Abstract
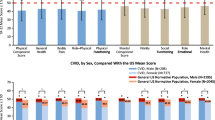
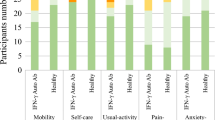
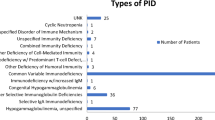
Health-related quality of life (HRQOL) is an emerging topic of interest in patients with immunodeficiency. Information about HRQOL in common variable immunodeficiency (CVID) is limited. The primary objective was to compare primary immunodeficiency disease (PIDD) patients with and without common variable immunodeficiency (CVID) on HRQOL domains using Patient-Reported Outcomes Measurement Information System (PROMIS-29) survey data from the United States Immunodeficiency Network (USIDNET) registry. The primary endpoint variables were scores on 7 HRQOL domains. The USIDNET registry was used to select patients with baseline PROMIS-29 data collected between 2015 and 2018. Descriptive statistics, Fisher’s exact test, and Student’s two-sample t test were used to compare patients with CVID versus patients with non-CVID on demographic and clinical characteristics. The single-sample t test was used to compare sample means to the normed population mean of 50. A general linear model approach to multiple regression with backward selection was used to remove factors that did not contribute significant information to the multivariable models, while controlling for multiple testing. Potential explanatory variables included group (CVID/non-CVID), sex, age, and BMI. Among 184 PIDD patients, 146 (79%) were diagnosed with CVID. Patients had a mean (SD) age of 53 (13.8), were predominantly female (83%), and were Caucasian (98%). PROMIS-29 results revealed a significant effect of group (CVID/non-CVID) on the anxiety, fatigue, and social participation domains, with fatigue being the most statistically significant. Fatigue, anxiety, and social participation may be key factors influencing HRQOL among patients with CVID. Future prospective longitudinal studies using PROMIS-29 will be needed to confirm these findings and to determine the mechanisms through which these factors develop in CVID, and how they can be improved.


Similar content being viewed by others
Abbreviations
- HRQOL:
-
Health-related quality of life
- CVID:
-
Common variable immunodeficiency
- PROMIS-29:
-
Patient-Reported Outcomes Measurement Information System
- PIDD:
-
Primary immunodeficiency diseases
- USIDNET:
-
The US Immunodeficiency Network
- IDF:
-
Immune Deficiency Foundation
- IGRT:
-
Immunoglobulin replacement therapy
- IRB:
-
Institutional Review Board
- BMI:
-
Body mass index
- IVIG:
-
Intravenous IgG
- SCIG:
-
Subcutaneous IgG
References
Hammarstrom L, Vorechovsky I, Webster D. Selective IgA deficiency (SIgAD) and common variable immunodeficiency (CVID). Clin Exp Immunol. 2000;120(2):225–31.
Cunningham-Rundles C, Bodian C. Common variable immunodeficiency: clinical and immunological features of 248 patients. Clin Immunol. 1999;92(1):34–48.
Jolles S, Orange JS, Gardulf A, Stein MR, Shapiro R, Borte M, et al. Current treatment options with immunoglobulin G for the individualization of care in patients with primary immunodeficiency disease. Clin Exp Immunol. 2015;179(2):146–60.
Shearer WT, Notarangelo LD, Griffith LM. Treatment of immunodeficiency: long-term outcome and quality of life. J Allergy Clin Immunol. 2008;122(6):1065–8.
Titman P, Allwood Z, Gilmour C, Malcolmson C, Duran-Persson C, Cale C, et al. Quality of life in children with primary antibody deficiency. J Clin Immunol. 2014;34(7):844–52.
Cole T, McKendrick F, Titman P, Cant AJ, Pearce MS, Cale CM, et al. Health related quality of life and emotional health in children with chronic granulomatous disease: a comparison of those managed conservatively with those that have undergone haematopoietic stem cell transplant. J Clin Immunol. 2012;33(1):8–13.
Jiang F, Torgerson TR, Ayars AG. Health-related quality of life in patients with primary immunodeficiency disease. Allergy, Asthma Clin Immunol. 2015;11:27.
Soresina A, Nacinovich R, Bomba M, Cassani M, Molinaro A, Sciotto A, et al. The quality of life of children and adolescents with X-linked agammaglobulinemia. J Clin Immunol. 2009;29(4):501–7.
