirements for an MSc degree in the Clinical Biochemistry, Kermanshah University of Medical Sciences, Kermanshah Iran (M. Kamankash). This study was funded by Kermanshah University of Medical Sciences, Kermanshah, Iran (Grant #96430). The Ethics Committee of Kermanshah University of Medical Sciences, Iran approved the present study (1.Research involving Human Participants 2. Informed consent ethical legal cod KUMS.Rec.1396.136)) (Grant #96430). All participants gave informed written consent for use of their samples and clinical data in research.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
There are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kiani, A., Kamankesh, M., Vaisi-Raygani, A. et al. Activities and polymorphisms of MMP-2 and MMP-9, smoking, diabetes and risk of prostate cancer. Mol Biol Rep 47, 9373–9383 (2020). https://doi.org/10.1007/s11033-020-05968-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-020-05968-5