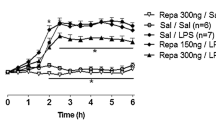
Abstract
Sickness syndrome is an adaptive response that can be distinguished by specific signs and symptoms, such as fever and generalized hyperalgesia. Endothelin-1 (ET-1) is produced by inflammatory stimuli, including lipopolysaccharide, and involved in the pathogenesis of inflammation and pain by acting through ETA and ETB receptors. ET-1 also induces fever by acting on the central nervous system. The present study investigated the role of ET-1 in sickness syndrome responses, including hyperalgesia, anhedonia, and hypolocomotion. Intracerebroventricular ET-1 administration induced mechanical and thermal hyperalgesia in rats, which was ameliorated by the ETA receptor antagonist BQ123 and exacerbated by the ETB receptor antagonist BQ788. A cyclooxygenase blocker did not alter hyperalgesia that was induced by ET-1. Lipopolysaccharide administration induced hyperalgesia, and both BQ123 and BQ788 abolished this mechanical hyperalgesia, but the thermal response was only partially blocked. The blockade of ETA receptors in the hypothalamus also abolished lipopolysaccharide-induced mechanical hyperalgesia, and the ETB receptor antagonist did not influence this response. Lipopolysaccharide also induced anhedonia, reflected by lower sucrose preference, and reduced locomotor activity. Both antagonists restored locomotor activity, but only BQ788 reversed the reduction of sucrose preference. These results indicate that ET-1 and both ETA and ETB receptors are involved in various responses that are related to sickness syndrome, including hyperalgesia, anhedonia, and hypolocomotion, that is induced by LPS. Hypothalamic ETA but not ETB receptors are involved in mechanical hyperalgesia that is observed during lipopolysaccharide-induced sickness syndrome.






Similar content being viewed by others
Data availability
Data are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
References
Abe M, Oka T, Hori T, Takahashi S (2001) Prostanoids in the preoptic hypothalamus mediate systemic lipopolysaccharide-induced hyperalgesia in rats. Brain Res 916(1):41–49. https://doi.org/10.1016/S0006-8993(01)02861-X
Alexander SPH, Christopoulos A, Davenport AP, Kelly E, Mathie A, Peters JA, Veale EL, Armstrong JF, Faccenda E, Harding SD et al (2019) The concise guide to pharmacology 2019/20: G protein-coupled receptors. Br J Pharmacol 176(Suppl 1):21–141. https://doi.org/10.1111/bph.14748
Blais V, Fugère M, Denault JB, Klarskov K, Day R, Leduc R (2002) Processing of proendothelin-1 by members of the subtilisin-like pro-protein convertase family. FEBS Lett 524(1–3):43–48. https://doi.org/10.1016/s0014-5793(02)02998-8
Brito HO, Radulski D, Wilhelms DB, Stojakovic A, Brito L, Gil da Costa RM, Trindade E, Engblom D, Franco C, Zampronio AR (2020) Immune-mediated febrile response in female rats: role of central hypothalamic mediators. Sci Rep 10(1):4073. https://doi.org/10.1038/s41598-020-61210-z
Cahill CM, Dray A, Coderre TJ (1998) Priming enhances endotoxin-induced thermal hyperalgesia and mechanical allodynia in rats. Brain Res 808(1):13–22. https://doi.org/10.1016/s0006-8993(98)00786-0
Chichorro J, Zampronio A, Rae G (2006) Endothelin ETB receptor antagonist reduces mechanical allodynia in rats with trigeminal neuropathic pain. Exp Biol Med (Maywood) 231:1136–1140
D’Amico M, Berrino L, Maione S, Filippelli A, de Novellis V, Rossi F (1996) Endothelin-1 in periaqueductal gray area of mice induces analgesia via glutamatergic receptors. Pain 65(2–3):205–209. https://doi.org/10.1016/0304-3959(95)00178-6
