Abstract
Key message
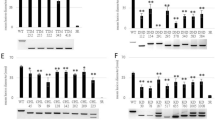
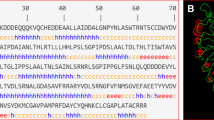
Metabolic pathway gene editing in tetraploid potato enhanced resistance to late blight. Multiallelic mutation correction of a caffeoyl-CoA O-methyltransferase gene increased accumulation of resistance metabolites in Russet Burbank potato.
Abstract
Late blight of potato is a devastating disease worldwide and requires weekly applications of fungicides to manage. Genetic improvement is the best option, but the self-incompatibility and inter-specific incompatibility makes potato breeding very challenging. Immune receptor gene stacking has increased resistance, but its durability is limited. Quantitative resistance is durable, and it mainly involves secondary cell wall thickening due to several metabolites and their conjugates. Deleterious mutations in biosynthetic genes can hinder resistance metabolite biosynthesis. Here a probable resistance role of the StCCoAOMT gene was first confirmed by an in-planta transient overexpression of the functional StCCoAOMT allele in late blight susceptible Russet Burbank (RB) genotype. Following this, a precise single nucleotide polymorphism (SNP) mutation correction of the StCCoAOMT gene in RB potato was carried out using CRISPR-Cas9 mediated homology directed repair (HDR). The StCCoAOMT gene editing increased the transcript abundance of downstream biosynthetic resistance genes. Following pathogen inoculation, several phenylpropanoid pathway genes were highly expressed in the edited RB plants, as compared to the non-edited. The disease severity (fold change = 3.76) and pathogen biomass in inoculated stems of gene-edited RB significantly reduced (FC = 21.14), relative to non-edited control. The metabolic profiling revealed a significant increase in the accumulation of resistance-related metabolites in StCCoAOMT edited RB plants. Most of these metabolites are involved in suberization and lignification. The StCCoAOMT gene, if mutated, can be edited in other potato cultivars to enhance resistance to late blight, provided it is associated with other functional genes in the metabolic pathway network.








Similar content being viewed by others
References
Almagro L, Gómez Ros LV, Belchi-Navarro S et al (2008) Class III peroxidases in plant defence reactions. J Exp Bot 60:377–390. https://doi.org/10.1093/jxb/ern277
Andersen EJ, Ali S, Byamukama E et al (2018) Disease resistance mechanisms in plants. Genes 9:339. https://doi.org/10.3390/genes9070339
Andersson M, Turesson H, Olsson N et al (2018) Genome editing in potato via CRISPR-Cas9 ribonucleoprotein delivery. Physiol Plant 164:378–384. https://doi.org/10.1111/ppl.12731
Arora R, Sharma S, Singh B (2014) Late blight disease of potato and its management. Potato J 4:16–40
Asai S, Ohta K, Yoshioka H (2008) MAPK signaling regulates nitric oxide and NADPH oxidase-dependent oxidative bursts in Nicotiana benthamiana. Plant Cell 20:1390–1406. https://doi.org/10.1105/tpc.107.055855
Baltes NJ, Gil-Humanes J, Cermak T et al (2014) DNA replicons for plant genome engineering. Plant Cell 26:151–163. https://doi.org/10.1105/tpc.113.119792
Bhuiyan NH, Selvaraj G, Wei Y, King J (2009a) Role of lignification in plant defense. Plant Signal Behav 4:158–159. https://doi.org/10.4161/psb.4.2.7688
Bhuiyan NH, Selvaraj G, Wei Y, King J (2009b) Gene expression profiling and silencing reveal that monolignol biosynthesis plays a critical role in penetration defence in wheat against powdery mildew invasion. J Exp Bot 60:509–521. https://doi.org/10.1093/jxb/ern290
Butler NM, Atkins PA, Voytas DF, Douches DS (2015) Generation and inheritance of targeted mutations in potato (Solanum tuberosum L.) using the CRISPR/Cas system. PLoS ONE 10:e0144591
