Abstract
Despite adherence to treatment, individuals living with HIV have an increased risk for developing cognitive impairments, referred to as HIV-associated neurological disorders (HAND). Due to continued growth in the HIV population, particularly amongst the aging cohort, the neurobiological mechanisms of HAND are increasingly relevant. Similar to other viral proteins (e.g. Tat, Gp120, Vpr), the Negative Factor (Nef) is associated with numerous adverse effects in the CNS as well as cognitive impairments. In particular, emerging data indicate the consequences of Nef may be facilitated by the modulation of cellular autophagy as well as its inclusion into extracellular vesicles (EVs). The present review examines evidence for the molecular mechanisms by which Nef might contribute to neuronal dysfunction underlying HAND, with a specific focus on autophagy and EVs. Based on the these data, we propose an integrated model by which Nef may contribute to underlying neuronal dysfunction in HAND and highlight potentially novel therapeutic targets for HAND.

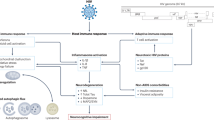
Graphical abstract


Similar content being viewed by others
References
Abudayyeh OO et al (2019) A cytosine deaminase for programmable single-base RNA editing. Science 365:382–386. https://doi.org/10.1126/science.aax7063
AIDS U (2019) Global HIV & Aids statistics - 2019 fact sheet. https://www.unaids.org/sites/default/files/media_asset/UNAIDS_FactSheet_en.pdf
Ances BM, Hammoud DA (2014) Neuroimaging of HIV-associated neurocognitive disorders (HAND). Curr Opin HIV AIDS 9:545–551. https://doi.org/10.1097/COH.0000000000000112
Anthony I, Ramage S, Carnie F, Simmonds P, Bell J (2005) Influence of HAART on HIV-related CNS disease and neuroinflammation. J Neuropathol Exp Neurol 64:529–536
Antinori A et al (2007) Updated research nosology for HIV-associated neurocognitive disorders. Neurology 69:1789–1799
Anyanwu SI, Doherty A, Powell MD, Obialo C, Huang MB, Quarshie A, Mitchell C, Bashir K, Newman GW (2018) Detection of HIV-1 and human proteins in urinary extracellular vesicles from HIV+ patients. Adv Virol 2018:7863412. https://doi.org/10.1155/2018/7863412
Arakelyan A, Fitzgerald W, Zicari S, Vanpouille C, Margolis L (2017) Extracellular Vesicles Carry HIV Env and Facilitate Hiv Infection of Human Lymphoid Tissue. Sci Rep 7:1695. https://doi.org/10.1038/s41598-017-01739-8
Arold S, Franken P, Strub M-P, Hoh F, Benichou S, Benarous R, Dumas C (1997) The crystal structure of HIV-1 Nef protein bound to the Fyn kinase SH3 domain suggests a role for this complex in altered T cell receptor signaling. Structure 5:1361–1372. https://doi.org/10.1016/S0969-2126(97)00286-4
Ayuso-Mateos JL, Pereda M, Gómez del Barrio A, Echevarria S, Fariñas MC, García-Palomo D (2000) Slowed Reaction Time in HIV-1-Seropositive Intravenous Drug Users without AIDS. Eur Neurol 44:72–78. https://doi.org/10.1159/000008200
Barnes D (1987) Solo actions of AIDS virus coat. Science 237:971–973. https://doi.org/10.1126/science.3039663
Behrens G et al (1999) Impaired glucose tolerance, beta cell function and lipid metabolism in HIV patients under treatment with protease inhibitors. Aids 13:F63–F70. https://doi.org/10.1097/00002030-199907090-00001
Bissel SJ, Wiley CA (2004) Human immunodeficiency virus infection of the brain: pitfalls in evaluating infected/affected cell populations. Brain Pathol 14:97–108. https://doi.org/10.1111/j.1750-3639.2004.tb00503.x
Byrd DA, Fellows RP, Morgello S, Franklin D, Heaton RK, Deutsch R, Atkinson JH, Clifford DB, Collier AC, Marra CM, Gelman B, McCutchan J, Duarte NA, Simpson DM, McArthur J, Grant I, CHARTER Group (2011) Neurocognitive Impact of Substance Use in HIV Infection. JAIDS J Acquir Immune Defic Syndr 58:154–162. https://doi.org/10.1097/QAI.0b013e318229ba41
Campbell TD, Khan M, Huang MB, Bond VC, Powell MD (2008) HIV-1 Nef protein is secreted into vesicles that can fuse with target cells and virions. Ethn Dis 18:S2-14-19
Campbell GR, Rawat P, Bruckman RS, Spector SA (2015) Human immunodeficiency virus type 1 Nef inhibits autophagy through transcription factor EB sequestration. PLoS Pathog 11:e1005018. https://doi.org/10.1371/journal.ppat.1005018
Carey AN, Sypek EI, Singh HD, Kaufman MJ, McLaughlin JP (2012) Expression of HIV-Tat protein is associated with learning and memory deficits in the mouse. Behav Brain Res 229:48–56
Carroll A, Brew B (2017) HIV-associated neurocognitive disorders: recent advances in pathogenesis, biomarkers, and treatment. F1000 Res 6:312. https://doi.org/10.12688/f1000research.10651.1