D’Amico M, Di Filippo C, Rossi F (1997) Selective and non-selective ET antagonists reveal an ET(A)/ET(B) receptor mediated ET-1-induced antinociceptive effect in PAG area of mice. Life Sci. 61(25):PL397–PL401. https://doi.org/10.1016/s0024-3205(97)00988-0
da Cunha JM, Rae GA, Ferreira SH, Cunha FQ (2004) Endothelins induce ETB receptor-mediated mechanical hypernociception in rat hindpaw: roles of cAMP and protein kinase C. Eur J Pharmacol 501(1–3):87–94. https://doi.org/10.1016/j.ejphar.2004.08.004
Dantzer R (2004) Cytokine-induced sickness behaviour: a neuroimmune response to activation of innate immunity. Eur J Pharmacol 500(1–3):399–411. https://doi.org/10.1016/j.ejphar.2004.07.040
de Oliveira BM, Telles TMBB, Lomba LA, Correia D, Zampronio AR (2017) Effects of binge-like ethanol exposure during adolescence on the hyperalgesia observed during sickness syndrome in rats. Pharmacol Biochem Behav 160:63–69. https://doi.org/10.1016/J.PBB.2017.08.004
De-Melo JD, Tonussi CR, D’Orleans-Juste P, Rae GA (1998) Articular nociception induced by endothelin-1, carrageenan and LPS in naive and previously inflamed knee-joints in the rat: inhibition by endothelin receptor antagonists. Pain 77(3):261–269. https://doi.org/10.1016/s0304-3959(98)00098-0
D'Orléans-Juste P, Plante M, Honoré JC, Carrier E, Labonté J (2003) Synthesis and degradation of endothelin-1. Can J Physiol Pharmacol 81(6):503–510. https://doi.org/10.1139/y03-032
D'Orléans-Juste P, Akide Ndunge OB, Desbiens L, Tanowitz HB, Desruisseaux MS (2019) Endothelins in inflammatory neurological diseases. Pharmacol Therap 194:145–160. https://doi.org/10.1016/j.pharmthera.2018.10.001
Fabricio AS, Silva CA, Rae GA, D’Orleans-Juste P, Souza GE (1998) Essential role for endothelin ET(B) receptors in fever induced by LPS (E. coli) in rats. Br J Pharmacol 125(3):542–548. https://doi.org/10.1038/sj.bjp.0702075
Fabricio AS, Rae GA, D'Orléans-Juste P, Souza GE (2005a) Endothelin-1 as a central mediator of LPS-induced fever in rats. Brain Res 1066(1–2):92–100. https://doi.org/10.1016/j.brainres.2005.10.037
Fabricio AS, Veiga FH, Cristofoletti R, Navarra P, Souza GE (2005b) The effects of selective and nonselective cyclooxygenase inhibitors on endothelin-1-induced fever in rats. Am J Physiol Regul Integr Comp Physiol 288(3):R671–R677. https://doi.org/10.1152/ajpregu.00532.2004
Gokin AP, Fareed MU, Pan HL, Hans G, Strichartz GR, Davar G (2001) Local injection of endothelin-1 produces pain-like behavior and excitation of nociceptors in rats. J Neurosci 21(14):5358–5366. https://doi.org/10.1523/JNEUROSCI.21-14-05358.2001
Hans G, Schmidt BL, Strichartz G (2009) Nociceptive sensitization by endothelin-1. Brain Res Rev 60(1):36–42. https://doi.org/10.1016/j.brainresrev.2008.12.008
Hasue F, Kuwaki T, Yamada H, Fukuda Y, Shimoyama M (2004) Inhibitory actions of endothelin-1 on pain processing. J Cardiovasc Pharmacol 44(Suppl 1):S318–S320. https://doi.org/10.1097/01.fjc.0000166271.40044.0c
Heinricher MM, Neubert MJ, Martenson ME, Goncalves L (2004) Prostaglandin E2 in the medial preoptic area produces hyperalgesia and activates pain-modulating circuitry in the rostral ventromedial medulla. Neuroscience 128(2):389–398. https://doi.org/10.1016/j.neuroscience.2004.06.050
Hostenbach S, D’haeseleer M, Kooijman R, De Keyser J (2016) The pathophysiological role of astrocytic endothelin-1. Prog Neurobiol. 44:88–102. https://doi.org/10.1016/j.pneurobio.2016.04.009
Houck CS, Khodorova A, Reale AM, Strichartz GR, Davar G (2004) Sensory fibers resistant to the actions of tetrodotoxin mediate nocifensive responses to local administration of endothelin-1 in rats. Pain 110(3):719–726. https://doi.org/10.1016/j.pain.2004.05.012