Butler NM, Baltes NJ, Voytas DF, Douches DS (2016) Geminivirus-mediated genome editing in potato (Solanum tuberosum L.) using sequence-specific nucleases. Front Plant Sci 7:1045. https://doi.org/10.3389/fpls.2016.01045
Campos L, Lisón P, López-Gresa MP et al (2014) Transgenic tomato plants overexpressing tyramine N-hydroxycinnamoyltransferase exhibit elevated hydroxycinnamic acid amide levels and enhanced resistance to Pseudomonas syringae. Mol Plant Microbe Interact 27:1159–1169
Čermák T, Baltes NJ, Čegan R et al (2015) High-frequency, precise modification of the tomato genome. Genome Biol 16:232. https://doi.org/10.1186/s13059-015-0796-9
Cingolani P, Platts A, Wang LL et al (2012) A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly 6:80–92. https://doi.org/10.4161/fly.19695
Clasen B, Stoddard T, Luo S et al (2015) Improving cold storage and processing traits in potato through targeted gene knockout. Plant Biotechnol J. https://doi.org/10.1111/pbi.12370
Collonnier C, Guyon-Debast A, Maclot F et al (2017) Towards mastering CRISPR-induced gene knock-in in plants: survey of key features and focus on the model Physcomitrella patens. Methods 121–122:103–117. https://doi.org/10.1016/j.ymeth.2017.04.024
Corpet F (1988) Multiple sequence alignment with hierarchical clustering. Nucleic Acids Res 16:10881–10890. https://doi.org/10.1093/nar/16.22.10881
Dahan-Meir T, Filler-Hayut S, Melamed-Bessudo C et al (2018) Efficient in planta gene targeting in tomato using geminiviral replicons and the CRISPR/Cas9 system. Plant J 95:5–16. https://doi.org/10.1111/tpj.13932
de Buanafina O, Marcia M (2009) Feruloylation in grasses: current and future perspectives. Mol Plant 2:861–872. https://doi.org/10.1093/mp/ssp067
Domergue F, Vishwanath SJ, Joubès J et al (2010) Three Arabidopsis fatty acyl-coenzyme A reductases, FAR1, FAR4, and FAR5, generate primary fatty alcohols associated with suberin deposition. Plant Physiol 153:1539–1554. https://doi.org/10.1104/pp.110.158238
Dong OX, Yu S, Jain R et al (2020) Marker-free carotenoid-enriched rice generated through targeted gene insertion using CRISPR-Cas9. Nat Commun 11:1178. https://doi.org/10.1038/s41467-020-14981-y
Duan H, Richael C, Rommens CM (2012) Overexpression of the wild potato eIF4E-1 variant Eva1 elicits Potato virus Y resistance in plants silenced for native eIF4E-1. Transgenic Res 21:929–938. https://doi.org/10.1007/s11248-011-9576-9
Enciso-Rodriguez F, Manrique-Carpintero NC, Nadakuduti SS et al (2019) Overcoming self-incompatibility in diploid potato using CRISPR-Cas9. Front Plant Sci 10:376
Fister AS, Landherr L, Maximova SN, Guiltinan MJ (2018) Transient expression of CRISPR/Cas9 machinery targeting TcNPR3 enhances defense response in Theobroma cacao. Front Plant Sci 9:268
Fry W (2008) Phytophthora infestans: the plant (and R gene) destroyer. Mol Plant Pathol 9:385–402
Gil-Humanes J, Wang Y, Liang Z et al (2017) High-efficiency gene targeting in hexaploid wheat using DNA replicons and CRISPR/Cas9. Plant J 89:1251–1262. https://doi.org/10.1111/tpj.13446
Hahn F, Eisenhut M, Mantegazza O, Weber APM (2018) Homology-directed repair of a defective glabrous gene in Arabidopsis with Cas9-based gene targeting. Front Plant Sci 9:424
Haverkort AJ, Struik PC, Visser RGF, Jacobsen E (2009) Applied biotechnology to combat late blight in potato caused by Phytophthora infestans. Potato Res 52:249–264. https://doi.org/10.1007/s11540-009-9136-3
Hegde N, Doddamani D, Kushalappa AC (2020) Identification and functional characterisation of late blight resistance polymorphic genes in Russet Burbank potato cultivar. Funct Plant Biol. https://doi.org/10.1071/FP19327
