Abstract
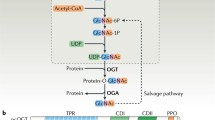
O-GlcNAcylation is an atypical, reversible, and dynamic glycosylation that plays a critical role in maintaining the normal physiological functions of cells by regulating various biological processes such as signal transduction, proteasome activity, apoptosis, autophagy, transcription, and translation. It can also respond to environmental changes and physiological signals to play the role of “stress receptor” and “nutrition sensor” in a variety of stress responses and biological processes. Even, a homeostatic disorder of O-GlcNAcylation may cause many diseases. Therefore, O-GlcNAcylation and its regulatory role in stress response are reviewed in this paper.



Similar content being viewed by others
References
Akan I, Olivier-Van Stichelen S, Bond MR, Hanover JA (2018) Nutrient-driven O-GlcNAc in proteostasis and neurodegeneration. J Neurochem 144:7–34. https://doi.org/10.1111/jnc.14242
Akella NM, le Minh G, Ciraku L, Mukherjee A, Bacigalupa ZA, Mukhopadhyay D, Sodi VL, Reginato MJ (2020) O-GlcNAc transferase regulates cancer stem-like potential of breast cancer cells. Mol Cancer Res 18:585–598. https://doi.org/10.1158/1541-7786.Mcr-19-0732
Allison DF, Wamsley JJ, Kumar M, Li D, Gray LG, Hart GW, Jones DR, Mayo MW (2012) Modification of RelA by O-linked N-acetylglucosamine links glucose metabolism to NF-κB acetylation and transcription. Proc Natl Acad Sci U S A 109:16888–16893. https://doi.org/10.1073/pnas.1208468109
Alonso J, Schimpl M, van Aalten DM (2014) O-GlcNAcase: promiscuous hexosaminidase or key regulator of O-GlcNAc signaling? J Biol Chem 289:34433–34439. https://doi.org/10.1074/jbc.R114.609198
An Y, Varma VR, Varma S, Casanova R, Dammer E, Pletnikova O, Chia CW, Egan JM, Ferrucci L, Troncoso J, Levey AI, Lah J, Seyfried NT, Legido-Quigley C, O'Brien R, Thambisetty M (2018) Evidence for brain glucose dysregulation in Alzheimer's disease. Alzheimers Dement 14:318–329. https://doi.org/10.1016/j.jalz.2017.09.011
Aquino-Gil M, Pierce A, Perez-Cervera Y, Zenteno E, Lefebvre T (2017) OGT: a short overview of an enzyme standing out from usual glycosyltransferases. Biochem Soc Trans 45:365–370. https://doi.org/10.1042/bst20160404
Ball LE, Berkaw MN, Buse MG (2006) Identification of the major site of O-linked beta-N-acetylglucosamine modification in the C terminus of insulin receptor substrate-1. Mol Cell Proteomics 5:313–323. https://doi.org/10.1074/mcp.M500314-MCP200
Banerjee PS, Lagerlöf O, Hart GW (2016) Roles of O-GlcNAc in chronic diseases of aging. Mol Asp Med 51:1–15. https://doi.org/10.1016/j.mam.2016.05.005
Barkovskaya A, Seip K, Hilmarsdottir B, Maelandsmo GM, Moestue SA, Itkonen HM (2019) O-GlcNAc Transferase inhibition differentially affects breast cancer subtypes. Sci Rep 9:5670. https://doi.org/10.1038/s41598-019-42153-6
Bond MR, Hanover JA (2013) O-GlcNAc cycling: a link between metabolism and chronic disease. Annu Rev Nutr 33:205–229. https://doi.org/10.1146/annurev-nutr-071812-161240
Bond MR, Hanover JA (2015) A little sugar goes a long way: the cell biology of O-GlcNAc. J Cell Biol 208:869–880. https://doi.org/10.1083/jcb.201501101
Buse MG, Robinson KA, Marshall BA, Hresko RC, Mueckler MM (2002) Enhanced O-GlcNAc protein modification is associated with insulin resistance in GLUT1-overexpressing muscles. Am J Physiol Endocrinol Metab 283:E241–E250. https://doi.org/10.1152/ajpendo.00060.2002
Chen PH, Chi JT, Boyce M (2018) Functional crosstalk among oxidative stress and O-GlcNAc signaling pathways. Glycobiology 28:556–564. https://doi.org/10.1093/glycob/cwy027
Chen R, Gong P, Tao T, Gao Y, Shen J, Yan Y, Duan C, Wang J, Liu X (2017) O-GlcNAc glycosylation of nNOS promotes neuronal apoptosis following glutamate excitotoxicity. Cell Mol Neurobiol 37:1465–1475. https://doi.org/10.1007/s10571-017-0477-1
Chen Y, Zhao X, Wu H (2019) Metabolic stress and cardiovascular disease in diabetes mellitus: the role of protein O-GlcNAc modification. Arterioscler Thromb Vasc Biol 39:1911–1924. https://doi.org/10.1161/atvbaha.119.312192
Choi H, Kim C, Song H, Cha MY, Cho HJ, Son SM, Kim HJ, Mook-Jung I (2019) Amyloid β-induced elevation of O-GlcNAcylated c-Fos promotes neuronal cell death. Aging Cell 18:e12872. https://doi.org/10.1111/acel.12872
