Abstract
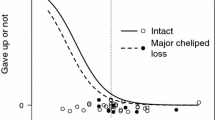
Male crustaceans engage in precopulatory mate-guarding to monopolize a female. However, a conspecific rival often takes the female from the guarding males during male–male contests, so guarding males should invest in behavior to decrease the rate of encounters with rivals, especially when they are potentially weak and guard a valuable female. We examined this idea using a net-climbing (avoidance) response by precopulatory guarding males of the hermit crab Pagurus minutus. Guarding males more often climb revetment walls in the field than solitary males and females, and the guarders were larger in body size than the solitary males on the walls. Guarding males increased climbing effort in the presence of rivals compared to controls (no stimulus), but not when faced with a non-threat organism. Compared to intact guarding males, those without a major cheliped (“weaponless”) were less likely to succeed in defending their partner in direct fights, but increased climbing investment overall, rather than especially when a rival was present. Although female size positively correlates with clutch size in this species, male climbing height decreased with increasing size of the guarded female. We discuss the effectiveness and importance of climbing in this species as it relates to conditions at our study site and the general vulnerability of guarding males depending on weapon status.





Similar content being viewed by others
References
Abrams P (1981) Shell fighting and competition between two hermit crab species in Panama. Oecologia 51:84–90. https://doi.org/10.1007/BF00344657
Bateman AJ (1948) Intra-sexual selection in Drosophila. Heredity 2:349–368. https://doi.org/10.1038/hdy.1948.21
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Button KS, Ioannidis JP, Mokrysz C, Nosek BA, Flint J, Robinson ESJ, Munafò MR (2013) Power failure: why small sample size undermines the reliability of neuroscience. Nat Rev Neurosci 14:365–376. https://doi.org/10.1038/nrn3475
Cothran RD (2004) Precopulatory mate guarding affects predation risk in two freshwater amphipod species. Anim Behav 68:1133–1138. https://doi.org/10.1016/j.anbehav.2003.09.021
Cox DR (1972) Regression models and life-tables. J R Stat Soc B 34:187–202. https://doi.org/10.1111/j.2517-6161.1972.tb00899.x
Dick JTA, Elwood RW (1990) Symmetrical assessment of female quality by male Gammarus pulex (Amphipoda) during struggles over precopula females. Anim Behav 40:877–883. https://doi.org/10.1016/S0003-3472(05)80989-3
Goshima S, Ohmori H, Wada S (1996) Reproductive biology of the hermit crab Pagurus nigrofascia (Anomura: Paguridae). Crust Res 25:86–92. https://doi.org/10.18353/crustacea.25.0_86
Goshima S, Kawashima T, Wada S (1998) Mate choice by males of the hermit crab Pagurus filholi: do males assess ripeness and/or fecundity of females? Ecol Res 13:151–161. https://doi.org/10.1046/j.1440-1703.1998.00254.x
Hazlett BA (1970) Interspecific shell fighting in three sympatric species of hermit crabs in Hawaii. Pac Sci 24:472–482
Hazlett BA, Rittschof D (2000) Predation–reproduction conflict resolution in the hermit crab, Clibanarius vittatus. Ethology 106:811–818. https://doi.org/10.1046/j.1439-0310.2000.00604.x
Helfman GS (1989) Threat-sensitive predator avoidance in damselfish-trumpetfish interactions. Behav Ecol Sociobiol 24:47–58. https://doi.org/10.1007/BF00300117
Imafuku M (1986) Sexual discrimination in the hermit crab Pagurus geminus. J Ethol 4:39–47. https://doi.org/10.1007/BF02348251
Jormalainen V (1998) Precopulatory mate guarding in crustaceans: male competitive strategy and intersexual conflict. Q Rev Biol 73:275–304. https://doi.org/10.1086/420306
Juanes F, Smith LD (1995) The ecological consequences of limb damage and loss in decapod crustaceans: a review and prospectus. J Exp Mar Biol Ecol 193:197–223. https://doi.org/10.1016/0022-0981(95)00118-2
Kaga N, Yasuda CI, Wada S (2016) Male–male contests in the hermit crab Pagurus filholi: many males do not initiate contests. Cancer 25:9–16. https://doi.org/10.18988/cancer.25.0_9
Kawaminami T, Goshima S (2015) Guarding males of the hermit crab climb up algae to avoid male–male competition. J Ethol 33:25–33. https://doi.org/10.1007/s10164-014-0411-7
Koga T, Yoshino K, Fukuda Y (2010) Temporal changes in the reproductive population structures and male’s secondary sexual character of the hermit crab Diogenes nitidimanus. Ecol Res 25:1007–1017. https://doi.org/10.1007/s11284-010-0727-x
Kuznetsova A, Brockhoff PB, Christensen RHB (2017) lmerTest package: tests in linear mixed effects models. J Stat Softw 82:1–26. https://doi.org/10.18637/jss.v082.i13