Humphreys JM, Hemm MR, Chapple C (1999) New routes for lignin biosynthesis defined by biochemical characterization of recombinant ferulate 5-hydroxylase, a multifunctional cytochrome P450-dependent monooxygenase. Proc Natl Acad Sci 96:10045–10050. https://doi.org/10.1073/pnas.96.18.10045
Iliakis G, Wang H, Perrault AR et al (2004) Mechanisms of DNA double strand break repair and chromosome aberration formation. Cytogenet Genome Res 104:14–20. https://doi.org/10.1159/000077461
Jensen JK, Johnson NR, Wilkerson CG (2014) Arabidopsis thaliana IRX10 and two related proteins from psyllium and Physcomitrella patens are xylan xylosyltransferases. Plant J 80:207–215. https://doi.org/10.1111/tpj.12641
Karlen SD, Zhang C, Peck ML et al (2016) Monolignol ferulate conjugates are naturally incorporated into plant lignins. Sci Adv 2:e1600393. https://doi.org/10.1126/sciadv.1600393
Kumar A, Yogendra KN, Karre S et al (2016a) WAX INDUCER1 (HvWIN1) transcription factor regulates free fatty acid biosynthetic genes to reinforce cuticle to resist Fusarium head blight in barley spikelets. J Exp Bot 67:4127–4139. https://doi.org/10.1093/jxb/erw187
Kumar S, Stecher G, Tamura K (2016b) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Kusano H, Ohnuma M, Mutsuro-Aoki H et al (2018) Establishment of a modified CRISPR/Cas9 system with increased mutagenesis frequency using the translational enhancer dMac3 and multiple guide RNAs in potato. Sci Rep 8:13753. https://doi.org/10.1038/s41598-018-32049-2
Kushalappa AC, Gunnaiah R (2013) Metabolo-proteomics to discover plant biotic stress resistance genes. Trends Plant Sci 18:522–531
Kushalappa AC, Yogendra KN, Karre S (2016a) Plant innate immune response: qualitative and quantitative resistance. Crit Rev Plant Sci 35:38–55. https://doi.org/10.1080/07352689.2016.1148980
Kushalappa AC, Yogendra KN, Sarkar K et al (2016b) Gene discovery and genome editing to develop cisgenic crops with improved resistance against pathogen infection. Can J Plant Pathol. https://doi.org/10.1080/07060661.2016.1199597
Li H, Handsaker B, Wysoker A et al (2009) The sequence alignment/map format and SAMtools. Bioinformatics 25:2078–2079. https://doi.org/10.1093/bioinformatics/btp352
Li J-F, Norville JE, Aach J et al (2013a) Multiplex and homologous recombination–mediated genome editing in Arabidopsis and Nicotiana benthamiana using guide RNA and Cas9. Nat Biotechnol 31:688–691. https://doi.org/10.1038/nbt.2654
Li X, Chen W, Zhao Y et al (2013b) Downregulation of caffeoyl-CoA O-methyltransferase (CCoAOMT) by RNA interference leads to reduced lignin production in maize straw. Genet Mol Biol 36:540–546. https://doi.org/10.1590/S1415-47572013005000039
Li J, Zhang X, Sun Y et al (2018) Efficient allelic replacement in rice by gene editing: a case study of the NRT1.1B gene. J Integr Plant Biol 60:536–540. https://doi.org/10.1111/jipb.12650
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Mao Y, Botella JR, Liu Y, Zhu J-K (2019) Gene editing in plants: progress and challenges. Natl Sci Rev 6:421–437. https://doi.org/10.1093/nsr/nwz005
Meng EC, Pettersen EF, Couch GS et al (2006) Tools for integrated sequence-structure analysis with UCSF Chimera. BMC Bioinform 7:339. https://doi.org/10.1186/1471-2105-7-339
Miki D, Zhang W, Zeng W et al (2018) CRISPR/Cas9-mediated gene targeting in Arabidopsis using sequential transformation. Nat Commun 9:1967. https://doi.org/10.1038/s41467-018-04416-0
Nelson R, Wiesner-Hanks T, Wisser R, Balint-Kurti P (2018) Navigating complexity to breed disease-resistant crops. Nat Rev Genet 19:21–33. https://doi.org/10.1038/nrg.2017.82
Nicot N, Hausman J-F, Hoffmann L, Evers D (2005) Housekeeping gene selection for real-time RT-PCR normalization in potato during biotic and abiotic stress. J Exp Bot 56:2907–2914. https://doi.org/10.1093/jxb/eri285
