Abstract
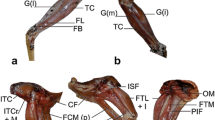
The relationship between muscle architectural properties and hind foot drumming of African mole-rats has yet to be determined using established methodology. Therefore, the internal structure of 32 hind limb muscles was evaluated in two drumming and one non-drumming species of Bathyergidae. The muscle mass (MM), fascicle length (Lf), and angle of pennation were measured to calculate the physiological cross-sectional area (PCSA) as well as estimate the maximum isometric force of contraction (Fmax). The most significant differences for the various muscle architecture parameters analyzed in synergistic muscle groups and individual muscles were observed between the rapid drumming Georychus capensis and the non-drumming Cryptomys hottentotus natalensis. The PCSA values of the hip extensors, hip adductors, knee extensors, and knee flexors of G. capensis were significantly larger than that of C. h. natalensis. Additionally, the hip extensors and knee flexors of both the drumming species (G. capensis and Bathyergus suillus) were shown to be capable of higher power output compared to the non-drumming species, and the hip adductors of G. capensis capable of faster contraction. M. gracilis anticus may play a key role in facilitating hind foot drumming as it was the only muscle to be significantly different in G. capensis and C. h. natalensis for all three muscle architecture parameters analyzed. Furthermore, it features in the high shortening capacity quadrant of the functional space plot of both G. capensis and B. suillus but not the non-drumming C. h. natalensis.





Similar content being viewed by others
Change history
24 November 2020
A Correction to this paper has been published: https://doi.org/10.1007/s10914-020-09531-8
References
Allen V, Elsey RM, Jones N, Wright J, Hutchinson JR (2010) Functional specialization and ontogenetic scaling of limb anatomy in Alligator mississippiensis. J Anat 216:423–445
Bennett NC, Faulkes CG (2000) African Mole-rats: Ecology and Eusociality. Cambridge University Press, Cambridge
Bennett NC, Jarvis JUM (1988) The reproductive biology of the Cape mole-rat, Georychus capensis (Rodentia: Bathyergidae). J Zool 214: 95-106
Bennett NC, Faulkes CG, Hart L, Jarvis JUM (2009) Bathyergus suilus (Rodentia: Bathyergidae). Mammal Species 828:1–7
Bennett NC, Maree S, Faulkes CG (2006) Georychus capensis. Mammal Species 799: 1–4
Brown M, Louw DG (2007) Automatic panoramic image stitching using invariant features. Int J Comput Vis: 74:59–73
Burkholder TJ, Fingad, B, Baron S, Lieber R (1994) Relationship between muscle fiber types and sizes and muscle architectural properties in the mouse hindlimb. J Morphol 221: 177–190
Charles JP, Cappellari O, Spence AJ, Hutchinson JR, Wells DJ (2016) Musculoskeletal geometry, muscle architecture and functional specialisations of the mouse hindlimb. PLoS ONE 11:e0147669
Eng CM, Smallwood LH, Rainiero MP, Lahey M, Ward SR, Lieber RL (2008) Scaling of muscle architecture and fiber types in the rat hindlimb. J Exp Biol 211: 2336–2345
Francescoli G (2000) Sensory capabilities and communication in subterranean rodents. In: Lacey EA, Patton JL, Cameron GN (eds) Life Underground: The Biology of Subterranean Rodents. University of Chicago Press, Chicago, pp 111–144
Fukunaga T, Roy RR, Shellock FG, Hodgson JA, Edgerton VR (1996) Specific tension of human plantar flexors and dorsiflexors. J Appl Physiol 80: 158–65
Hart L, Bennett NC, Malpaux B, Chimimba CT, Oosthuizen MK (2004) The chronobiology of the Natal mole-rat, Cryptomys hottentotus natalensis. Physiol Behav 82:563–569
Hart L, O’Riain MJ, Jarvis JUM, Bennett NC (2006) The pituitary potential for opportunistic breeding in the Cape dune mole-rat, Bathyergus suillus. Physiol Behav 88: 615–619
Hill PSM (2009) How do animals use substrate-borne vibrations as an information source? Naturwissenschaften 96: 1355–1371
Hudson PE, Corr SA, Payne-Davis RC, Clancy SN, Lane E, Wilson AM (2011) Functional anatomy of the cheetah (Acinonyx jubatus) hindlimb. J Anat 218: 363–374