Tabolli S, Giannantoni P, Pulvirenti F, La Marra F, Granata G, Milito C, et al. Longitudinal study on health-related quality of life in a cohort of 96 patients with common variable immune deficiencies. Front Immunol. 2014;5:605.
Quinti I, Di Pietro C, Martini H, Pesce AM, Lombardi F, Baumghartner M, et al. Health related quality of life in common variable immunodeficiency. Yonsei Med J. 2012;53(3):603–10.
Rider NL, Kutac C, Hajjar J, Scalchunes C, Seeborg FO, Boyle M, et al. Health-related quality of life in adult patients with common variable immunodeficiency disorders and impact of treatment. J Clin Immunol. 2017;37(5):461–75.
Lopez-Perez P, Miranda-Novales G, Segura-Mendez NH, Del Rivero-Hernandez L, Cambray-Gutierrez C, Chavez-Garcia A. Study of quality of life in adults with common variable immunodeficiency by using the Questionnaire SF-36. Revista alergia Mexico (Tecamachalco, Puebla, Mexico : 1993). 2014;61(2):52–8.
About USIDNET 2018 [Available from: https://usidnet.org/usidnet-mission/. Accessed 01 June 2018
Validation 2020 [Available from: https://www.healthmeasures.net/explore-measurement-systems/promis/measure-development-research/validation. Accessed 01 Sep 2020
Cella D, Yount S, Rothrock N, Gershon R, Cook K, Reeve B, et al. The Patient-Reported Outcomes Measurement Information System (PROMIS): progress of an NIH roadmap cooperative group during its first two years. Med Care. 2007;45(5 Suppl 1):S3–S11.
Klem M, Saghafi E, Abromitis R, Stover A, Dew MA, Pilkonis P. Building PROMIS item banks: librarians as co-investigators. Qual Life Res. 2009;18(7):881–8.
Cella D, Riley W, Stone A, Rothrock N, Reeve B, Yount S, et al. The Patient-Reported Outcomes Measurement Information System (PROMIS) developed and tested its first wave of adult self-reported health outcome item banks: 2005-2008. J Clin Epidemiol. 2010;63(11):1179–94.
Rothrock NE, Hays RD, Spritzer K, Yount SE, Riley W, Cella D. Relative to the general US population, chronic diseases are associated with poorer health-related quality of life as measured by the Patient-Reported Outcomes Measurement Information System (PROMIS). J Clin Epidemiol. 2010;63(11):1195–204.
Schnall R, Liu J, Cho H, Hirshfield S, Siegel K, Olender S. A health-related quality-of-life measure for use in patients with HIV: a validation study. AIDS Patient Care STDs. 2017;31(2):43–8.
Reeve BB, Hays RD, Bjorner JB, Cook KF, Crane PK, Teresi JA, et al. Psychometric evaluation and calibration of health-related quality of life item banks: plans for the Patient-Reported Outcomes Measurement Information System (PROMIS). Med Care. 2007;45(5 Suppl 1):S22–31.
Cook KF, Jensen SE, Schalet BD, Beaumont JL, Amtmann D, Czajkowski S, et al. PROMIS measures of pain, fatigue, negative affect, physical function, and social function demonstrated clinical validity across a range of chronic conditions. J Clin Epidemiol. 2016;73:89–102.
Craig BM, Reeve BB, Brown PM, Cella D, Hays RD, Lipscomb J, et al. US valuation of health outcomes measured using the PROMIS-29. Value Health. 2014;17(8):846–53.
Hanmer J, Dewitt B, Yu L, Tsevat J, Roberts M, Revicki D, et al. Cross-sectional validation of the PROMIS-Preference scoring system. PLoS One. 2018;13(7):e0201093.
New Quality of Life Survey Available on IDF ePHR 2018 [Available from: https://primaryimmune.org/promis-29. Accessed 01 Sep 2020
PROMIS–29 Profile v2.1: PROMIS Health Organization (PHO) 2018 [updated 07 February 2018. Available from: http://www.healthmeasures.net/administrator/components/com_instruments/uploads/PROMIS-29%20Profile%20v2.1%202-7-2018.pdf. Accessed 01 Sep 2020
Ballow M, Conaway MR, Sriaroon P, Rachid RA, Seeborg FO, Duff CM, et al. Construction and validation of a novel disease-specific quality-of-life instrument for patients with primary antibody deficiency disease (PADQOL-16). J Allergy Clin Immunol. 2017;139(6):2007–10.e8.