Castro-Gonzalez S et al. (2020) HIV-1 Nef counteracts autophagy restriction by enhancing the association between BECN1 and its inhibitor BCL2 in a PRKN-dependent manner. Autophagy 1-25 https://doi.org/10.1080/15548627.2020.1725401
Chen NC, Partridge AT, Sell C, Torres C, Martín-García J (2017) Fate of microglia during HIV-1 infection: from activation to senescence?. Microglial Dysfunction during HIV-1 infection. Glia 65:431–446. https://doi.org/10.1002/glia.23081
Cherner M et al (2005) Hepatitis C augments cognitive deficits associated with HIV infection and methamphetamine. Neurology 64:1343–1347. https://doi.org/10.1212/01.WNL.0000158328.26897.0D
Cheung R, Ravyn V, Wang L, Ptasznik A, Collman RG (2008) Signaling mechanism of HIV-1 gp120 and Virion-induced IL-1β release in primary human macrophages. J Immunol 180:6675–6684. https://doi.org/10.4049/jimmunol.180.10.6675
Chiesi A, Vella S, Dally LG, Pedersen C, Danner S, Johnson AM, Schwander S, Goebel FD, Glauser M, Antunes F (1996) Epidemiology of AIDS Dementia Complex in Europe. JAIDS J Acquir Immune Defic Syndr 11:39–44
Chiramel A, Brady N, Bartenschlager R (2013) Divergent Roles of Autophagy in Virus Infection. Cells 2:83–104. https://doi.org/10.3390/cells2010083
Chompre G, Cruz E, Maldonado L, Rivera-Amill V, Porter JT, Noel RJ (2013) Astrocytic expression of HIV-1 Nef impairs spatial and recognition memory. Neurobiol Dis 49:128–136. https://doi.org/10.1016/j.nbd.2012.08.007
Chompre G, Martinez-Orengo N, Cruz M, Porter JT, Noel RJ (2019) TGFβRI antagonist inhibits HIV-1 Nef-induced CC chemokine family ligand 2 (CCL2) in the brain and prevents spatial learning impairment. J Neuro Inflamm 16:1–19
Churchill MJ, Wesselingh SL, Cowley D, Pardo CA, McArthur JC, Brew BJ, Gorry PR (2009) Extensive astrocyte infection is prominent in human immunodeficiency virus-associated dementia. Ann Neurol 66:253–258. https://doi.org/10.1002/ana.21697
Clifford DB, Ances BM (2013) HIV-associated neurocognitive disorder. Lancet Infect Dis 13:976–986. https://doi.org/10.1016/S1473-3099(13)70269-X
Colombo M, Raposo G, Thery C (2014) Biogenesis, secretion, and intercellular interactions of Exosomes and other extracellular vesicles. In: Schekman R, Lehmann R (eds) Annual review of cell and developmental biology, Vol 30, vol 30. Annual review of cell and developmental biology. Annual Reviews, Palo Alto, pp 255–289. https://doi.org/10.1146/annurev-cellbio-101512-122326
Crowe S, Zhu T, Muller WA (2003) The contribution of monocyte infection and trafficking to viral persistence, and maintenance of the viral reservoir in HIV infection. J Leukoc Biol 74:635–641. https://doi.org/10.1189/jlb.0503204
Davenport MP, Khoury DS, Cromer D, Lewin SR, Kelleher AD, Kent SJ (2019) Functional cure of HIV: the scale of the challenge. Nat Rev Immunol 19:45–54. https://doi.org/10.1038/s41577-018-0085-4
Deacon NJ et al (1995) Genomic structure of an attenuated quasi species of HIV-1 from a blood transfusion donor and recipients. Science 270:988–991. https://doi.org/10.1126/science.270.5238.988
Deeks SG, Lewin SR, Havlir DV (2013) The end of AIDS: HIV infection as a chronic disease. Lancet 382:1525–1533. https://doi.org/10.1016/S0140-6736(13)61809-7
Delassus S, Cheynier R, Wain-Hobson S (1991) Evolution of human immunodeficiency virus type 1 nef and long terminal repeat sequences over 4 years in vivo and in vitro. J Virol 65:225–231
Di Bonito P et al (2017) Antitumor HPV E7-specific CTL activity elicited by in vivo engineered exosomes produced through DNA inoculation. Int J Nanomedicine 12:4579–4591. https://doi.org/10.2147/ijn.S131309
Ellis R, Langford D, Masliah E (2007) HIV and antiretroviral therapy in the brain: neuronal injury and repair. Nat Rev Neurosci 8:33–44
Eugenin EA, Clements JE, Zink MC, Berman JW (2011) Human immunodeficiency virus infection of human astrocytes disrupts blood-brain barrier integrity by a gap junction-dependent mechanism. J Neurosci 31:9456–9465. https://doi.org/10.1523/JNEUROSCI.1460-11.2011
Fackler OT et al (1997) Association of Human Immunodeficiency Virus Nef Protein with actin is Myristoylation dependent and influences its subcellular localization. Eur J Biochem 247:843–851. https://doi.org/10.1111/j.1432-1033.1997.00843.x
Falutz J (2007) Therapy insight: body-shape changes and metabolic complications associated with HIV and highly active antiretroviral therapy. Nat Rev Endocrinol 3:651–661. https://doi.org/10.1038/ncpendmet0587