Hung VKL, Chen SMY, Tai LW, Chen AYS, Chung SK, Cheung CW (2012) Over-expression of endothelin-1 in astrocytes, but not endothelial cells, ameliorates inflammatory pain response after formalin injection. Life Sci 91(13–14):618–622. https://doi.org/10.1016/j.lfs.2012.06.038
Kedzierski RM, Yanagisawa M (2001) Endothelin system: the double-edged sword in health and disease. Annu Rev Pharmacol Toxicol 41:851–876. https://doi.org/10.1146/annurev.pharmtox.41.1.851
Khodorova A, Fareed MU, Gokin A, Strichartz GR, Davar G (2002) Local injection of a selective endothelin-B receptor agonist inhibits endothelin-1-induced pain-like behavior and excitation of nociceptors in a naloxone-sensitive manner. J Neurosci 22(17):7788–7796. https://doi.org/10.1523/JNEUROSCI.22-17-07788.2002
Khodorova A, Navarro B, Jouaville LS, Murphy J-E, Rice FL, Mazurkiewicz JE, Long-Woodward D, Stoffel M, Strichartz GR, Yukhananov R et al (2003) Endothelin-B receptor activation triggers an endogenous analgesic cascade at sites of peripheral injury. Nat Med 9(8):1055–1061. https://doi.org/10.1038/nm885
Khodorova A, Montmayeur J-P, Strichartz G (2009) Endothelin receptors and pain. J Pain 10(1):4–28. https://doi.org/10.1016/j.jpain.2008.09.009
Leite-Avalca MCG, Lomba LA, Bastos-Pereira AL, Brito HO, Fraga D, Zampronio AR (2016) Involvement of central endothelin et a and cannabinoid cb 1 receptors and arginine vasopressin release in sepsis induced by cecal ligation and puncture in rats. Shock. https://doi.org/10.1097/SHK.0000000000000598
Leite-Avalca MCG, Staats FT, Verona D, de Souza P, Almeida MC, Silva-Santos JE, Zampronio AR (2019) Cannabinoid CB1 receptor antagonist rimonabant decreases levels of markers of organ dysfunction and alters vascular reactivity in aortic vessels in late sepsis in rats. Inflammation 42(2):618–627. https://doi.org/10.1007/s10753-018-0919-z
Motta EM, Chichorro JG, D’Orléans-Juste P, Rae GA (2009) Roles of endothelin ETA and ETB receptors in nociception and chemical, thermal and mechanical hyperalgesia induced by endothelin-1 in the rat hindpaw. Peptides 30(5):918–925. https://doi.org/10.1016/j.peptides.2009.01.011
Nikolov R, Maslarova J, Semkova I, Moyanova S (1992) Intracerebroventricular endothelin-1 (ET-1) produces Ca(2+)-mediated antinociception in mice. Methods Find Exp Clin Pharmacol 14(3):229–233
Paxinos G, Watson C (1998) The rat brain in extereotaxic coordinates. Academic Press, San Diego
Piovezan AP, D’Orleans-Juste P, Souza GE, Rae GA (2000) Endothelin-1-induced ET(A) receptor-mediated nociception, hyperalgesia and oedema in the mouse hind-paw: modulation by simultaneous ET(B) receptor activation. Br J Pharmacol 129(5):961–968. https://doi.org/10.1038/sj.bjp.0703154
Pitychoutis PM, Nakamura K, Tsonis PA, Papadopoulou-Daifoti Z (2009) Neurochemical and behavioral alterations in an inflammatory model of depression: sex differences exposed. Neuroscience 159(4):1216–1232. https://doi.org/10.1016/j.neuroscience.2009.01.072
Raffa RB, Schupsky JJ, Jacoby HI (1996) Endothelin-induced nociception in mice: mediation by ETA and ETB receptors. J Pharmacol Exp Ther 276(2):647–651
Sakurai T, Yanagisawa M, Takuwa Y, Miyazaki H, Kimura S, Goto K, Masaki T (1990) Cloning of a cDNA encoding a non-isopeptide-selective subtype of the endothelin receptor. Nature 348(6303):732–735. https://doi.org/10.1038/348732a0
Saper CB, Romanovsky AA, Scammell TE (2012) Neural circuitry engaged by prostaglandins during the sickness syndrome. Nat Neurosci 15(8):1088–1095. https://doi.org/10.1038/nn.3159
Sasaki Y, Takimoto M, Oda K, Fruh T, Takai M, Okada T, Hori S (1997) Endothelin evokes efflux of glutamate in cultures of rat astrocytes. J Neurochem 68(5):2194–2200. https://doi.org/10.1046/j.1471-4159.1997.68052194.x
