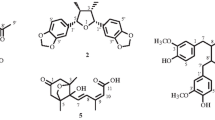
A new norneolignan derivative, clitorternalactone (1), has been isolated from the stems of Clitoria ternatea, together with six known compounds, clitorienolactone A (2), myricetin 3-glucoside (3), quercetin 3-glucoside (4), kaempferol 3-glucoside (5), taraxerol (6), and taraxerone (7). The structure of the new compound 1 was determined through spectroscopic and MS analyses. Among the isolates, compound 1 exhibited cytotoxicities with IC50 values of 2.54 ± 0.23, 3.68 ± 0.17, and 4.05 ± 0.43 μM, respectively, against DLD-1, CCRF-CEM, and IMR-32 cell lines. In addition, clitorternalactone (1), clitorienolactone A (2), quercetin 3-glucoside (4), and kaempferol 3-glucoside (5) showed potent inhibition with IC50 values of 3.05 ± 0.12, 5.44 ± 0.46, 10.20 ± 0.69, and 13.57 ± 0.36 μM, respectively, against LPS-induced NO generation.
Similar content being viewed by others
References
T. C. Huang and H. Ohashi, Leguminosae in Flora of Taiwan, Vol. 3, 2nd Ed., Editorial Committee of the Flora of Taiwan, Taipei, Taiwan, 1993, pp. 160–396.
K. Vasisht, M. Dhobi, S. Khullar, S. K. Mandal, and M. Karan, Tetrahedron Lett., 57, 1758 (2016).
S. K. Banerjee and R. N. Chakravarti, Bull. Calcutta Sch. Trop. Med., 11, 106 (1963).
R. K. Gupta and L. B. Lal, Ind. J. Pharm., 30, 167 (1968).
K. Kazuma, N. Noda, and M. Suzuki, Phytochemistry, 64, 1133 (2003).
H. Ripperger, Pharmazie, 33, 82 (1978).
N. Saito, K. Abe, T. Honda, C. F. Timberlake, and P. Bridle, Phytochemistry, 24, 1583 (1985).
B. K. Srivastava and C. S. Pande, Planta Med., 32, 138 (1977).
S. S. Joshi, R. K. Shrivastava, and D. K. Shrivastava, J. Am. Oil Chem. Soc., 58, 714 (1981).
S. Husain and K. S. Devi, J. Oil Technol. Assoc. India, 30, 162 (1998).
D. J. Taur and R. Y. Patil, J. Ethnopharmacol., 136, 374 (2011).
B. Parimaladevi, R. Boominathan, and S. C. Mandal, Fitoterapia, 74, 345 (2003).
A. D. Taranalli and T. C. Cheeramkuzhy, Pharm. Biol., 38, 51 (2000).
K. Kazuma, N. Noda, and M. Suzuki, Phytochemistry, 62, 229 (2003).
K. N. Sangeetha, S. Sujatha, V. S. Muthusamy, S. Anand, N. Nithya, D. Velmurugan, A. Balakrishnan, and B. S. Lakshmi, Biochim. Biophys. Acta, 1800, 359 (2010).
S. Chunhakant and C. Chaicharoenpong, Molecules, 24, 2798 (2019).
T. Mosmann, J. Immunol. Methods, 65, 55 (1983).
M. Johansson, B. Kopcke, H. Anke, and O. Sterner, J. Antibiot., 55, 104 (2002).
Acknowledgment
This research was supported by a grant from the Ministry of Science and Technology, Taiwan (MOST 106-2320-B-010-033-MY3), awarded to Prof. J.-J. Chen. This work was also supported by grants from Far Eastern Memorial Hospital–National Yang-Ming University Joint Research Program (108DN22), Medical Research Fund (No. 108-22) of Kaohsiung Armed Forces General Hospital, and Yuan’s General Hospital (YGH 19-027). Fu-Sen Wu, Ching-Ju Hung, and Chien-Liang Lin have contributed equally to this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 6, November–December, 2020, pp. 863–866.
Rights and permissions
About this article
Cite this article
Wu, FS., Hung, CJ., Lin, CL. et al. New Norneolignan and Bioactive Constituents of Clitoria ternatea. Chem Nat Compd 56, 1000–1004 (2020). https://doi.org/10.1007/s10600-020-03213-w
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-020-03213-w