Fan Y, He JJ (2016) HIV-1 tat induces unfolded protein response and endoplasmic reticulum stress in astrocytes and causes neurotoxicity through glial fibrillary acidic protein (GFAP) activation and aggregation. J Biol Chem 291:22819–22829
Fauci AS, Marston HD (2015) Ending the HIV–AIDS Pandemic — Follow the Science. N Engl J Med 373:2197–2199. https://doi.org/10.1056/NEJMp1502020
Fazeli PL, Crowe M, Ross AL, Wadley V, Ball K, Vance ED (2014) Cognitive Functioning In Adults Aging With HIV: A Cross-Sectional Analysis Of Cognitive Subtypes And Influential Factors. J CRHAP 1:54–68. https://doi.org/10.14302/issn.2324-7339.jcrhap-13-191
Fernandes SP, Edwards TM, Ng KT, Robinson SR (2007) HIV-1 protein gp120 rapidly impairs memory in chicks by interrupting the glutamate–glutamine cycle. Neurobiol Learn Mem 87:1–8. https://doi.org/10.1016/j.nlm.2006.03.006
Ferrantelli F, Manfredi F, Chiozzini C, Anticoli S, Olivetta E, Arenaccio C, Federico M (2018) DNA vectors generating engineered Exosomes potential CTL vaccine candidates against AIDS. Hepat B Tumors Mol Biotechnol 60:773–782. https://doi.org/10.1007/s12033-018-0114-3
Ferrantelli F et al (2019) The intracellular delivery of anti-HPV16 E7 scFvs through engineered extracellular vesicles inhibits the proliferation of HPV-infected cells. Int J Nanomedicine 14:8755–8768. https://doi.org/10.2147/ijn.S209366
Fields J et al (2013) Age-dependent molecular alterations in the autophagy pathway in HIVE patients and in a gp120 tg mouse model: reversal with beclin-1 gene transfer. J NeuroVirol 19:89–101. https://doi.org/10.1007/s13365-012-0145-7
Fields J et al (2015) HIV-1 tat alters neuronal autophagy by modulating autophagosome fusion to the lysosome: implications for HIV-associated neurocognitive disorders the journal of neuroscience. Off J Soc Neurosci 35:1921–1938. https://doi.org/10.1523/JNEUROSCI.3207-14.2015
Fitting S, Booze RM, Mactutus CF (2008) Neonatal intrahippocampal injection of the HIV-1 proteins gp120 and tat: differential effects on behavior and the relationship to stereological hippocampal measures. Brain Res 1232:139–154
Fitting S, McLaurin KA, Booze RM, Mactutus CF (2018) Dose-dependent neurocognitive deficits following postnatal day 10 HIV-1 viral protein exposure: relationship to hippocampal anatomy parameters. Int J Dev Neurosci 65:66–82
Frankel AD, Young JAT (1998) HIV-1: fifteen proteins and an RNA. Annu Rev Biochem 67:1–25. https://doi.org/10.1146/annurev.biochem.67.1.1
Fujii Y, Otake K, Tashiro M, Adachi A (1996) Soluble Nef antigen of HIV-1 is cytotoxic for human CD4+ T cells. FEBS Lett 393:93–96. https://doi.org/10.1016/0014-5793(96)00859-9
Gartner S, Markovits P, Markovitz D, Kaplan M, Gallo R, Popovic M (1986) The role of mononuclear phagocytes in HTLV-III/LAV infection. Science 233:215–219. https://doi.org/10.1126/science.3014648
Garvey LJ, Pavese N, Politis M, Ramlackhansingh A, Brooks DJ, Taylor-Robinson SD, Winston A (2014) Increased microglia activation in neurologically asymptomatic HIV-infected patients receiving effective ART. AIDS 28:67–72. https://doi.org/10.1097/01.aids.0000432467.54003.f7
Geyer M, Peterlin BM (2001) Domain assembly, surface accessibility and sequence conservation in full length HIV-1 Nef. FEBS Lett 496:91–95. https://doi.org/10.1016/S0014-5793(01)02394-8
Geyer M, Munte CE, Schorr J, Kellner R, Kalbitzer HR (1999) Structure of the anchor-domain of myristoylated and non-myristoylated HIV-1 Nef protein 1 1Edited by a. R Fersht. J Mol Biol 289:123–138. https://doi.org/10.1006/jmbi.1999.2740
Ghorpade A et al (2005) Neuroinflammatory responses from microglia recovered from HIV-1-infected and seronegative subjects. J Neuroimmunol 163:145–156. https://doi.org/10.1016/j.jneuroim.2005.01.022
Gisslén M, Price RW, Nilsson S (2011) The definition of HIV-associated neurocognitive disorders: are we overestimating the real prevalence? BMC Infect Dis 11:356. https://doi.org/10.1186/1471-2334-11-356
Glass JD, Fedor H, Wesselingh SL, McArthur JC (1995) Immunocytochemical quantitation of human immunodeficiency virus in the brain: correlations with dementia. Ann Neurol 38:755–762
Glick D, Barth S, Macleod KF (2010) Autophagy: cellular and molecular mechanisms. J Pathol 221:3–12
Goodkin K et al (2001) Aging and neuro-AIDS conditions and the changing spectrum of HIV-1-associated morbidity and mortality. J Clin Epidemiol 54:S35–S43. https://doi.org/10.1016/S0895-4356(01)00445-0
