Abstract
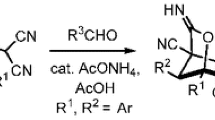
The sequential reaction of furfural with cyclic secondary amines and further with benzaldehyde and cyanoacetates affords new 2,4-dicyano-8-(R2N)-6-oxo-3-phenylbicyclo[3.2.1]-octane-2,4-dicarboxylates as rac-(1R,2R,3R,4S,5S,8R)-diastereomers. The structures of the reaction products were determined by X-ray diffraction.
Similar content being viewed by others
References
A. Lefranc, L. Gremaud, A. Alexakis, Org. Lett., 2014, 16, 5242.
M. Rueping, A. Kuenkel, F. Tato, J. W. Bats, Angew. Chem. (Int. Ed.), 2009, 48, 3699.
B. Tan, Y. Lu, X. Zeng, P. J. Chua, G. Zhong, Org. Lett., 2010, 12, 2682.
J. Zhao, S. Yang, X. Xie, X. Li, Y. Liu, J. Org. Chem., 2018, 83, 1287.
C. He, Z. Bai, J. Hu, B. Wang, H. Xie, L. Yu, H. Ding, Chem. Commun., 2017, 53, 8435.
M. H. Filippini, J. Rodriguez, Chem. Rev., 1999, 99, 27.
M. Presset, Y. Coquerel, J. Rodriguez, Chem. Rev., 2013, 113, 525.
M. Presset, Y. Coquerel, J. Rodriguez, Chem. Cat. Chem., 2012, 4, 172.
P. C. Meltzer, P. Blundell, Y. F. Yong, Z. Chen, C. George, M. D. Gonzalez, B. K. Madras, J. Med. Chem., 2000, 43, 2982.
P. C. Meltzer, P. Blundell, Z. Chen, Y. F. Yong, B. K. Madras, Bioorg. Med. Chem. Lett., 1999, 9, 857.
C. Peter, P. Geoffroy, M. Miesch, Org. Biomol. Chem., 2018, 16, 1381.
V. V. Dotsenko, A. I. Ismiev, A. N. Khrustaleva, K. A. Frolov, S. G. Krivokolysko, E. A. Chigorina, A. P. Snizhko, V. M. Gromenko, I. S. Bushmarinov, R. K. Askerov, T. M. Pekhtereva, S. Yu. Suykov, E. S. Papayanina, A. V. Mazepa, A. M. Magerramov, Chem. Heterocycl. Compd., 2016, 52, 473.
V. V. Dotsenko, E. A. Chigorina, S. G. Krivokolysko, Chem. Heterocycl. Compd., 2017, 53, 626.
E. A. Chigorina, K. A. Frolov, V. V. Dotsenko, A. S. Goloveshkin, I. S. Bushmarinov, S. G. Krivokolysko, Russ. Chem. Bull., 2016, 65, 2260.
V. V. Dotsenko, S. G. Krivokolysko, B. S. Krivokolysko, K. A. Frolov, Russ. J. Gen. Chem., 2018, 88, 682.
A. I. Ismiev, V. V. Dotsenko, N. A. Aksenov, G. Z. Mamedova, A. M. Magerramov, Russ. J. Gen. Chem., 2018, 88, 1533.
V. V. Dotsenko, K. A. Frolov, E. A. Chigorina, A. N. Khrustaleva, E. Yu. Bibik, S. G. Krivokolysko, Russ. Chem. Bull., 2019, 68.
C. Piutti, F. Quartieri, Molecules, 2013, 18, 12290.
C. Verrier, S. Moebs-Sanchez, Y. Queneau, F. Popowycz, Org. Biomol. Chem., 2018, 16, 676.
G. K. Veits, D. R. Wenz, J. Read de Alaniz, Angew. Chem. (Int. Ed.), 2010, 49, 9484.
D. Lebœuf, E. Schulz, V. Gandon, Org. Lett., 2014, 16, 6464.
B. V. Subba Reddy, Y. Vikram Reddy, P. Subba Lakshumma, G. Narasimhulu, J. S. Yadav, B. Sridhar, P. Purushotham Reddy, A. C. Kunwar, RSC Advances, 2012, 2, 10661.
D. Yu, V. T. Thai, L. I. Palmer, G. K. Veits, J. E. Cook, J. Read de Alaniz, J. E. Hein, J. Org. Chem., 2013, 78, 12784.
L. I. Palmer, J. Read de Alaniz, Synlett, 2014, 25, 8.
H. Li, R. Tong, J. Sun, Angew. Chem. (Int. Ed.), 2016, 55, 15125.
Wu-Bang Tang, Kou-Sen Cao, Shan-Shui Meng, Wen-Hua Zheng, Synthesis, 2017, 49, 3670.
M. Nardi, P. Costanzo, A. De Nino, M. L. Di Gioia, F. Olivito, G. Sindona, A. Procopio, Green Chem., 2017, 19, 5403.
M. A. Tius, Eur. J. Org. Chem., 2005, 2193.
M. J. Di Grandi, Org. Biomol. Chem., 2014, 12, 5331.
D. R. Wenz, J. Read de Alaniz, Eur. J. Org. Chem., 2015, 23.
K. Griffiths, C. W. D. Gallop, A. Abdul-Sada, A. Vargas, O. Navarro, G. E. Kostakis, Chem. Eur. J., 2015, 21, 6358.
D. Ramesh, T. Srikanth Reddy, M. Narasimhulu, S. Rajaram, N. Suryakiran, K. Chinni Mahesh, Y. Venkateswarlu, Chem. Lett., 2009, 38, 586.
A. Procopio, P. Costanzo, M. Curini, M. Nardi, M. Oliverio, G. Sindona, ACS Sust. Chem. Eng., 2013, 1, 541.
M. S. Estevão, C. A. M. Afonso, Tetrahedron Lett., 2017, 58, 302.
R. F. Gomes, J. A. Coelho, C. A. Afonso, Chem. Eur. J., 2018, 24, 9170.
G. Piancatelli, M. D’Auria, F. D’Onofrio, Synthesis, 1994, 867.
G. Sheldrick, Acta Cryst. Sect. A, 2008, 64, 112.
O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard, H. Puschmann, J. Appl. Cryst., 2009, 42, 339.
G. M. Sheldrick, Acta Cryst. Sect. C, 2015, 71, 3.
Author information
Authors and Affiliations
Corresponding authors
Additional information
This work was financially supported by the Ministry of Science and Higher Education of the Russian Federation (theme 0795-2020-0031; project executors V. V. Dotsenko, N. A. Aksenov, and I. V. Aksenova).
Russian Chemical Bulletin, International Edition, Vol. 69, No. 10, pp. 1938–1943, October, 2020
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 10, pp. 1938–1943, October, 2020.
Rights and permissions
About this article
Cite this article
Ismiyev, A.I., Dotsenko, V.V., Aksenov, N.A. et al. Synthesis and structure of new 2,4-dicyano-6-oxo-3-phenylbicyclo[3.2.1]octane-2,4-dicarboxylates. Russ Chem Bull 69, 1938–1943 (2020). https://doi.org/10.1007/s11172-020-2982-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-020-2982-5