Clarke AJ, Hurtado-Guerrero R, Pathak S, Schüttelkopf AW, Borodkin V, Shepherd SM, Ibrahim AFM, van Aalten DMF (2008) Structural insights into mechanism and specificity of O-GlcNAc transferase. EMBO J 27:2780–2788. https://doi.org/10.1038/emboj.2008.186
Cork GK, Thompson J, Slawson C (2018) Real talk: the inter-play between the mTOR, AMPK, and hexosamine biosynthetic pathways in cell signaling. Front Endocrinol (Lausanne) 9:522. https://doi.org/10.3389/fendo.2018.00522
Cui YL, Xue RQ, Xi He, Ming Zhao, Yu XJ, Liu LZ, Wu Q, Si Yang, Li DL, Zang WJ (2019) Cholinergic drugs ameliorate endothelial dysfunction by decreasing O-GlcNAcylation via M3 AChR-AMPK-ER stress signaling. Life Sci 222:1–12. https://doi.org/10.1016/j.lfs.2019.02.036
D'Alessandris C, Andreozzi F, Federici M, Cardellini M, Brunetti A, Ranalli M, del Guerra S, Lauro D, del Prato S, Marchetti P, Lauro R, Sesti G (2004) Increased O-glycosylation of insulin signaling proteins results in their impaired activation and enhanced susceptibility to apoptosis in pancreatic beta-cells. FASEB J 18:959–961. https://doi.org/10.1096/fj.03-0725fje
Dahl E, Sadr-Nabavi A, Klopocki E, Betz B, Grube S, Kreutzfeld R, Himmelfarb M, An HX, Gelling S, Klaman I, Hinzmann B, Kristiansen G, Grützmann R, Kuner R, Petschke B, Rhiem K, Wiechen K, Sers C, Wiestler O, Schneider A, Höfler H, Nährig J, Dietel M, Schäfer R, Rosenthal A, Schmutzler R, Dürst M, Meindl A, Niederacher D (2005) Systematic identification and molecular characterization of genes differentially expressed in breast and ovarian cancer. J Pathol 205:21–28. https://doi.org/10.1002/path.1687
Deplus R, Delatte B, Schwinn MK, Defrance M, Méndez J, Murphy N, Dawson MA, Volkmar M, Putmans P, Calonne E, Shih AH, Levine RL, Bernard O, Mercher T, Solary E, Urh M, Daniels DL, Fuks F (2013) TET2 and TET3 regulate GlcNAcylation and H3K4 methylation through OGT and SET1/COMPASS. EMBO J 32:645–655. https://doi.org/10.1038/emboj.2012.357
Draime A, Bridoux L, Belpaire M, Pringels T, Degand H, Morsomme P, Rezsohazy R (2018) The O-GlcNAc transferase OGT interacts with and post-translationally modifies the transcription factor HOXA1. FEBS Lett 592:1185–1201. https://doi.org/10.1002/1873-3468.13015
Duan F, Wu H, Jia D, Wu W, Ren S, Wang L, Song S, Guo X, Liu F, Ruan Y, Gu J (2018) O-GlcNAcylation of RACK1 promotes hepatocellular carcinogenesis. J Hepatol 68:1191–1202. https://doi.org/10.1016/j.jhep.2018.02.003
Ducheix S, Magré J, Cariou B, Prieur X (2018) Chronic O-GlcNAcylation and diabetic cardiomyopathy: the bitterness of glucose. Front Endocrinol (Lausanne) 9:642. https://doi.org/10.3389/fendo.2018.00642
Efimova EV, Appelbe OK, Ricco N, Lee SSY, Liu Y, Wolfgeher DJ, Collins TN, Flor AC, Ramamurthy A, Warrington S, Bindokas VP, Kron SJ (2019) O-GlcNAcylation enhances double-Strand break repair, promotes cancer cell proliferation, and prevents therapy-induced senescence in irradiated tumors. Mol Cancer Res 17:1338–1350. https://doi.org/10.1158/1541-7786.Mcr-18-1025
Elsen NL, Patel SB, Ford RE, Hall DL, Hess F, Kandula H, Kornienko M, Reid J, Selnick H, Shipman JM, Sharma S, Lumb KJ, Soisson SM, Klein DJ (2017) Insights into activity and inhibition from the crystal structure of human O-GlcNAcase. Nat Chem Biol 13:613–615. https://doi.org/10.1038/nchembio.2357
Gao Y, Wells L, Comer FI, Parker GJ, Hart GW (2001) Dynamic O-glycosylation of nuclear and cytosolic proteins: cloning and characterization of a neutral, cytosolic beta-N-acetylglucosaminidase from human brain. J Biol Chem 276:9838–9845. https://doi.org/10.1074/jbc.M010420200
Goldberg H, Whiteside C, Fantus IG (2011) O-linked β-N-acetylglucosamine supports p38 MAPK activation by high glucose in glomerular mesangial cells. Am J Physiol Endocrinol Metab 301:E713–E726. https://doi.org/10.1152/ajpendo.00108.2011
Gong J, Jing L (2011) Glutamine induces heat shock protein 70 expression via O-GlcNAc modification and subsequent increased expression and transcriptional activity of heat shock factor-1. Minerva Anestesiol 77:488–495