Gorry PR et al (2007) Pathogenicity and immunogenicity of attenuated, nef-deleted HIV-1 strains in vivo. Retrovirology 4:66. https://doi.org/10.1186/1742-4690-4-66
Grant I et al (2014) Asymptomatic HIV-associated neurocognitive impairment increases risk for symptomatic decline. Neurology 82:2055–2062. https://doi.org/10.1212/WNL.0000000000000492
group obotPs et al (2016) Defining cognitive impairment in people-living-with-HIV: the POPPY study. BMC Infect Dis 16:617. https://doi.org/10.1186/s12879-016-1970-8
Gupta MK et al (2017) HIV-1 Nef-induced cardiotoxicity through dysregulation of autophagy. Sci Rep 7:8572. https://doi.org/10.1038/s41598-017-08736-x
Hanna Z, Kay DG, Rebai N, Guimond A, Jothy S, Jolicoeur P (1998) Nef harbors a Major determinant of pathogenicity for an AIDS-like disease induced by HIV-1 in transgenic mice. Cell 95:163–175. https://doi.org/10.1016/S0092-8674(00)81748-1
Harricharan R, Thaver V, Russell VA, Daniels WM (2015) Tat-induced histopathological alterations mediate hippocampus-associated behavioural impairments in rats. Behav Brain Funct 11:3
Hategan A et al (2017) HIV Tat protein and amyloid-β peptide form multifibrillar structures that cause neurotoxicity. Nat Struct Mol Biol 24:379
Heaton RK et al (2010) HIV-associated neurocognitive disorders persist in the era of potent antiretroviral therapy. Charter Stud Neurol 75:2087–2096. https://doi.org/10.1212/WNL.0b013e318200d727
Hestad KA, Chinyama J, Anitha MJ, Ngoma MS, McCutchan JA, Franklin DR, Heaton RK (2019) Cognitive Impairment in Zambians With HIV Infection and Pulmonary Tuberculosis. JAIDS J Acquir Immune Def Syndr 80:110–117. https://doi.org/10.1097/QAI.0000000000001880
Holden CP, Haughey NJ, Nath A, Geiger JD (1999) Role of Na+/H+ exchangers, excitatory amino acid receptors and voltage-operated Ca2+ channels in human immunodeficiency virus type 1 gp120-mediated increases in intracellular Ca2+ in human neurons and astrocytes. Neuroscience 91:1369–1378. https://doi.org/10.1016/S0306-4522(98)00714-3
Huang M-B, Weeks O, Zhao L-J, Saltarelli M, Bond VC (2000) Effects of extracellular human immunodeficiency virus type 1 vpr protein in primary rat cortical cell cultures. J Neuro-Oncol 6:202–220
Huang Y, Zhao L, Jia B, Wu L, Li Y, Curthoys N, Zheng JC (2011) Glutaminase Dysregulation in HIV-1-infected human microglia mediates neurotoxicity: relevant to HIV-1-associated neurocognitive disorders. J Neurosci 31:15195–15204. https://doi.org/10.1523/JNEUROSCI.2051-11.2011
Hui L, Chen X, Haughey NJ, Geiger JD (2012) Role of endolysosomes in HIV-1 tat-induced neurotoxicity. ASN Neuro 4:243–252. https://doi.org/10.1042/AN20120017
Jacobs IR et al (2019) Inhibitory control deficits associated with Upregulation of CB 1 R in the HIV-1 tat transgenic mouse model of Hand. J NeuroImmune Pharmacol 14:661–678
James CO, Huang MB, Khan M, Garcia-Barrio M, Powell MD, Bond VC (2004) Extracellular Nef protein targets CD4+ T cells for apoptosis by interacting with CXCR4 surface receptors. J Virol 78:3099–3109
Janssen RS, Nwanyanwu OC, Selik RM, Stehr-Green JK (1992) Epidemiology of human immunodeficiency virus encephalopathy in the United States. Neurology 42:1472–1472. https://doi.org/10.1212/WNL.42.8.1472
Johnson TP, Patel K, Johnson KR, Maric D, Calabresi PA, Hasbun R, Nath A (2013) Induction of IL-17 and nonclassical T-cell activation by HIV-tat protein. Proc Natl Acad Sci 110:13588–13593
Kamal S, Locatelli I, Wandeler G, Sehhat A, Bugnon O, Metral M, du Pasquier R, Gutbrod K, Cavassini M, Schneider MP, the Swiss HIV Cohort Study (2017) The presence of human immunodeficiency virus-associated neurocognitive disorders is associated with a lower adherence to combined antiretroviral treatment. Open Forum Infect Dis 4:ofx070. https://doi.org/10.1093/ofid/ofx070
Kaul M, Garden GA, Lipton SA (2001) Pathways to neuronal injury and apoptosis in HIV-associated dementia. Nature 410:988–994. https://doi.org/10.1038/35073667
Kaur J, Debnath J (2015) Autophagy at the crossroads of catabolism and anabolism. Nat Rev Mol Cell Biol 16(8):461–472. https://doi.org/10.1038/nrm4024
Kestler HW, Ringler DJ, Mori K, Panicali DL, Sehgal PK, Daniel MD, Desrosiers RC (1991) Importance of the nef gene for maintenance of high virus loads and for development of AIDS. Cell 65:651–662. https://doi.org/10.1016/0092-8674(91)90097-i
Khan MB, Lang MJ, Huang M-B, Raymond A, Bond VC, Shiramizu B, Powell MD (2016) Nef exosomes isolated from the plasma of individuals with HIV-associated dementia (HAD) can induce Aβ1–42 secretion in SH-SY5Y neural cells. J NeuroVirol 22:179–190. https://doi.org/10.1007/s13365-015-0383-6
