Abstract
Purpose
Cerebral venous thrombosis (CVT) is a rare disease in children, characterized by partial or total occlusion of blood flow in the cerebral venous system. The aim of this study is to describe clinical presentation, neuroimaging features, therapeutic management, and outcome of children with CVT.
Methods
We retrospectively analyzed the data, including clinical manifestations, laboratory data, neurological findings, and treatment of children with radiologically confirmed CVT, admitted between January 2010 and March 2020 to our hospital. Cases of CVT complicating brain surgery were excluded.
Results
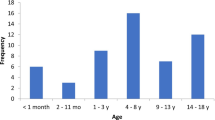
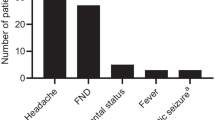
We enrolled 24 children with CVT. Infection was the main etiology (58.3%), followed by trauma in 16.7% of cases. In the remaining 25% of cases, the cause was identified only in one patient presenting a thrombophilic factor. The most frequent site of thrombosis was the superficial venous system (86.8%), with multiple localizations disclosed in 79% of patients. All children received anticoagulant therapy with low molecular weight heparin (LMWH). One patient died for systemic complications of an underlying disease. No patient developed hemorrhagic events during the therapy, lasting from 35 to 360 days (mean 86 days). In all but one surviving patients (22 out of 24), recanalization of the sinus was observed at AngioMRI performed during follow-up. No neurological complications of CVT were recorded (mean follow-up: 1.5 year).
Conclusions
CVT may present with subtle and unspecific clinical manifestations in children. High level of suspicion should be kept in trauma and sinusitis. Anticoagulation treatment is safe and effective and should be promptly started to improve outcome.


Similar content being viewed by others
Abbreviations
- ATIII:
-
Antithrombin III
- CTV:
-
Computed tomography venography
- CVT:
-
Cerebral venous thrombosis
- INR:
-
International normalized ratio
- IQR:
-
Interquartile range
- LMWH:
-
Low molecular weight heparin
- MRI:
-
Magnetic resonance imaging
- MRV:
-
Magnetic resonance venography
- PICU:
-
Pediatric intensive care unit
- TTR:
-
Target therapeutic range
- UFH:
-
Unfractionated heparin
- VKAs:
-
Vitamin K antagonists
- VTE:
-
Venous thrombosis
- Xa:
-
Activated factor X
References
Barnes C, Newall F, Furmedge J, Mackay M, Monagle P (2004) Cerebral sinus venous thrombosis in children. J Paediatr Child Health 40:53–55
De Veber G, Andrew M, Adams C, Bjornson B, Booth F, Buckley DJ et al (2001) Cerebral sinovenous thrombosis in children. N Engl J Med 345:417–423
Ichord R (2017) Cerebral sinovenous thrombosis. Front Pediatr 5:163
Witmer CM, Takemoto CM (2017) Pediatric hospital acquired venous thromboembolism. Front Pediatr 5:198
Gao L, Xu W, Yu X, Cao S, Xu H, Yan F et al (2018) Accuracy of magnetic resonance venography in diagnosing cerebral venous sinus thrombosis. Thromb Res 167:64–73
Widjaja E, Griffiths PD (2004) Intracranial MR venography in children: normal anatomy and variations. AJNR Am J Neuroradiol 25:1557–1562
Karsy M, Harmer JR, Guan J, Brock A, Ravindra VM, Chung LS et al (2018) Outcomes in adults with cerebral venous sinus thrombosis: a retrospective cohort study. J Clin Neurosci 53:34–40
Wang Y, Meng R, Duan J, Liu G, Chen J, Li S et al (2016) Nephrotic syndrome may be one of the important etiologies of cerebral venous sinus thrombosis. J Stroke Cerebrovasc Dis 25:2415–2422
Slasky SE, Rivaud Y, Suberlak M, Tairu O, Fox AD, Ohman-Strickland P (2017) Venous sinus thrombosis in blunt trauma: incidence and risk factors. J Comput Assist Tomogr 41:891–897
Monagle P, Chan AKC, Goldenberg NA, Ichord RN, Journeycake JM, Nowak-Göttl U et al (2012) Antithrombotic therapy in neonates and children: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 141:e737S–e801S
Członkowska A, Ciesielska A, Gromadzka G, Kurkowska-Jastrzebska I (2006) Gender differences in neurological disease: role of estrogens and cytokines. Endocrine 29:243–256