Groves JA, Lee A, Yildirir G, Zachara NE (2013) Dynamic O-GlcNAcylation and its roles in the cellular stress response and homeostasis. Cell Stress Chaperones 18:535–558. https://doi.org/10.1007/s12192-013-0426-y
Groves JA, Maduka AO, O'Meally RN, Cole RN, Zachara NE (2017) Fatty acid synthase inhibits the O-GlcNAcase during oxidative stress. J Biol Chem 292:6493–6511. https://doi.org/10.1074/jbc.M116.760785
Guinez C, Mir AM, Dehennaut V, Cacan R, Harduin-Lepers A, Michalski JC, Lefebvre T (2008) Protein ubiquitination is modulated by O-GlcNAc glycosylation. FASEB J 22:2901–2911. https://doi.org/10.1096/fj.07-102509
Guinez C, Mir AM, Leroy Y, Cacan R, Michalski JC, Lefebvre T (2007) Hsp70-GlcNAc-binding activity is released by stress, proteasome inhibition, and protein misfolding. Biochem Biophys Res Commun 361:414–420. https://doi.org/10.1016/j.bbrc.2007.07.020
Han C, Gu Y, Shan H, Mi W, Sun J, Shi M, Zhang X, Lu X, Han F, Gong Q, Yu W (2017) O-GlcNAcylation of SIRT1 enhances its deacetylase activity and promotes cytoprotection under stress. Nat Commun 8:1491. https://doi.org/10.1038/s41467-017-01654-6
Hanover JA, Chen W, Bond MR (2018) O-GlcNAc in cancer: An Oncometabolism-fueled vicious cycle. J Bioenerg Biomembr 50:155–173. https://doi.org/10.1007/s10863-018-9751-2
Hanover JA, Krause MW, Love DC (2010) The hexosamine signaling pathway: O-GlcNAc cycling in feast or famine. Biochim Biophys Acta 1800:80–95. https://doi.org/10.1016/j.bbagen.2009.07.017
Hanover JA, Yu S, Lubas WB, Shin SH, Ragano-Caracciola M, Kochran J, Love DC (2003) Mitochondrial and nucleocytoplasmic isoforms of O-linked GlcNAc transferase encoded by a single mammalian gene. Arch Biochem Biophys 409:287–297. https://doi.org/10.1016/s0003-9861(02)00578-7
Hardivillé S, Hart GW (2014) Nutrient regulation of signaling, transcription, and cell physiology by O-GlcNAcylation. Cell Metab 20:208–213. https://doi.org/10.1016/j.cmet.2014.07.014
Hart GW, Housley MP, Slawson C (2007) Cycling of O-linked beta-N-acetylglucosamine on nucleocytoplasmic proteins. Nature 446:1017–1022. https://doi.org/10.1038/nature05815
Hart GW, Slawson C, Ramirez-Correa G, Lagerlof O (2011) Cross talk between O-GlcNAcylation and phosphorylation: roles in signaling, transcription, and chronic disease. Annu Rev Biochem 80:825–858. https://doi.org/10.1146/annurev-biochem-060608-102511
He Y, Ma X, Li D, Hao J (2017) Thiamet G mediates neuroprotection in experimental stroke by modulating microglia/macrophage polarization and inhibiting NF-κB p65 signaling. J Cereb Blood Flow Metab 37:2938–2951. https://doi.org/10.1177/0271678x16679671
Hong SY, Hagen T (2015) 2-Deoxyglucose induces the expression of thioredoxin interacting protein (TXNIP) by increasing O-GlcNAcylation - implications for targeting the Warburg effect in cancer cells. Biochem Biophys Res Commun 465:838–844. https://doi.org/10.1016/j.bbrc.2015.08.097
Hoter A, Amiri M, Prince A, Amer H, Warda M, Naim HY (2018) Differential glycosylation and modulation of camel and human HSP isoforms in response to thermal and hypoxic stresses. Int J Mol Sci 19. https://doi.org/10.3390/ijms19020402
Hrit J et al (2018) OGT binds a conserved C-terminal domain of TET1 to regulate TET1 activity and function in development. eLife 7. https://doi.org/10.7554/eLife.34870
Hu J, Wang Y, Zhao S, Chen J, Jin S, Jia P, Ding X (2018) Remote ischemic preconditioning ameliorates acute kidney injury due to contrast exposure in rats through augmented O-GlcNAcylation. Oxidative Med Cell Longev 2018:4895913–4895915. https://doi.org/10.1155/2018/4895913
Itkonen HM et al (2016) Inhibition of O-GlcNAc transferase activity reprograms prostate cancer cell metabolism. Oncotarget 7:12464–12476. https://doi.org/10.18632/oncotarget.7039
Janetzko J, Walker S (2014) The making of a sweet modification: structure and function of O-GlcNAc transferase. J Biol Chem 289:34424–34432. https://doi.org/10.1074/jbc.R114.604405
Jang I, Kim HB, Seo H, Kim JY, Choi H, Yoo JS, Kim JW, Cho JW (2015) O-GlcNAcylation of eIF2α regulates the phospho-eIF2α-mediated ER stress response. Biochim Biophys Acta 1853:1860–1869. https://doi.org/10.1016/j.bbamcr.2015.04.017