Available Translations 2020 [Available from: http://www.healthmeasures.net/explore-measurement-systems/promis/intro-to-promis/available-translations. Accessed 01 Sep 2020
PROMIS Profile scoring manual 2015 [Available from: https://www.assessmentcenter.net/documents/PROMIS%20Profile%20Scoring%20Manual.pdf. Accessed 01 Sep 2020
Health Measures: Intrepret scores: PROMIS 2019 [Available from: http://www.healthmeasures.net/score-and-interpret/interpret-scores/promis. Accessed 01 Sep 2020
Hajjar J, Guffey D, Minard CG, Orange JS. Increased incidence of fatigue in patients with primary immunodeficiency disorders: prevalence and associations within the US Immunodeficiency Network Registry. J Clin Immunol. 2017;37(2):153–65.
Hajjar J, Kutac C, Rider NL, Seeborg FO, Scalchunes C, Orange J. Fatigue and the wear-off effect in adult patients with common variable immunodeficiency. Clin Exp Immunol. 2018;194(3):327–38.
Hjollund NH, Andersen JH, Bech P. Assessment of fatigue in chronic disease: a bibliographic study of fatigue measurement scales. Health Qual Life Outcomes. 2007;5:12.
Swain MG. Fatigue in chronic disease. Clin Sci. 2000;99(1):1–8.
Bradley AS, Ford B, Bansal AS. Altered functional B cell subset populations in patients with chronic fatigue syndrome compared to healthy controls. Clin Exp Immunol. 2013;172(1):73–80.
Heath J, Lehman E, Saunders EF, Craig T. Anxiety and depression in adults with primary immunodeficiency: how much do these patients experience and how much do they attribute to their primary immunodeficiency? Allergy Asthma Proc. 2016;37(5):409–15.
Quinti I, Pulvirenti F, Giannantoni P, Hajjar J, Canter DL, Milito C, et al. Development and initial validation of a questionnaire to measure health-related quality of life of adults with common variable immune deficiency: the CVID_QoL Questionnaire. J Allergy Clin Immunol Pract. 2016;4(6):1169–79.e4.
Ruffner MA, Sullivan KE. Complications associated with underweight primary immunodeficiency patients: prevalence and associations within the USIDNET registry. J Clin Immunol. 2018;38(3):283–93.
Sponholtz TR, van den Heuvel ER, Xanthakis V, Vasan RS. Association of variability in body mass index and metabolic health with cardiometabolic disease risk. J Am Heart Assoc. 2019;8(7):e010793.
Barton JC, Bertoli LF, Barton JC, Acton RT. Fibromyalgia in 300 adult index patients with primary immunodeficiency. Clin Exp Rheumatol. 2017;35 Suppl 105(3):68–73.
Thygesen LC, Ersboll AK. When the entire population is the sample: strengths and limitations in register-based epidemiology. Eur J Epidemiol. 2014;29(8):551–8.
Bonilla FA, Barlan I, Chapel H, Costa-Carvalho BT, Cunningham-Rundles C, de la Morena MT, et al. International Consensus document (ICON): common variable immunodeficiency disorders. J Allergy Clin Immunol Pract. 2016;4(1):38–59.
Berger M. Subcutaneous administration of IgG. Immunol Allergy Clin N Am. 2008;28(4):779–802 viii.
Rojavin MA, Hubsch A, Lawo JP. Quantitative evidence of wear-off effect at the end of the intravenous IgG (IVIG) dosing cycle in primary immunodeficiency. J Clin Immunol. 2016;36(3):210–9.
Acknowledgments
We would like to acknowledge The US Immunodeficiency Network (USIDNET), a program of the Immune Deficiency Foundation (IDF), for their help in query submission and data analysis. The USIDNET is supported by a cooperative agreement, U24AI86837, from the National Institute of Allergy and Infectious Diseases (NIAID).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, S., Kline, M., Fuleihan, R.L. et al. PROMIS-29 survey confirms major impact of fatigue on health-related quality of life in common variable immunodeficiency. Immunol Res 68, 379–388 (2020). https://doi.org/10.1007/s12026-020-09162-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12026-020-09162-4