Killian M (2012) Dual role of autophagy in HIV-1 replication and pathogenesis. AIDS Res Ther 9:16. https://doi.org/10.1186/1742-6405-9-16
Kim HJ, Martemyanov KA, Thayer SA (2008) Human immunodeficiency virus protein Tat induces synapse loss via a reversible process that is distinct from cell death. J Neurosci 28:12604–12613
Kitayama H, Miura Y, Ando Y, Hoshino S, Ishizaka Y, Koyanagi Y (2008) Human immunodeficiency virus type 1 Vpr inhibits axonal outgrowth through induction of mitochondrial dysfunction. J Virol 82:2528–2542
Koedel U et al (1999) HIV type 1 Nef protein is a viral factor for leukocyte recruitment into the central nervous system. J Immunol 163:1237–1245
Koenig S et al (1986) Detection of AIDS virus in macrophages in brain tissue from AIDS patients with encephalopathy. Science 233:1089–1093. https://doi.org/10.1126/science.3016903
Konadu KA, Chu J, Huang MB, Amancha PK, Armstrong W, Powell MD, Villinger F, Bond VC (2015) Association of Cytokines with Exosomes in the plasma of HIV-1-seropositive individuals. J Infect Dis 211:1712–1716
Kyei GB et al (2009) Autophagy pathway intersects with HIV-1 biosynthesis and regulates viral yields in macrophages. J Cell Biol 186:255–268. https://doi.org/10.1083/jcb.200903070
Lamers SL, Poon AF, McGrath MS (2011) HIV-1 nef protein structures associated with brain infection and dementia pathogenesis. PLoS One. 6(2):e16659. https://doi.org/10.1371/journal.pone.0016659
Lamers SL et al (2018) Brain-specific HIV Nef identified in multiple patients with neurological disease. J Neuro-Oncol 24:1–15
Laprevotte I, Pupin M, Coward E, Didier G, Terzian C, Devauchelle C, Hénaut A (2001) HIV-1 and HIV-2 LTR nucleotide sequences: assessment of the alignment by N-block presentation, “retroviral signatures” of Overrepeated oligonucleotides, and a probable important role of scrambled stepwise duplications/deletions in molecular evolution. Mol Biol Evol 18:1231–1245. https://doi.org/10.1093/oxfordjournals.molbev.a003909
Lattanzi L, Federico M (2012) A strategy of antigen incorporation into exosomes: comparing cross-presentation levels of antigens delivered by engineered exosomes and by lentiviral virus-like particles. Vaccine 30:7229–7237. https://doi.org/10.1016/j.vaccine.2012.10.010
Learmont JC et al (1999) Immunologic and Virologic status after 14 to 18 years of infection with an attenuated strain of HIV-1 — a report from the Sydney blood Bank cohort. N Engl J Med 340:1715–1722. https://doi.org/10.1056/NEJM199906033402203
Lee C-H, Saksela K, Mirza UA, Chait BT, Kuriyan J (1996) Crystal structure of the conserved Core of HIV-1 Nef Complexed with a Src family SH3. Domain Cell 85:931–942. https://doi.org/10.1016/S0092-8674(00)81276-3
Lehmann MH, Lehmann JM, Erfle V (2019) Nef-induced CCL2 expression contributes to HIV/SIV brain invasion and neuronal dysfunction. Front Immunol 10:2447
Lenassi M et al (2010) HIV Nef is secreted in Exosomes and triggers apoptosis in bystander CD4+ T. Cells Traffic 11:110–122. https://doi.org/10.1111/j.1600-0854.2009.01006.x
Lennemann NJ, Coyne CB (2015) Catch me if you can: the link between autophagy and viruses. PLoS Pathog 11:e1004685. https://doi.org/10.1371/journal.ppat.1004685
Levy E (2017) Exosomes in the diseased brain: first insights from in vivo studies. Front Neurosci 11:142. https://doi.org/10.3389/fnins.2017.00142
Li W, Galey D, Mattson MP, Nath A (2005) Molecular and cellular mechanisms of neuronal cell death in HIV dementia. Neurotox Res 8:119–134. https://doi.org/10.1007/bf03033824
Liu J, Li L (2019) Targeting autophagy for the treatment of Alzheimer’s disease: challenges and opportunities. Front Mol Neurosci 12:203. https://doi.org/10.3389/fnmol.2019.00203
Liu J, Liu W, Yang H (2019a) Balancing apoptosis and autophagy for Parkinson's disease therapy: targeting BCL-2 ACS. Chem Neurosci 10:792–802. https://doi.org/10.1021/acschemneuro.8b00356
Liu S et al (2019b) The dual role of HIV-1 gp120 V3 loop-induced autophagy in the survival and apoptosis of the primary rat hippocampal neurons. Neurochem Res 44:1636–1652. https://doi.org/10.1007/s11064-019-02788-3
Louboutin J-P, Agrawal L, BAS R, van Bockstaele EJ, Strayer DS (2012) Gene delivery of antioxidant enzymes inhibits human immunodeficiency virus type 1 gp120-induced expression of caspases. Neuroscience 214:68–77. https://doi.org/10.1016/j.neuroscience.2012.03.061