Jensen RV, Andreadou I, Hausenloy DJ, Bøtker HE (2019) The role of O-GlcNAcylation for protection against ischemia-reperfusion injury. Int J Mol Sci 20. https://doi.org/10.3390/ijms20020404
Jiang M, Xu B, Li X, Shang Y, Chu Y, Wang W, Chen D, Wu N, Hu S, Zhang S, Li M, Wu K, Yang X, Liang J, Nie Y, Fan D (2019) O-GlcNAcylation promotes colorectal cancer metastasis via the miR-101-O-GlcNAc/EZH2 regulatory feedback circuit. Oncogene 38:301–316. https://doi.org/10.1038/s41388-018-0435-5
Jiang M, Yu S, Yu Z, Sheng H, Li Y, Liu S, Warner DS, Paschen W, Yang W (2017) XBP1 (X-box-binding protein-1)-dependent O-GlcNAcylation is neuroprotective in ischemic stroke in young mice and its impairment in aged mice is rescued by thiamet-G. Stroke 48:1646–1654. https://doi.org/10.1161/strokeaha.117.016579
Jínek M, Rehwinkel J, Lazarus BD, Izaurralde E, Hanover JA, Conti E (2004) The superhelical TPR-repeat domain of O-linked GlcNAc transferase exhibits structural similarities to importin alpha. Nat Struct Mol Biol 11:1001–1007. https://doi.org/10.1038/nsmb833
Joiner CM, Li H, Jiang J, Walker S (2019) Structural characterization of the O-GlcNAc cycling enzymes: insights into substrate recognition and catalytic mechanisms. Curr Opin Struct Biol 56:97–106. https://doi.org/10.1016/j.sbi.2018.12.003
Jones DR, Keune WJ, Anderson KE, Stephens LR, Hawkins PT, Divecha N (2014) The hexosamine biosynthesis pathway and O-GlcNAcylation maintain insulin-stimulated PI3K-PKB phosphorylation and tumour cell growth after short-term glucose deprivation. FEBS J 281:3591–3608. https://doi.org/10.1111/febs.12879
Kang ES, Han D, Park J, Kwak TK, Oh MA, Lee SA, Choi S, Park ZY, Kim Y, Lee JW (2008) O-GlcNAc modulation at Akt1 Ser473 correlates with apoptosis of murine pancreatic beta cells. Exp Cell Res 314:2238–2248. https://doi.org/10.1016/j.yexcr.2008.04.014
Kátai E, Pál J, Poór VS, Purewal R, Miseta A, Nagy T (2016) Oxidative stress induces transient O-GlcNAc elevation and tau dephosphorylation in SH-SY5Y cells. J Cell Mol Med 20:2269–2277. https://doi.org/10.1111/jcmm.12910
Kazemi Z, Chang H, Haserodt S, McKen C, Zachara NE (2010) O-linked beta-N-acetylglucosamine (O-GlcNAc) regulates stress-induced heat shock protein expression in a GSK-3beta-dependent manner. J Biol Chem 285:39096–39107. https://doi.org/10.1074/jbc.M110.131102
Keembiyehetty C (2015) Disruption of O-GlcNAc cycling by deletion of O-GlcNAcase (Oga/Mgea5) changed gene expression pattern in mouse embryonic fibroblast (MEF) cells. Genomics Data 5:30–33. https://doi.org/10.1016/j.gdata.2015.04.026
Lazarus BD, Love DC, Hanover JA (2006) Recombinant O-GlcNAc transferase isoforms: identification of O-GlcNAcase, yes tyrosine kinase, and tau as isoform-specific substrates. Glycobiology 16:415–421. https://doi.org/10.1093/glycob/cwj078
Lazarus MB, Nam Y, Jiang J, Sliz P, Walker S (2011) Structure of human O-GlcNAc transferase and its complex with a peptide substrate. Nature 469:564–567. https://doi.org/10.1038/nature09638
Lee DE, Lee SJ, Kim SJ, Lee HS, Kwon OS (2019) Curcumin ameliorates nonalcoholic fatty liver disease through inhibition of O-GlcNAcylation. Nutrients 11. https://doi.org/10.3390/nu11112702
Lee HJ, Ryu JM, Jung YH, Lee KH, Kim DI, Han HJ (2016) Glycerol-3-phosphate acyltransferase-1 upregulation by O-GlcNAcylation of Sp1 protects against hypoxia-induced mouse embryonic stem cell apoptosis via mTOR activation. Cell Death Dis 7:e2158. https://doi.org/10.1038/cddis.2015.410
Lefebvre T, Dehennaut V, Guinez C, Olivier S, Drougat L, Mir AM, Mortuaire M, Vercoutter-Edouart AS, Michalski JC (2010) Dysregulation of the nutrient/stress sensor O-GlcNAcylation is involved in the etiology of cardiovascular disorders, type-2 diabetes and Alzheimer's disease. Biochim Biophys Acta 1800:67–79. https://doi.org/10.1016/j.bbagen.2009.08.008
Leney AC, El Atmioui D, Wu W, Ovaa H, Heck AJR (2017) Elucidating crosstalk mechanisms between phosphorylation and O-GlcNAcylation. Proc Natl Acad Sci U S A 114:E7255–e7261. https://doi.org/10.1073/pnas.1620529114
Li B, Li H, Lu L, Jiang J (2017) Structures of human O-GlcNAcase and its complexes reveal a new substrate recognition mode. Nat Struct Mol Biol 24:362–369. https://doi.org/10.1038/nsmb.3390
