Abstract
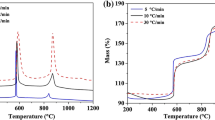
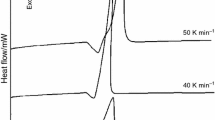
In this work, effect of milling process and CaO addition on the reaction mechanism and kinetics of aluminothermic reduction of molybdenum trioxide were studied by simultaneous thermal analysis, differential scanning calorimetry, X-ray diffraction analysis and Coats–Redfern method, respectively. For this purpose, molybdenum trioxide was reduced by Al powder under two different conditions of mechanical activation by milling process and as received form mixed by stoichiometric amount of CaO that was required for creation of CaMoO4 intermediate phase. In the case of using milled molybdenum trioxide, 20 wt% of aluminum oxide was used as heat absorber. The results showed that by using mechanically activated MoO3, the reduction reactions proceeded through the formation of intermediate phases of Al2(MoO4)3 and MoO2. In the presence of CaO, the intermediate phase was changed to CaMoO4. In both cases, the reaction temperatures and their activation energies decreased. The kinetic model for the aluminothermic reduction of un-milled and milled molybdenum trioxide was determined as chemical control, where by addition of CaO, mechanism of the reduction reaction was changed to diffusion control.













Similar content being viewed by others
References
Dang J, Zhang G H, Chou K C, Reddy R G, He Y, Sun Y, Int J Refract Met Hard Mater 41 (2013) 216.
Keshavarz Alamdari E, Trans Indian Inst Met 70 (2017) 1995.
Wasim S, Guodong Z H, Ghufranud D, Xiangxian M, Trans Indian Inst Met 72 (2019) 559.
Khabbaz S, Honarbakhsh-Raouf A, Ataie A, Saghafi M, Int J Refract Met Hard Mater 41 (2013) 402.
Wang D H, Sun G D, Zhang G H, Int J Refract Met Hard Mater 75 (2018) 70.
Sun G D, Zhang G H, Jiao S, Chou K C, J Phys Chem C 122 (2018) 1023.
Sun G D, Zhang G H, Ji X P, Liu J K, Zhang H, Chou K C, Int J Refract Met Hard Mater 80 (2019) 11.
Manukyan K, Aydinyan S, Aghajanyan A, Grigoryan Y, Niazyan O, Kharatyan S, Int J Refract Met Hard Mater 31 (2012) 28.
Manukyan K, Mnatsakanyan R, Kharatyan S, Int J Refract Met Hard Mater 28 (2010) 601.
Hoseinpur A, Bafghi M S, Vahdati Khaki J, Int J Refract Met Hard Mater 50 (2015) 191.
Raj R, Kumari D, Prasad R, Trans Indian Inst Met 72 (2019) 11.
Aydinyan S V, Manukyan Z, Mater Sci Eng B 172 (2010) 267.
Torabi O, Golabgir M H, Tajizadegan H, Torabi H, Int J Refract Met Hard Mater 47 (2014) 18.
Sheybani K, Paydar M H, Shariat M H, Int J Refract Met Hard Mater 82 (2019) 245.
Saghafi M, Ataie A, Heshmati-Manesh S, Int J Refract Met Hard Mater 29 (2011) 419.
Ebrahimi-Kahrizsangi R, Abbasi M H, Saidi A, Chem Eng J 121 (2006) 65.
Hung Z, Zheng L, J Iron Steel Res Int 21 (2013) 51.
Outokumpu R A, HSC Chemistry Software, vol. 5.1 (2002). https://www.hsc-chemistry.com/.
Caballero J A, Conesa J A, J Anal Appl Pyrolysis 73 (2005) 85.
Vyazovkina S, Burnhamb A K, Criadoc J M, Pere L A, Popescud C, Sbirrazzuol N, Thermochim Acta 520 (2011) 1.
Doweidar H, J Non Cryst Solids 471 (2017) 344.
Hu H P, Chen Q Y, Yin Z Y, He H Y, Huang H, Trans Nonferr Met Soc China 17 (2007) 205.
Bakhshandeh S, Setoudeh N, Askari Zamani M A, Mohassel A, J Min Metall Sect B Metall 54 (2018) 313.
Sah S, Dutta K, Trans Indian Inst Met 64 (2011) 583.
Bojan J, Srec S, Bemd F, Trans Indian Inst Met 67 (2014) 629.
Chattopadhyay C, Sarkar S, Sangal S, Mondal K, Trans Indian Inst Met 67 (2014) 945.
Kelton K F, J Non Cryst Solids 274 (2000) 147.
Spillar V, Dolejs D, Geochim Cosmochim Acta 131 (2014) 164.
Corezzi S, Fioretto D, Santucci G, Kenny J, Polymer 51 (2010) 5833.
Uche A K, Chude O, Malekian R, Maharaj B T, J Adv Signal Process 89 (2015) 23.
Nagla E, Hefny E, J Phys Sci 28 (2017) 129.
Moukhina E, J Therm Anal Calorim 109 (2012) 1203.
Vyazovkin S, Wight A, Int Rev Phys Chem 17 (1998) 407.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sheybani, K., Paydar, M.H. & Shariat, M.H. Investigation on the Kinetics and Mechanism of Aluminothermic Reduction of Molybdenum Trioxide: Non-isothermal Kinetics. Trans Indian Inst Met 73, 2875–2888 (2020). https://doi.org/10.1007/s12666-020-02088-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12666-020-02088-3