Mamik MK, Hui E, Branton WG, McKenzie BA, Chisholm J, Cohen EA, Power C (2017) HIV-1 viral protein R activates NLRP3 inflammasome in microglia: implications for HIV-1 associated neuroinflammation. J NeuroImmune Pharmacol 12:233–248
Manfredi F, Di Bonito P, Arenaccio C, Anticoli S, Federico M (2016) Incorporation of Heterologous Proteins in Engineered Exosomes Methods. Mol Biol 1448:249–260. https://doi.org/10.1007/978-1-4939-3753-0_18
Marra CM et al (2013) Neurocognitive impairment in HIV-infected individuals with previous syphilis. Int J STD AIDS 24:351–355. https://doi.org/10.1177/0956462412472827
McArthur JC, Steiner J, Sacktor N, Nath A (2010) HIV-associated neurocognitive disorders: ‘mind the gap’. Ann Neurol. https://doi.org/10.1002/ana.22053
McDonnell J, Haddow L, Daskalopoulou M, Lampe F, Speakman A, Gilson R, Phillips A, Sherr L, Wayal S, Harrison J, Antinori A, Maruff P, Schembri A, Johnson M, Collins S, Rodger A, Cognitive Impairment in People with HIV in the European Region (CIPHER) Study Group (2014) Minimal Cognitive Impairment in UK HIV-Positive Men Who Have Sex With Men: Effect of Case Definitions and Comparison With the General Population and HIV-Negative Men. JAIDS J Acquir Immune Def Syndr 67:120–127. https://doi.org/10.1097/QAI.0000000000000273
McNamara RP et al (2018) Nef secretion into extracellular vesicles or Exosomes is conserved across human and simian immunodeficiency viruses. mBio 9:e02344–e02317. https://doi.org/10.1128/mBio.02344-17
Meyer JP, Althoff AL, Altice FL (2013) Optimizing care for HIV-infected people who use drugs: evidence-based approaches to overcoming healthcare disparities. Clin Infect Dis 57:1309–1317. https://doi.org/10.1093/cid/cit427
Mordelet E et al (2004) Histopathological and cognitive defects induced by Nef in the brain. FASEB J 18:1851–1861. https://doi.org/10.1096/fj.04-2308com
Mukhamedova N, Hoang A, Dragoljevic D, Dubrovsky L, Pushkarsky T, Low H, Ditiatkovski M, Fu Y, Ohkawa R, Meikle PJ, Horvath A, Brichacek B, Miller YI, Murphy A, Bukrinsky M, Sviridov D (2019) Exosomes containing HIV protein Nef reorganize lipid rafts potentiating inflammatory response in bystander cells. PLoS Pathog 15:e1007907. https://doi.org/10.1371/journal.ppat.1007907
Na H, Acharjee S, Jones G, Vivithanaporn P, Noorbakhsh F, McFarlane N, Maingat F, Ballanyi K, Pardo CA, Cohen ÉA, Power C (2011) Interactions between human immunodeficiency virus (HIV)-1 Vpr expression and innate immunity influence neurovirulence. Retrovirology 8:44
Narciso P et al (2001) Metabolic and morphologic disorders in patients treated with highly active antiretroviral therapy since primary HIV infection annals of the New York. Acad Sci 946:214–222. https://doi.org/10.1111/j.1749-6632.2001.tb03914.x
Nath A (2002) Human immunodeficiency virus (HIV) proteins in neuropathogenesis of HIV dementia. J Infect Dis 186:S193–S198
Nath A, Conant K, Chen P, Scott C, Major EO (1999) Transient exposure to HIV-1 tat protein results in cytokine production in macrophages and astrocytes a hit and run phenomenon. J Biol Chem 274:17098–17102
Nookala AR, Schwartz DC, Chaudhari NS, Glazyrin A, Stephens EB, Berman NE, Kumar A (2018) Methamphetamine augment HIV-1 tat mediated memory deficits by altering the expression of synaptic proteins and neurotrophic factors brain. Behavior Immun 71:37–51
Patel CA, Mukhtar M, Pomerantz RJ (2000) Human immunodeficiency virus type 1 Vpr induces apoptosis in human neuronal cells. J Virol 74:9717–9726. https://doi.org/10.1128/JVI.74.20.9717-9726.2000
Piller SC, Jans P, Gage PW, Jans DA (1998) Extracellular HIV-1 virus protein R causes a large inward current and cell death in cultured hippocampal neurons: implications for AIDS pathology. Proc Natl Acad Sci 95:4595–4600. https://doi.org/10.1073/pnas.95.8.4595
Pope M et al (1994) Conjugates of dendritic cells and memory T lymphocytes from skin facilitate productive infection with HIV-1. Cell 78:389–398. https://doi.org/10.1016/0092-8674(94)90418-9
Pužar Dominkuš P, Ferdin J, Plemenitaš A, Peterlin BM, Lenassi M (2017) Nef is secreted in exosomes from Nef.GFP-expressing and HIV-1-infected human astrocytes. J Neuro-Oncol 23:713–724
Rahimian P, He JJ (2016a) Exosome-associated release, uptake, and neurotoxicity of HIV-1 tat protein. J Neuro-Oncol 22:774–788. https://doi.org/10.1007/s13365-016-0451-6
Rahimian P, He JJ (2016b) HIV-1 tat-shortened neurite outgrowth through regulation of microRNA-132 and its target gene expression. J Neuroinflammation 13:247–247. https://doi.org/10.1186/s12974-016-0716-2