Li MD, Ruan HB, Hughes ME, Lee JS, Singh JP, Jones SP, Nitabach MN, Yang X (2013) O-GlcNAc signaling entrains the circadian clock by inhibiting BMAL1/CLOCK ubiquitination. Cell Metab 17:303–310. https://doi.org/10.1016/j.cmet.2012.12.015
Li MD, Vera NB, Yang Y, Zhang B, Ni W, Ziso-Qejvanaj E, Ding S, Zhang K, Yin R, Wang S, Zhou X, Fang EX, Xu T, Erion DM, Yang X (2018) Adipocyte OGT governs diet-induced hyperphagia and obesity. Nat Commun 9:5103. https://doi.org/10.1038/s41467-018-07461-x
Lima VV, Spitler K, Choi H, Webb RC, Tostes RC (2012) O-GlcNAcylation and oxidation of proteins: is signalling in the cardiovascular system becoming sweeter? Clin Sci (Lond) 123:473–486. https://doi.org/10.1042/cs20110638
Liu P, Yao R, Shi H, Liu Y, Lian S, Yang Y, Yang H, Li S (2019a) Effects of cold-inducible RNA-binding protein (CIRP) on liver glycolysis during acute cold exposure in C57BL/6 mice. Int J Mol Sci 20. https://doi.org/10.3390/ijms20061470
Liu Y, Wang X, Zhu T, Zhang N, Wang L, Huang T, Cao Y, Li W, Zhang J (2019b) Resistance to bortezomib in breast cancer cells that downregulate Bim through FOXA1 O-GlcNAcylation. J Cell Physiol 234:17527–17537. https://doi.org/10.1002/jcp.28376
Luanpitpong S, Chanthra N, Janan M, Poohadsuan J, Samart P, U-Pratya Y, Rojanasakul Y, Issaragrisil S (2018) Inhibition of O-GlcNAcase sensitizes apoptosis and reverses bortezomib resistance in mantle cell lymphoma through modification of truncated bid. Mol Cancer Ther 17:484–496. https://doi.org/10.1158/1535-7163.Mct-17-0390
Lucena MC, Carvalho-Cruz P, Donadio JL, Oliveira IA, de Queiroz RM, Marinho-Carvalho MM, Sola-Penna M, de Paula IF, Gondim KC, McComb ME, Costello CE, Whelan SA, Todeschini AR, Dias WB (2016) Epithelial mesenchymal transition induces aberrant glycosylation through hexosamine biosynthetic pathway activation. J Biol Chem 291:12917–12929. https://doi.org/10.1074/jbc.M116.729236
Ma J, Hart GW (2014) O-GlcNAc profiling: from proteins to proteomes. Clin Proteomics 11:8. https://doi.org/10.1186/1559-0275-11-8
Ma Z, Vocadlo DJ, Vosseller K (2013) Hyper-O-GlcNAcylation is anti-apoptotic and maintains constitutive NF-κB activity in pancreatic cancer cells. J Biol Chem 288:15121–15130. https://doi.org/10.1074/jbc.M113.470047
Ma Z, Vosseller K (2014) Cancer metabolism and elevated O-GlcNAc in oncogenic signaling. J Biol Chem 289:34457–34465. https://doi.org/10.1074/jbc.R114.577718
Marshall S, Bacote V, Traxinger RR (1991) Discovery of a metabolic pathway mediating glucose-induced desensitization of the glucose transport system. Role of hexosamine biosynthesis in the induction of insulin resistance. J Biol Chem 266:4706–4712
Martin JC, Fadda E, Ito K, Woods RJ (2014) Defining the structural origin of the substrate sequence independence of O-GlcNAcase using a combination of molecular docking and dynamics simulation. Glycobiology 24:85–96. https://doi.org/10.1093/glycob/cwt094
Martinez MR, Dias TB, Natov PS, Zachara NE (2017) Stress-induced O-GlcNAcylation: an adaptive process of injured cells. Biochem Soc Trans 45:237–249. https://doi.org/10.1042/bst20160153
Masui K, Cavenee WK, Mischel PS (2014) mTORC2 in the center of cancer metabolic reprogramming. Trends Endocrinol Metab 25:364–373. https://doi.org/10.1016/j.tem.2014.04.002
Mercier T, Bouvet M, Dubois-Deruy E, Dechaumes A, Beseme O, Richard V, Mulder P, Pinet F (2018) Interplay between phosphorylation and O-GlcNAcylation of sarcomeric proteins in ischemic heart failure. Front Endocrinol (Lausanne) 9:598. https://doi.org/10.3389/fendo.2018.00598
Ngoh GA, Hamid T, Prabhu SD, Jones SP (2009) O-GlcNAc signaling attenuates ER stress-induced cardiomyocyte death. Am J Physiol Heart Circ Physiol 297:H1711–H1719. https://doi.org/10.1152/ajpheart.00553.2009
Ngoh GA, Watson LJ, Facundo HT, Jones SP (2011) Augmented O-GlcNAc signaling attenuates oxidative stress and calcium overload in cardiomyocytes. Amino Acids 40:895–911. https://doi.org/10.1007/s00726-010-0728-7
Nie H, Ju H, Fan J, Shi X, Cheng Y, Cang X, Zheng Z, Duan X, Yi W (2020) O-GlcNAcylation of PGK1 coordinates glycolysis and TCA cycle to promote tumor growth. Nat Commun 11:36. https://doi.org/10.1038/s41467-019-13601-8
