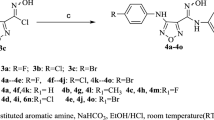
Abstract
The objective of the current study is to evaluate the potency of halogen-furan-2(5H)-one-type derivatives against human cancer cell lines. Four known bromofuran-2(5H)-one-type derivatives, as well as five new and two known bromo-4-(phenylamino)furan-2(5H)-one-type compounds and six novel and two known halogen-4-alkyl-5-phenyl-3-(phenylamino)furan-2(5H)-one-type derivatives, were synthesized and evaluated for their anticancer activity against prostate (PC-3) and colon (HCT-116) human cancer cell lines. The results showed that only the bromofuran-2(5H)-ones were cytotoxic in both cell lines. Three of these displayed particularly useful antiproliferative activities, in both cancer cells evaluated. (E)-5-(Bromomethylene)furan-2-(5H)-one was the most active against PC-3 (IC50 0.93 ± 0.02 μM) while 3,4-dibromofuran-2(5H)-one was the most active against HCT-116 (IC50 0.4 ± 0.04 μM). Furthermore, flow cytometry studies revealed that the bromofuran-2(5H)-ones induced cell death by apoptosis. Also, it was found that the cytotoxic furanones induced lipid peroxidation, determined by TBARS assay. Thus, cytotoxicity of the active compounds could be associated with ROS production. Additionally, it must be taken into account that all cytotoxic compounds contain an electrophilic carbon atom in position 4, which can explain, through a non-specific reactivity with nucleophiles, the cytotoxic activity of these compounds.
Graphic abstract



Similar content being viewed by others
References
Globocan 2019. https://gco.iarc.fr/today/data/factsheets/populations/900-world-fact-sheets.pdf. Accessed 9 May 2020
Lopez-Lazaro M, Pastor N, Azrak SS, Ayusco CA, Austin F, Cortes F (2005) J Nat Prod 68:1642
Lopez-Lazaro M (2007) Expert Opin Ther Targets 11:1043
Newman RA, Yang P, Pawlus AD, Keith IB (2008) Mol Interv 8:36
Peng CK, Zeng T, Xu XJ, Chang YQ, Hou W, Lu K, Lin H, Sun PH, Lin J, Chen WM (2017) Eur J Med Chem 127:187
Byczek-Wyrosteka A, Kitela R, Rumaka K, Skoniecznab M, Kasprzyckaa A, Walczaka K (2018) Eur J Med Chem 150:687
Manny AJ, Kjelleberg S, Kumar N, Nys R, Read RW, Steinberg P (1997) Tetrahedron 53:15813
MacDonald SF (1974) Can J Chem 52:3257
Benneche T, Lönn J, Scheie AA (2006) Synth Commun 36:1401
Taylor GA (1963) Org Synth Coll 4:688
Bellina F, Anselmi C, Viel S, Mannina L, Rossi R (2001) Tetrahedron 57:9997
Cunha S, Oliveira C, Sabino JR (2011) J Braz Chem Soc 22:598
Narayana MS, Madhav B, Vijay KA, Rama RK, Nageswar YVD (2009) Tetrahedron 65:5251
Hall IH, Lee KH, Mar EC, Starnes CO, Waddell TG (1997) J Med Chem 20:333
Zeni O, Di Pietro R, d’Ambrosio G, Massa R, Capri M, Naarale J, Juutilainen J, Scarfì MR (2007) Radiat Res 167:306
Hernández-Vázquez E, Miranda LD (2016) Org Biomol Chem 14:4875
Liviac D, Creus A, Marcos R (2009) Environ Mol Mutagen 50:413
Wu H, Song Z, Hentzer M, Andersen JB, Moli S, Givskov M, Høiby N (2004) J Antimicrob Chemother 53:1054
Kibanova A, Camacho AN, Silva JC (2009) Environ Sci Technol 43:7550
Acknowledgements
Víctor A. Castro-Torres is a doctoral student from the Programa de Doctorado en Ciencias Biomédicas, Universidad Nacional Autónoma de México (UNAM) and has received CONACyT fellowship 267787. This study was supported by Grants from PAPIIT IN211819 and CONACyT 402838 and 00042. We acknowledge the technical support of MS Antonio Nieto-Camacho and MS María Teresa O. Ramírez-Apan from Instituto de Química-UNAM.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Castro-Torres, V.A., Jacobo-Herrera, N.J., Díaz-Sánchez, L. et al. Synthesis and cytotoxic evaluation of halogenated furanones. Monatsh Chem 151, 1841–1849 (2020). https://doi.org/10.1007/s00706-020-02708-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-020-02708-0