Peng A, Chen S, Lei T et al (2017) Engineering canker-resistant plants through CRISPR/Cas9-targeted editing of the susceptibility gene CsLOB1 promoter in citrus. Plant Biotechnol J 15:1509–1519. https://doi.org/10.1111/pbi.12733
Pertea M, Kim D, Pertea GM et al (2016) Transcript-level expression analysis of RNA-seq experiments with HISAT, StringTie and Ballgown. Nat Protoc 11:1650–1667. https://doi.org/10.1038/nprot.2016.095
Pluskal T, Castillo S, Villar-Briones A, Oresic M (2010) MZmine 2: modular framework for processing, visualizing, and analyzing mass spectrometry-based molecular profile data. BMC Bioinform 11:395. https://doi.org/10.1186/1471-2105-11-395
Puchta H, Hohn B (1996) From centiMorgans to base pairs: homologous recombination in plants. Trends Plant Sci 1(10):340–348. https://doi.org/10.1016/S1360-1385(96)82595-0
Reem NT, Pogorelko G, Lionetti V et al (2016) decreased polysaccharide feruloylation compromises plant cell wall integrity and increases susceptibility to necrotrophic fungal pathogens. Front Plant Sci 7:630. https://doi.org/10.3389/fpls.2016.00630
Sevestre F, Facon M, Wattebled F, Szydlowski N (2020) Facilitating gene editing in potato: a single-nucleotide polymorphism (SNP) map of the Solanum tuberosum L. cv. desiree genome. Sci Rep 10:2045. https://doi.org/10.1038/s41598-020-58985-6
Seybold H, Demetrowitsch TJ, Hassani MA et al (2020) A fungal pathogen induces systemic susceptibility and systemic shifts in wheat metabolome and microbiome composition. Nat Commun 11:1910. https://doi.org/10.1038/s41467-020-15633-x
Smith RA, Cass CL, Mazaheri M et al (2017) Suppression of CINNAMOYL-CoA REDUCTASE increases the level of monolignol ferulates incorporated into maize lignins. Biotechnol Biofuels 10:109. https://doi.org/10.1186/s13068-017-0793-1
Solovyev V (2007) Statistical approaches in eukaryotic gene prediction. Handb Stat Genet 97–159
Steinert J, Schiml S, Puchta H (2016) Homology-based double-strand break-induced genome engineering in plants. Plant Cell Rep 35:1429–1438
Sun G, Strebl M, Merz M et al (2019) Glucosylation of the phytoalexin N-feruloyl tyramine modulates the levels of pathogen-responsive metabolites in Nicotiana benthamiana. Plant J 100:20–37. https://doi.org/10.1111/tpj.14420
Symington LS, Gautier J (2011) Double-strand break end resection and repair pathway choice. Annu Rev Genet 45:247–271. https://doi.org/10.1146/annurev-genet-110410-132435
Uhse S, Djamei A (2018) Effectors of plant-colonizing fungi and beyond. PLoS Pathog 14:e1006992–e1006992. https://doi.org/10.1371/journal.ppat.1006992
van Esse HP, Reuber TL, van der Does D (2020) Genetic modification to improve disease resistance in crops. New Phytol 225:70–86. https://doi.org/10.1111/nph.15967
Veillet F, Chauvin L, Kermarrec M-P et al (2019a) The Solanum tuberosum GBSSI gene: a target for assessing gene and base editing in tetraploid potato. Plant Cell Rep 38:1065–1080. https://doi.org/10.1007/s00299-019-02426-w
Veillet F, Perrot L, Chauvin L et al (2019b) Transgene-free genome editing in tomato and potato plants using Agrobacterium-mediated delivery of a CRISPR/Cas9 cytidine base editor. Int J Mol Sci 20
Walters DR, Ratsep J, Havis ND (2013) Controlling crop diseases using induced resistance: challenges for the future. J Exp Bot 64:1263–1280. https://doi.org/10.1093/jxb/ert026
Wang S, Zhang S, Wang W et al (2015) Efficient targeted mutagenesis in potato by the CRISPR/Cas9 system. Plant Cell Rep 34:1473–1476. https://doi.org/10.1007/s00299-015-1816-7
Wang M, Lu Y, Botella JR et al (2017) Gene targeting by homology-directed repair in rice using a geminivirus-based CRISPR/Cas9 system. Mol Plant 10:1007–1010