Nöt LG, Brocks CA, Vámhidy L, Marchase RB, Chatham JC (2010) Increased O-linked beta-N-acetylglucosamine levels on proteins improves survival, reduces inflammation and organ damage 24 hours after trauma-hemorrhage in rats. Crit Care Med 38:562–571. https://doi.org/10.1097/CCM.0b013e3181cb10b3
Ohashi N, Morino K, Ida S, Sekine O, Lemecha M, Kume S, Park SY, Choi CS, Ugi S, Maegawa H (2017) Pivotal role of O-GlcNAc modification in cold-induced thermogenesis by brown adipose tissue through mitochondrial biogenesis. Diabetes 66:2351–2362. https://doi.org/10.2337/db16-1427
Ohn T, Kedersha N, Hickman T, Tisdale S, Anderson P (2008) A functional RNAi screen links O-GlcNAc modification of ribosomal proteins to stress granule and processing body assembly. Nat Cell Biol 10:1224–1231. https://doi.org/10.1038/ncb1783
Pathak S, Alonso J, Schimpl M, Rafie K, Blair DE, Borodkin VS, Schüttelkopf AW, Albarbarawi O, van Aalten DMF (2015) The active site of O-GlcNAc transferase imposes constraints on substrate sequence. Nat Struct Mol Biol 22:744–750. https://doi.org/10.1038/nsmb.3063
Peterson SB, Hart GW (2016) New insights: a role for O-GlcNAcylation in diabetic complications. Crit Rev Biochem Mol Biol 51:150–161. https://doi.org/10.3109/10409238.2015.1135102
Qian K, Wang S, Fu M, Zhou J, Singh JP, Li MD, Yang Y, Zhang K, Wu J, Nie Y, Ruan HB, Yang X (2018) Transcriptional regulation of O-GlcNAc homeostasis is disrupted in pancreatic cancer. J Biol Chem 293:13989–14000. https://doi.org/10.1074/jbc.RA118.004709
Rao X, Duan X, Mao W, Li X, Li Z, Li Q, Zheng Z, Xu H, Chen M, Wang PG, Wang Y, Shen B, Yi W (2015) O-GlcNAcylation of G6PD promotes the pentose phosphate pathway and tumor growth. Nat Commun 6:8468. https://doi.org/10.1038/ncomms9468
Reeves RA, Lee A, Henry R, Zachara NE (2014) Characterization of the specificity of O-GlcNAc reactive antibodies under conditions of starvation and stress. Anal Biochem 457:8–18. https://doi.org/10.1016/j.ab.2014.04.008
Rexach JE, Clark PM, Hsieh-Wilson LC (2008) Chemical approaches to understanding O-GlcNAc glycosylation in the brain. Nat Chem Biol 4:97–106. https://doi.org/10.1038/nchembio.68
Rodriguez AC, Yu SH, Li B, Zegzouti H, Kohler JJ (2015) Enhanced transfer of a photocross-linking N-acetylglucosamine (GlcNAc) analog by an O-GlcNAc transferase mutant with converted substrate specificity. J Biol Chem 290:22638–22648. https://doi.org/10.1074/jbc.M115.667006
Ruan HB, Ma Y, Torres S, Zhang B, Feriod C, Heck RM, Qian K, Fu M, Li X, Nathanson MH, Bennett AM, Nie Y, Ehrlich BE, Yang X (2017) Calcium-dependent O-GlcNAc signaling drives liver autophagy in adaptation to starvation. Genes Dev 31:1655–1665. https://doi.org/10.1101/gad.305441.117
Ruan HB, Nie Y, Yang X (2013a) Regulation of protein degradation by O-GlcNAcylation: crosstalk with ubiquitination. Mol Cell Proteomics 12:3489–3497. https://doi.org/10.1074/mcp.R113.029751
Ruan HB, Singh JP, Li MD, Wu J, Yang X (2013b) Cracking the O-GlcNAc code in metabolism. Trends Endocrinol Metab 24:301–309. https://doi.org/10.1016/j.tem.2013.02.002
Rusai K, Herzog R, Kuster L, Kratochwill K, Aufricht C (2013) GSK-3β inhibition protects mesothelial cells during experimental peritoneal dialysis through upregulation of the heat shock response. Cell Stress Chaperones 18:569–579. https://doi.org/10.1007/s12192-013-0410-6
Sacoman JL, Dagda RY, Burnham-Marusich AR, Dagda RK, Berninsone PM (2017) Mitochondrial O-GlcNAc Transferase (mOGT) regulates mitochondrial structure, function, and survival in HeLa cells. J Biol Chem 292:4499–4518. https://doi.org/10.1074/jbc.M116.726752
Sharma NS, Gupta VK, Dauer P, Kesh K, Hadad R, Giri B, Chandra A, Dudeja V, Slawson C, Banerjee S, Vickers SM, Saluja A, Banerjee S (2019) O-GlcNAc modification of Sox2 regulates self-renewal in pancreatic cancer by promoting its stability. Theranostics 9:3410–3424. https://doi.org/10.7150/thno.32615
Shen DL, Gloster TM, Yuzwa SA, Vocadlo DJ (2012) Insights into O-linked N-acetylglucosamine ([0-9]O-GlcNAc) processing and dynamics through kinetic analysis of O-GlcNAc transferase and O-GlcNAcase activity on protein substrates. J Biol Chem 287:15395–15408. https://doi.org/10.1074/jbc.M111.310664