Schinelli S (2006) Pharmacology and physiopathology of the brain endothelin system: an overview. Curr Med Chem 13(6):627–638. https://doi.org/10.2174/092986706776055652
Soncini R, de Souza DF, Neves AP, Braga DS, Andrade CAF, Giusti-Paiva A (2012) Dipyrone attenuates acute sickness response to lipopolysaccharide in mice. Neurosci Lett 516(1):114–118. https://doi.org/10.1016/j.neulet.2012.03.070
Souza LC, Martynhak BJ, Bassani TB et al (2018) Agomelatine’s effect on circadian locomotor rhythm alteration and depressive-like behavior in 6-OHDA lesioned rats. Physiol Behav 188:298–310. https://doi.org/10.1016/j.physbeh.2018.02.033
Telles TM, de Oliveira BM, Lomba LA, Leite-Avalca MG, Correia D, Zampronio AR (2017) Effects of binge-like ethanrol exposure during adolescence on the febrile response in rats. Alcoholism Clin Exp Res 41(3):507–515. https://doi.org/10.1111/acer.13333
Werner MFP, Trevisani M, Campi B, Andre E, Geppetti P, Rae GA (2010) Contribution of peripheral endothelin ETA and ETB receptors in neuropathic pain induced by spinal nerve ligation in rats. Eur J Pain 14(9):911–917. https://doi.org/10.1016/j.ejpain.2010.03.001
Wilhelms DB, Mirrasekhian E, Zajdel J, Kumar Singh A, Engblom D (2016) Cyclooxygenase isoform exchange blocks brain-mediated inflammatory symptoms. PLoS ONE 11(11):e0166153. https://doi.org/10.1371/journal.pone.0166153
Zampronio AR, Kuzmiski JB, Florence CM, Mulligan SJ, Pittman QJ (2010) Opposing actions of endothelin-1 on glutamatergic transmission onto vasopressin and oxytocin neurons in the supraoptic nucleus. J Neurosci 30(50):16855–16863. https://doi.org/10.1523/JNEUROSCI.5079-10.2010
Ziv I, Fleminger G, Djaldetti R, Achiron A, Melamed E, Sokolovsky M (1992) Increased plasma endothelin-1 in acute ischemic stroke. Stroke 23(7):1014–1016. https://doi.org/10.1161/01.str.23.7.1014
Acknowledgements
LAL received a scholarship from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Brazil. This study was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Grant # 457938/2014-5), Brazil.
Author information
Authors and Affiliations
Contributions
Conceptualization, methodology, formal analysis, and supervision: ARZ and DC. Data collection and data analysis: LAL, MCGL-A, JVC, and LCMC. Writing, review, and edition: LAL, JVC, and ARZ. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
This research study was approved by the Ethics Committee for Animal Use (no. 1024) of Universidade Federal do Paraná and were in accordance with international legislation for animal use in research.
Consent to participate
Not applicable.
Additional information
Communicated by Bill J Yates.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
221_2020_5929_MOESM1_ESM.tiff
Supplementary Figure 1: Effects of BQ123 and BQ788 on mechanical and thermal threshold in rats. Rats were treated with BQ123 (3 pmol/2 µl, i.c.v.) or BQ788 (3 pmol/2 µl, i.c.v.) or vehicle (Veh; 2 µl of sterile saline, i.c.v.) and then vehicle (Veh, 2 µl of sterile saline, i.c.v.). The mechanical threshold (A) and PWL (B) were evaluated at the indicated time points. The results are expressed as the mean ± SEM mechanical threshold (in grams) or PWL (in seconds). The number of animals/group is shown in the figure legends (TIFF 810 kb)
Rights and permissions
About this article
Cite this article
Lomba, L.A., Cruz, J.V., Coelho, L.C.M. et al. Role of central endothelin-1 in hyperalgesia, anhedonia, and hypolocomotion induced by endotoxin in male rats. Exp Brain Res 239, 267–277 (2021). https://doi.org/10.1007/s00221-020-05929-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-020-05929-1