Wang Y, Cheng X, Shan Q, Zhang Y, Liu J, Gao C et al (2014) Simultaneous editing of three homoeoalleles in hexaploid bread wheat confers heritable resistance to powdery mildew. Nat Biotechnol 32:947–952. https://doi.org/10.1038/nbt.2969
Wiesner-Hanks T, Nelson R (2016) Multiple disease resistance in plants. Annu Rev Phytopathol 54:229–252
Xu X, Pan S, Cheng S et al (2011) Genome sequence and analysis of the tuber crop potato. Nature 475:189–195. https://doi.org/10.1038/nature10158
Yang Q, Xuan Trinh H, Imai S et al (2004) Analysis of the involvement of hydroxyanthranilate hydroxycinnamoyltransferase and caffeoyl-CoA 3-O-methyltransferase in phytoalexin biosynthesis in oat. Mol Plant Microbe Interact 17:81–89. https://doi.org/10.1094/MPMI.2004.17.1.81
Yang J, Yan R, Roy A et al (2015) The I-TASSER suite: protein structure and function prediction. Nat Methods 12:7–8. https://doi.org/10.1038/nmeth.3213
Yang Q, He Y, Kabahuma M et al (2017) A gene encoding maize caffeoyl-CoA O-methyltransferase confers quantitative resistance to multiple pathogens. Nat Genet 49:1364
Yogendra KN, Kushalappa AC (2016) Integrated transcriptomics and metabolomics reveal induction of hierarchies of resistance genes in potato against late blight. Funct Plant Biol 15:497–7828. https://doi.org/10.1071/FP16028
Yogendra KN, Pushpa D, Mosa KA et al (2014) Quantitative resistance in potato leaves to late blight associated with induced hydroxycinnamic acid amides. Funct Integr Genomics 14:285–298. https://doi.org/10.1007/s10142-013-0358-8
Yogendra KN, Kumar A, Sarkar K et al (2015a) Transcription factor StWRKY1 regulates phenylpropanoid metabolites conferring late blight resistance in potato. J Exp Bot 66:7377–7389. https://doi.org/10.1093/jxb/erv434
Yogendra KN, Kushalappa AC, Sarmiento F et al (2015b) Metabolomics deciphers quantitative resistance mechanisms in diploid potato clones against late blight. Funct Plant Biol 42:284–298. https://doi.org/10.1071/FP14177
Zhang C, Wang P, Tang D et al (2019) The genetic basis of inbreeding depression in potato. Nat Genet 51:374–378. https://doi.org/10.1038/s41588-018-0319-1
Zhang Y, Chen M, Siemiatkowska B et al (2020) A highly efficient Agrobacterium-mediated method for transient gene expression and functional studies in multipe plant species. Plant Commun. https://doi.org/10.1016/j.xplc.2020.100028
Zhao Q, Dixon RA (2014) Altering the cell wall and its impact on plant disease: from forage to bioenergy. Annu Rev Phytopathol 52:69–91. https://doi.org/10.1146/annurev-phyto-082712-102237
Zhao Y, Zhang C, Liu W et al (2016) An alternative strategy for targeted gene replacement in plants using a dual-sgRNA/Cas9 design. Sci Rep 6:23890. https://doi.org/10.1038/srep23890
Zhao Y, Wang K, Wang W et al (2019) A high-throughput SNP discovery strategy for RNA-seq data. BMC Genomics 20:160. https://doi.org/10.1186/s12864-019-5533-4
Acknowledgements
This project was funded by the Natural Sciences and Engineering Council of Canada (NSERC) and partly by the Weston-Loblaw, Seeding Food Innovation.
Author information
Authors and Affiliations
Contributions
NH conducted all the experiments and wrote the manuscript. SJ assisted in metabolite analysis, and NS assisted in gene expression studies. ACK supervised the project and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All the studies were conducted following Environment, health and Safety, McGill, guidelines.
Additional information
Communicated by Günther Hahne.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hegde, N., Joshi, S., Soni, N. et al. The caffeoyl-CoA O-methyltransferase gene SNP replacement in Russet Burbank potato variety enhances late blight resistance through cell wall reinforcement. Plant Cell Rep 40, 237–254 (2021). https://doi.org/10.1007/s00299-020-02629-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-020-02629-6