Shi H, Munk A, Nielsen TS, Daughtry MR, Larsson L, Li S, Høyer KF, Geisler HW, Sulek K, Kjøbsted R, Fisher T, Andersen MM, Shen Z, Hansen UK, England EM, Cheng Z, Højlund K, Wojtaszewski JFP, Yang X, Hulver MW, Helm RF, Treebak JT, Gerrard DE (2018) Skeletal muscle O-GlcNAc transferase is important for muscle energy homeostasis and whole-body insulin sensitivity. Mol Metab 11:160–177. https://doi.org/10.1016/j.molmet.2018.02.010
Shi H, Yao R, Lian S, Liu P, Liu Y, Yang YY, Yang H, Li S (2019) Regulating glycolysis, the TLR4 signal pathway and expression of RBM3 in mouse liver in response to acute cold exposure. Stress 22:366–376. https://doi.org/10.1080/10253890.2019.1568987
Slawson C, Hart GW (2011) O-GlcNAc signalling: implications for cancer cell biology. Nat Rev Cancer 11:678–684. https://doi.org/10.1038/nrc3114
Sodi VL, Bacigalupa ZA, Ferrer CM, Lee JV, Gocal WA, Mukhopadhyay D, Wellen KE, Ivan M, Reginato MJ (2018) Nutrient sensor O-GlcNAc transferase controls cancer lipid metabolism via SREBP-1 regulation. Oncogene 37:924–934. https://doi.org/10.1038/onc.2017.395
Suh HN, Lee YJ, Kim MO, Ryu JM, Han HJ (2014) Glucosamine-induced Sp1 O-GlcNAcylation ameliorates hypoxia-induced SGLT dysfunction in primary cultured renal proximal tubule cells. J Cell Physiol 229:1557–1568. https://doi.org/10.1002/jcp.24599
Torres CR, Hart GW (1984) Topography and polypeptide distribution of terminal N-acetylglucosamine residues on the surfaces of intact lymphocytes. Evidence for O-linked GlcNAc. J Biol Chem 259:3308–3317
Trapannone R, Mariappa D, Ferenbach AT, van Aalten DM (2016) Nucleocytoplasmic human O-GlcNAc transferase is sufficient for O-GlcNAcylation of mitochondrial proteins. Biochem J 473:1693–1702. https://doi.org/10.1042/bcj20160092
Trinca GM, Goodman ML, Papachristou EK, D’Santos CS, Chalise P, Madan R, Slawson C, Hagan CR (2018) O-GlcNAc-dependent regulation of progesterone receptor function in breast cancer. Horm Cancer 9:12–21. https://doi.org/10.1007/s12672-017-0310-9
Trinca GM, Hagan CR (2018) O-GlcNAcylation in women's cancers: breast, endometrial and ovarian. J Bioenerg Biomembr 50:199–204. https://doi.org/10.1007/s10863-017-9730-z
Very N, Vercoutter-Edouart AS, Lefebvre T, Hardivillé S, El Yazidi-Belkoura I (2018) Cross-dysregulation of O-GlcNAcylation and PI3K/AKT/mTOR Axis in human chronic diseases. Front Endocrinol (Lausanne) 9:602. https://doi.org/10.3389/fendo.2018.00602
Wang AC, Jensen EH, Rexach JE, Vinters HV, Hsieh-Wilson LC (2016a) Loss of O-GlcNAc glycosylation in forebrain excitatory neurons induces neurodegeneration. Proc Natl Acad Sci U S A 113:15120–15125. https://doi.org/10.1073/pnas.1606899113
Wang S, Yang F, Petyuk VA, Shukla AK, Monroe ME, Gritsenko MA, Rodland KD, Smith RD, Qian WJ, Gong CX, Liu T (2017) Quantitative proteomics identifies altered O-GlcNAcylation of structural, synaptic and memory-associated proteins in Alzheimer's disease. J Pathol 243:78–88. https://doi.org/10.1002/path.4929
Wang X, Feng Z, Wang X, Yang L, Han S, Cao K, Xu J, Zhao L, Zhang Y, Liu J (2016b) O-GlcNAcase deficiency suppresses skeletal myogenesis and insulin sensitivity in mice through the modulation of mitochondrial homeostasis. Diabetologia 59:1287–1296. https://doi.org/10.1007/s00125-016-3919-2
Wani WY, Boyer-Guittaut M, Dodson M, Chatham J, Darley-Usmar V, Zhang J (2015) Regulation of autophagy by protein post-translational modification. Lab Investig 95:14–25. https://doi.org/10.1038/labinvest.2014.131
Wani WY, Ouyang X, Benavides GA, Redmann M, Cofield SS, Shacka JJ, Chatham JC, Darley-Usmar V, Zhang J (2017) O-GlcNAc regulation of autophagy and α-synuclein homeostasis; implications for Parkinson's disease. Mol Brain 10:32. https://doi.org/10.1186/s13041-017-0311-1
Wellen KE, Lu C, Mancuso A, Lemons JMS, Ryczko M, Dennis JW, Rabinowitz JD, Coller HA, Thompson CB (2010) The hexosamine biosynthetic pathway couples growth factor-induced glutamine uptake to glucose metabolism. Genes Dev 24:2784–2799. https://doi.org/10.1101/gad.1985910