Ranki A et al (1995) Abundant expression of HIV Nef and rev proteins in brain astrocytes in vivo is associated with dementia. AIDS 9:1001–1008. https://doi.org/10.1097/00002030-199509000-00004
Rao VR, Ruiz AP, Prasad VR (2014) Viral and cellular factors underlying neuropathogenesis in HIV associated neurocognitive disorders (HAND). AIDS Res Ther 11:13. https://doi.org/10.1186/1742-6405-11-13
Raposo G, Stoorvogel W (2013) Extracellular vesicles: Exosomes, microvesicles, and friends. J Cell Biol 200:373–383. https://doi.org/10.1083/jcb.201211138
Raymond AD, Campbell-Sims TC, Khan M, Lang M, Huang MB, Bond VC, Powell MD (2011) HIV type 1 Nef is released from infected cells in CD45(+) microvesicles and is present in the plasma of HIV-infected individuals. AIDS Res Hum Retrovir 27:167–178
Raymond AD et al (2016) Microglia-derived HIV Nef+ exosome impairment of the blood–brain barrier is treatable by nanomedicine-based delivery of Nef peptides. J Neuro Virol 22:129–139. https://doi.org/10.1007/s13365-015-0397-0
Reddy PVB, Agudelo M, Atluri VSR, Nair MP (2012) Inhibition of nuclear factor Erythroid 2-related factor 2 exacerbates HIV-1 gp120-induced oxidative and inflammatory response: role in HIV associated neurocognitive disorder. Neurochem Res 37:1697–1706. https://doi.org/10.1007/s11064-012-0779-0
Reggiori F, Komatsu M, Finley K, Simonsen A (2012) Autophagy: More Than a Nonselective Pathway. Int J Cell Biol 2012:1–18. https://doi.org/10.1155/2012/219625
Rempel HC, Pulliam L (2005) HIV-1 tat inhibits neprilysin and elevates amyloid β. AIDS 19:127–135
Rho MB, Wesselingh S, Glass JD, Mcarthur JC, Choi S, Griffin J, Tyor WR (1995) A potential role for interferon-α in the pathogenesis of HIV-associated dementia. Brain Behav Immun 9:366–377. https://doi.org/10.1006/brbi.1995.1034
Rom I, Deshmane SL, Mukerjee R, Khalili K, Amini S, Sawaya BE (2009) HIV-1 Vpr deregulates calcium secretion in neural cells. Brain Res 1275:81–86
Ryan EL, Morgello S, Isaacs K, Naseer M, Gerits P, Bank TMHB (2004) Neuropsychiatric impact of hepatitis C on advanced HIV. Neurology 62:957–962. https://doi.org/10.1212/01.WNL.0000115177.74976.6C
Sabatier JM et al (1991) Evidence for neurotoxic activity of tat from human immunodeficiency virus type 1. J Virol 65:961–967
Sabbah EN, Roques BP (2005) Critical implication of the (70–96) domain of human immunodeficiency virus type 1 Vpr protien in apoptosis of primary rat cortical and straital neurons. J Neuro-Oncol 11:489–502
Sanford R, Fernandez Cruz AL, Scott SC, Mayo NE, Fellows LK, Ances BM, Collins DL (2017) Regionally Specific Brain Volumetric and Cortical Thickness Changes in HIV-Infected Patients in the HAART Era. JAIDS J Acquir Immune Def Syndr 74:563–570. https://doi.org/10.1097/QAI.0000000000001294
Saribas AS, Khalili K, Sariyer IK (2015) Dysregulation of autophagy by HIV-1 Nef in human astrocytes. Cell Cycle 14:2899–2904. https://doi.org/10.1080/15384101.2015.1069927
Saribas AS, Cicalese S, Ahooyi TM, Khalili K, Amini S, Sariyer IK (2018) HIV-1 Nef is released in extracellular vesicles derived from astrocytes: evidence for Nef-mediated neurotoxicity. Cell Death Dis 8:e2542–e2542. https://doi.org/10.1038/cddis.2016.467
Saylor D et al (2016) HIV-associated neurocognitive disorder — pathogenesis and prospects for treatment. Nat Rev Neurol 12:234–248. https://doi.org/10.1038/nrneurol.2016.27
Schouten J et al (2016) Determinants of reduced cognitive performance in HIV-1-infected middle-aged men on combination antiretroviral therapy. AIDS 30:1027–1038. https://doi.org/10.1097/QAD.0000000000001017
Sengupta S, Siliciano RF (2018) Targeting the latent reservoir for HIV-1. Immunity 48:872–895. https://doi.org/10.1016/j.immuni.2018.04.030
Simioni S, Cavassini M, Annoni J-M, Rimbault Abraham A, Bourquin I, Schiffer V, Calmy A, Chave J-P, Giacobini E, Hirschel B, Du Pasquier RA (2009) Cognitive dysfunction in HIV patients despite long-standing suppression of viremia. AIDS 24(9):1243–50. https://doi.org/10.1097/QAD.0b013e3283354a7b
Spudich S, Gonzalez-Scarano F (2012) HIV-1-related central nervous system disease: current issues in pathogenesis, Diagnosis, and Treatment. Cold Spring Harb Perspect Med 2:a007120–a007120. https://doi.org/10.1101/cshperspect.a007120
Su T, Schouten J, Geurtsen GJ, Wit FW, Stolte IG, Prins M, Portegies P, Caan MW, Reiss P, Majoie CB, Schmand BA (2015) Multivariate normative comparison, a novel method for more reliably detecting cognitive impairment in HIV infection. Aids 29(5):547–557. https://doi.org/10.1097/qad.0000000000000573