Golomb M, Fullerton H, Nowak-Gottl U, Deveber G, International Pediatric Stroke Study Group (2009) Male predominance in childhood ischemic stroke: findings from the international pediatric stroke study. Stroke 40:52–57
Ichord R, Benedict S, Chan A, Kirkham FJ, Nowak-Göttl U, International Paediatric Stroke Study Group (2015) Paediatric cerebral sinovenous thrombosis: findings of the International Paediatric Stroke Study. Arch Dis Child 100:174–179
Ichord R (2017) Cerebral sinovenous thrombosis. Front Pediatr 5:1–7
Jordan L, Rafay M, Smith S, Askalan R, Zamel KM, deVeber G et al (2010) Antithrombotic treatment in neonatal cerebral sinovenous thrombosis: results of the International Pediatric Stroke Study. J Pediatr 156:704–710
Dlamini N, Billinghurst L, Kirkham FJ (2010) Cerebral venous sinus (sinovenous) thrombosis in children. Neurosurg Clin N Am 21:511–527
Ferro JM, Canhao P, Stam J, Bousser MG, Barinagarrementeria F, Investigators ISCVT (2004) Prognosis of cerebral vein and dural sinus thrombosis: results of the International Study on Cerebral Vein and Dural Sinus Thrombosis (ISCVT). Stroke 35:664–670
Qu H, Yang M (2013) Early imaging characteristics of 62 cases of cerebral venous sinus thrombosis. Exp Ther Med 5:233–236
Einhaupl KM, Villringer A, Meister W, Garner C, Pellkofer M, Haberl RL et al (1991) Heparin treatment in sinus venous thrombosis. Lancet 338:597–600
De Bruijn SF, Stam J (1999) Randomized, placebo-controlled trial of anticoagulant treatment with low-molecular-weight heparin for cerebral sinus thrombosis. Stroke 30:484–488
Monagle P, Cuello C, Augustine C, Bonduel M, Brandão LR, Capman T et al (2018) American Society of Hematology 2018 Guidelines for Management of Venous Thromboembolism: treatment of pediatric venous thromboembolism. Blood Adv 2:3292–3316
Newall F, Branchford B, Male C (2018) Anticoagulant prophylaxis and therapy in children: current challenges and emerging issues. J Thromb Haemost 16:196–208
Greene LALC, Law C, Jung M, Walton S, Ignjatovic V, Monagle P et al (2014) Lack of anti-factor Xa assay standardization results in significant low molecular weight heparin (enoxaparin) dose variation in neonates and children. J Thromb Haemost 12:1554–1557
Monagle P, Newall F (2018) Management of thrombosis in children and neonates: practical use of anticoagulants in children. Hematology Am Soc Hematol Educ Program 2018:399–404
Andreozzi GM, Bignamini AA, Davì G, Palareti G, Matuška J, Holý M, SURVET Study Investigators et al (2015) Sulodexide for the prevention of recurrent venous thromboembolism: the Sulodexide in Secondary Prevention of Recurrent Deep Vein Thrombosis (SURVET) Study: a multicenter, randomized, double-blind, placebo-controlled trial. Circulation 132:1891–1897
Cohen O, Levy-Mendelovich S, Ageno W (2020) Rivaroxaban for the treatment of venous thromboembolism in pediatric patients. Expert Rev Cardiovasc Ther 24:1–9
Chhim RF, Shelton CM, Christensen ML (2013) Recent new drug approvals, part 2: drugs undergoing active clinical studies in children. J Pediatr Pharmacol Ther 18:14–38
Male C, Lensing AWA, Palumbo JS et al (2020) Rivaroxaban compared with standard anticoagulants for the treatment of acute venous thromboembolism in children: a randomised, controlled, phase 3 trial. Lancet Haematol 7:e18–e27
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Ethics approval
This study was approved by the Institutional Review Board and Ethic Committee of our institution.
Consent to participate
All subjects gave their consent to participate in the study.
Consent for publication
All subjects gave their consent to the publication of anonymized data.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lazzareschi, I., Curatola, A., Gatto, A. et al. Diagnosis and management of cerebral venous sinus thrombosis in children: a single-center retrospective analysis. Childs Nerv Syst 37, 153–160 (2021). https://doi.org/10.1007/s00381-020-04958-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-020-04958-z