Whelan SA, Dias WB, Thiruneelakantapillai L, Lane MD, Hart GW (2010) Regulation of insulin receptor substrate 1 (IRS-1)/AKT kinase-mediated insulin signaling by O-linked beta-N-acetylglucosamine in 3T3-L1 adipocytes. J Biol Chem 285:5204–5211. https://doi.org/10.1074/jbc.M109.077818
Whisenhunt TR, Yang X, Bowe DB, Paterson AJ, Van Tine BA, Kudlow JE (2006) Disrupting the enzyme complex regulating O-GlcNAcylation blocks signaling and development. Glycobiology 16:551–563. https://doi.org/10.1093/glycob/cwj096
Xie S, Jin N, Gu J, Shi J, Sun J, Chu D, Zhang L, Dai CL, Gu JH, Gong CX, Iqbal K, Liu F (2016) O-GlcNAcylation of protein kinase a catalytic subunits enhances its activity: a mechanism linked to learning and memory deficits in Alzheimer's disease. Aging Cell 15:455–464. https://doi.org/10.1111/acel.12449
Xu W, Zhang X, Wu JL, Fu L, Liu K, Liu D, Chen GG, Lai PBS, Wong N, Yu J (2017) O-GlcNAc transferase promotes fatty liver-associated liver cancer through inducing palmitic acid and activating endoplasmic reticulum stress. J Hepatol 67:310–320. https://doi.org/10.1016/j.jhep.2017.03.017
Yang X, Ongusaha PP, Miles PD, Havstad JC, Zhang F, So WV, Kudlow JE, Michell RH, Olefsky JM, Field SJ, Evans RM (2008) Phosphoinositide signalling links O-GlcNAc transferase to insulin resistance. Nature 451:964–969. https://doi.org/10.1038/nature06668
Yang X, Qian K (2017) Protein O-GlcNAcylation: emerging mechanisms and functions. Nat Rev Mol Cell Biol 18:452–465. https://doi.org/10.1038/nrm.2017.22
Yao R (2018) Effects of O-GlcNAcylation modification on glucose metabolism in gastrocnemius and liver of acute cold-exposed mice and piglets. Heilongjiang Bayi Agricultural University, Daqing
Yao R, Yang Y, Lian S, Shi H, Liu P, Liu Y, Yang H, Li S (2018) Effects of acute cold stress on liver O-GlcNAcylation and Glycometabolism in mice. Int J Mol Sci 19. https://doi.org/10.3390/ijms19092815
Yi W, Clark PM, Mason DE, Keenan MC, Hill C, Goddard WA, Peters EC, Driggers EM, Hsieh-Wilson LC (2012) Phosphofructokinase 1 glycosylation regulates cell growth and metabolism. Science 337:975–980. https://doi.org/10.1126/science.1222278
Zafir A, Readnower R, Long BW, McCracken J, Aird A, Alvarez A, Cummins TD, Li Q, Hill BG, Bhatnagar A, Prabhu SD, Bolli R, Jones SP (2013) Protein O-GlcNAcylation is a novel cytoprotective signal in cardiac stem cells. Stem Cells 31:765–775. https://doi.org/10.1002/stem.1325
Zhang X, Qiao Y, Wu Q, Chen Y, Zou S, Liu X, Zhu G, Zhao Y, Chen Y, Yu Y, Pan Q, Wang J, Sun F (2017) The essential role of YAP O-GlcNAcylation in high-glucose-stimulated liver tumorigenesis. Nat Commun 8:15280. https://doi.org/10.1038/ncomms15280
Zhang X, Shu XE, Qian SB (2018) O-GlcNAc modification of eIF4GI acts as a translational switch in heat shock response. Nat Chem Biol 14:909–916. https://doi.org/10.1038/s41589-018-0120-6
Zhu G, Qian M, Lu L, Chen Y, Zhang X, Wu Q, Liu Y, Bian Z, Yang Y, Guo S, Wang J, Pan Q, Sun F (2019) O-GlcNAcylation of YY1 stimulates tumorigenesis in colorectal cancer cells by targeting SLC22A15 and AANAT. Carcinogenesis. https://doi.org/10.1093/carcin/bgz010
Zhu Y, Shan X, Yuzwa SA, Vocadlo DJ (2014) The emerging link between O-GlcNAc and Alzheimer disease. J Biol Chem 289:34472–34481. https://doi.org/10.1074/jbc.R114.601351
Funding
This work was supported by grants from the General Project of National Natural Science Foundation of China (31972637, 31772695) and Key Project of Heilongjiang Natural Science Foundation (ZD2019C004).
Author information
Authors and Affiliations
Contributions
Yang Liu had the idea for the manuscript and wrote the manuscript. Rui-Zhi Yao performed the literature search. Shuai Lian and Peng Liu performed complementary literature searches. Ya-Jie Hu, Hong-Zhao Shi, and Hong-Ming Lv composed figures. Yu-Ying Yang corrected the manuscript for publication. Bin Xu and Shi-Ze Li supervised the work, evaluated the data, and approved the final version. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, Y., Yao, RZ., Lian, S. et al. O-GlcNAcylation: the “stress and nutrition receptor” in cell stress response. Cell Stress and Chaperones 26, 297–309 (2021). https://doi.org/10.1007/s12192-020-01177-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-020-01177-y