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation: a review and prospectus. Can J Zool 68:619–640. https://doi.org/10.1139/z90-092
Mathis A, Hoback WW (1997) The influence of chemical stimuli from predators on precopulatory pairing by the amphipod, Gammarus pseudolimnaeus. Ethology 103:33–40. https://doi.org/10.1111/j.1439-0310.1997.tb00004.x
Nakano R, Yasuda CI, Koga T (2016) Temporal changes in egg number and size during a single breeding season in the hermit crab Pagurus minutus. Jap J Benthol 71:32–36. https://doi.org/10.5179/benthos.71.32
Okamura S, Goshima S (2010) Indirect female choice mediated by sex pheromones in the hermit crab Pagurus filholi. J Ethol 28:323–329. https://doi.org/10.1007/s10164-009-0188-2
R Core Team (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Shuster SM (1989) Male alternative reproductive strategies in a marine isopod crustacean (Paracerceis sculpta): the use of genetic markers to measure differences in fertilization success among α-, β-, and γ-males. Evolution 43:1683–1698. https://doi.org/10.1111/j.1558-5646.1989.tb02618.x
Smith LD (1992) The impact of limb autotomy on mate competition in blue crabs Callinectes sapidus Rathbun. Oecologia 89:494–501. https://doi.org/10.1007/BF00317155
Strong Jr DR (1973) Amphipod amplexus, the significance of ecotypic variation. Ecology 54:1383–1388. https://doi.org/10.2307/1934203
Suzuki Y, Yasuda C, Takeshita F, Wada S (2012) Male mate choice and male–male competition in the hermit crab Pagurus nigrofascia: importance of female quality. Mar Biol 159:1991–1996. https://doi.org/10.1007/s00227-012-1986-6
Takeshita F, Henmi Y (2010) The effects of body size, ownership and sex-ratio on the precopulatory mate guarding of Caprella penantis (Crustacea: Amphipoda). J Mar Biol Assoc UK 90:275–279. https://doi.org/10.1017/S0025315409990671
Tanikawa D, Yasuda C, Suzuki Y, Wada S (2012) Effects of male size and mate quality on male–male contest in the hermit crab Pagurus filholi. Jpn J Benthol 67:15–19. https://doi.org/10.5179/benthos.67.15
Therneau T (2020) A package for survival analysis in R. R Package Version 3.1-12. https://CRAN.R-project.org/package=survival
Uzendoski K, Maksymovitch E, Verrell P (1993) Do the risks of predation and intermale competition affect courtship behavior in the salamander Desmognathus ochrophaeus? Behav Ecol Sociobiol 32:421–427. https://doi.org/10.1007/BF00168826
Wada S, Tanaka K, Goshima S (1999) Precopulatory mate guarding in the hermit crab Pagurus middendorffii (Brandt) (Decapoda: Paguridae): effects of population parameters on male guarding duration. J Exp Mar Biol Ecol 239:289–298. https://doi.org/10.1016/S0022-0981(99)00045-3
Yasuda C, Suzuki Y, Wada S (2011) Function of the major cheliped in male–male competition in the hermit crab Pagurus nigrofascia. Mar Biol 158:2327–2334. https://doi.org/10.1007/s00227-011-1736-1
Yasuda C, Takeshita F, Wada S (2012) Assessment strategy in male–male contests of the hermit crab Pagurus middendorffii. Anim Behav 84:385–390. https://doi.org/10.1016/j.anbehav.2012.05.007
Yasuda CI, Koga T (2016a) Do weaponless males of the hermit crab Pagurus minutus give up contests without escalation? Behavior of intruders that lack their major cheliped in male–male contests. J Ethol 34:249–254. https://doi.org/10.1007/s10164-016-0470-z
Yasuda CI, Koga T (2016b) Importance of weapon size in all stages of male–male contests in the hermit crab Pagurus minutus. Behav Ecol Sociobiol 70:2175–2183. https://doi.org/10.1007/s00265-016-2221-0
Yasuda CI, Otoda M, Nakano R, Takiya Y, Koga T (2017) Seasonal change in sexual size dimorphism of the major cheliped in the hermit crab Pagurus minutus. Ecol Res 32:347–357. https://doi.org/10.1007/s11284-017-1438-3
Acknowledgements
This study was supported by a Japan Society for the Promotion of Science Research Fellowship KAKENHI grant (no. JP18K06416) to TK and a Grant-in-Aid for Young Scientists (B) (no. 17K15188) to CIY.
Author information
Authors and Affiliations
Contributions
Conceptualization: TT, CIY, TK; methodology: TT, TK; formal analysis and investigation: CIY; writing, original draft preparation: TT, CIY, TK; writing, review and editing: CIY, TK.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Tanaka, T., Yasuda, C.I. & Koga, T. Climbing behavior of guarding males in the hermit crab Pagurus minutus: the effect of rivals, female size, and being weaponless. J Ethol 39, 115–123 (2021). https://doi.org/10.1007/s10164-020-00681-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10164-020-00681-z