Swanson CM, Malim MH (2008) SnapShot: HIV-1 proteins. Cell 133:742–742.e741
Tetta C, Ghigo E, Silengo L, Deregibus MC, Camussi G (2013) Extracellular vesicles as an emerging mechanism of cell-to-cell communication. Endocrine 44:11–19. https://doi.org/10.1007/s12020-012-9839-0
Thompson KA et al (2003) Decreased Neurotropism of nef long terminal repeat ( nef /LTR)-deleted simian immunodeficiency virus. J Neuro-Oncol 9:442–451. https://doi.org/10.1080/13550280390218715
Torres L, Noel RJ (2014) Astrocytic expression of HIV-1 viral protein R in the hippocampus causes chromatolysis, synaptic loss and memory impairment. J Neuroinflammation 11:53
Trickey A et al (2017) Survival of HIV-positive patients starting antiretroviral therapy between 1996 and 2013: a collaborative analysis of cohort studies. Lancet HIV 4:e349–e356. https://doi.org/10.1016/S2352-3018(17)30066-8
Trillo-Pazos G, McFarlane-Abdulla E, Campbell IC, Pilkington GJ, Everall IP (2000) Recombinant nef HIV-IIIB protein is toxic to human neurons in culture. Brain Res 864:315–326. https://doi.org/10.1016/s0006-8993(00)02213-7
Tyor WR, Glass JD, Griffin JW, Becker PS, McArthur JC, Bezman L, Griffin DE (1992) Cytokine expression in the brain during the acquired immunodeficiency syndrome. Ann Neurol 31:349–360 https://doi.org/10.1002/ana.410310402
Valcour V et al (2004) Age, apolipoprotein E4, and the risk of HIV dementia: the Hawaii aging with HIV cohort. J Neuroimmunol 157:197–202. https://doi.org/10.1016/j.jneuroim.2004.08.029
Valcour V, Chalermchai T, Sailasuta N, Marovich M, Lerdlum S, Suttichom D, Suwanwela NC, Jagodzinski L, Michael N, Spudich S, van Griensven F, de Souza M, Kim J, Ananworanich J, RV254/SEARCH 010 Study Group (2012) Central Nervous System Viral Invasion and Inflammation During Acute HIV Infection. J Infect Dis 206:275–282. https://doi.org/10.1093/infdis/jis326
van Marle G et al (2004) Human immunodeficiency virus type 1 Nef protein mediates neural cell death: a neurotoxic role for IP-10. Virology 329:302–318. https://doi.org/10.1016/j.virol.2004.08.024
Wang Z et al (2004) Effects of human immunodeficiency virus type 1 on astrocyte gene expression and function: potential role in neuropathogenesis. J Neuro-Oncol 10:25–32. https://doi.org/10.1080/753312749
Wang Y, Santerre M, Tempera I, Martin K, Mukerjee R, Sawaya BE (2017) HIV-1 Vpr disrupts mitochondria axonal transport and accelerates neuronal aging. Neuropharmacology 117:364–375
Weissman D, Li Y, Orenstein JM, Fauci AS (1995) Both a precursor and a mature population of dendritic cells can bind HIV. However, only the mature population that expresses CD80 can pass infection to unstimulated CD4+ T cells. J Immunol 155:4111–4117
Whitmer RA, Gustafson DR, Barrett-Connor E, Haan MN, Gunderson EP, Yaffe K (2008) Central obesity and increased risk of dementia more than three decades later. Neurology 71:1057–1064. https://doi.org/10.1212/01.wnl.0000306313.89165.ef
Williams KC et al (2001) Perivascular macrophages are the primary cell type productively infected by simian immunodeficiency virus in the brains of macaques: implications for the neuropathogenesis of AIDS. J Exp Med 193:905–915. https://doi.org/10.1084/jem.193.8.905
World Health Organization (2018) The top 10 causes of death. https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death
Wyatt R, Sodroski J (1998) The HIV-1 envelope glycoproteins: fusogens, antigens, and immunogens. Science 280:1884–1888
Yeung MC, Pulliam L, Lau AS (1995) The HIV envelope protein gp120 is toxic to human brain-cell cultures through the induction of interleukin-6 and tumor necrosis factor-α. AIDS 9:137–144. https://doi.org/10.1097/00002030-199509020-00004
Zhao X, Fan Y, Vann PH, Wong JM, Sumien N, He JJ (2020) Long-term HIV-1 tat expression in the brain led to neurobehavioral, pathological, and epigenetic changes reminiscent of accelerated aging. Aging Dis 11:93–107
Acknowledgments
This work was supported, in part, by grants awarded by the NIH to IKS. SSY and MD are supported by Interdisciplinary and Translational Research Training in NeuroAIDS (T32MH079785).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yarandi, S.S., Duggan, M.R. & Sariyer, I.K. Emerging Role of Nef in the Development of HIV Associated Neurological Disorders. J Neuroimmune Pharmacol 16, 238–250 (2021). https://doi.org/10.1007/s11481-020-09964-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11481-020-09964-1