Jarvis JUM, Bennett NC (1991) Ecology and behaviour of the family Bathyergidae. In: Sherman PW, Jarvis JUM, Alexander RD (eds) The Biology of the Naked Mole-rat. Princeton University Press, New York, pp 66–96
Kikuchi Y (2010) Comparative analysis of muscle architecture in primate arm and forearm. Anat Histol Embryol 39: 93–106
Kikuchi Y, Kuraoka A (2014) Differences in muscle dimensional parameters between non-formalin-fixed (freeze-thawed) and formalin-fixed specimen in gorilla (Gorilla gorilla). Mammal Study 39: 65–72
Lacey EA, Patton JL, Cameron GN (eds) (2000) Life Underground: The Biology of Subterranean Rodents. University of Chicago Press, Chicago
Lee DV, Startebake EF, Walter RM, Carrier DR (2004) Effects of mass distribution on the mechanics of level trotting in dogs. J Exp Biol 207: 1715–1728
Lieber RL (1993) Skeletal muscle architecture: implications for muscle function and surgical tendon transfer. J Hand Ther 4: 105–113
Lieber RL, Friden J, (2000) Functional and clinical significance of skeletal muscle architecture. Muscle & Nerve 23: 1647–1666
Lieber RL, Ward SR (2011) Skeletal muscle design to meet functional demands. Philos Trans R Soc B 366: 1466–1476
Maganaris CN, Baltzopoulos V, Ball D, Sargeant AJ (2001) In vivo specific tension of human skeletal muscle. J Appl Physiol 90:865–72
Martin ML, Warburton NM, Travouillon KJ, Flemming PA (2019) Mechanical similarity across ontogeny of digging muscles in an Australian marsupial (Isoodon fusciventer). J Morphol 280: 423–435
Mason MJ, Narins PM (2001) Seismic signal use by fossorial mammals. Am J Zool 41: 1171–1184
Mason MJ, Narins PM (2010) Seismic sensitivity and communication in subterranean mammals In: O’Connell-Rodwell CE (ed) The Use of Vibrations in Communication: Properties, Mechanisms and Function across Taxa. Transworld Research Network, Kerala, pp 121–139
Medler S (2002) Comparative trends in shortening velocity and force production in skeletal muscle. Am J Physiol Regul Integr Comp 283: R368–R378
Mendez J, Keyes A (1960) Density and composition of mammalian muscle. Metabolism 9: 184–188
Moore AL, Budny JE, Russell, AP, Butcher MT (2013) Architectural specialisation of the intrinsic thoracic limb musculature of the American badger (Taxidea taxus). J Morphol 274: 35–48
Myatt JP, Crompton RH, Trope SKS (2011) Hindlimb muscle architecture in non-human great apes and a comparison of methods for analysing inter-species variation. J Anat 219: 150–166
Narins PM, Reichman OJ, Jarvis JUM, Lewis ER (1992) Seismic signal transmission between burrows of the Cape mole-rat, Georychus capensis. J Comp Physiol A 170: 13–21
Olsen RA, Glenn ZD, Cliffe RN, Butcher MT (2018) Architectural properties of sloth forelimb muscles (Pilosa: Bradypodia). J Mammal Evol 25: 573–585
Payne RC, Hutchinson JR, Robilliard JJ, Smith NC, Wilson AM (2005) Functional specialisation of pelvic limb anatomy in horses (Equus caballus). J Anat 206: 557–574
R Core Team (2013) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. URL http://www.R-project.org/
Randall JA (2001) Evolution and function of drumming as communication in mammals. Am Zool 41: 1143–1156
Randall JA (2010) Drummers and stompers: vibrational communication in mammals. In: O’Connell-Rodwell CE (ed) The Use of Vibrations in Communication: Properties, Mechanisms and Function across Taxa. Transworld Research Network, Kerala, pp 100–120
Randall JA (2014) Vibrational communication: spiders to kangaroo rats. In: Witzany G (ed) Biocommunication of Animals. Springer, Dordrecht, pp 103–133
Rose JA, Sandefur M, Huskey S, Demler JL, Butcher MT (2013) Muscle architecture and out-force potential of the thoracic limb in the Eastern mole (Scalopus aquaticus). J Morphol 274:1277–1287
Rosin S, Nyakatura JA (2017) Hind limb extensor muscle architecture reflects locomotor specialisations of a jumping and a striding quadrupedal caviomorph rodent. Zoomorphology 136: 267-277
Rupert JE, Rose JA, Organ JM, Butcher MT (2015) Forelimb muscle architecture and myosin isoform composition in the groundhog (Marmota monax). J Exp Biol 218: 194–205
Sacks RD, Roy RR (1982) Architecture of the hind limb muscle of cats: functional significance. J Morphol 173: 194–195
Sahd L, Bennett NC, Kotzé SH (2019) Hind foot drumming: morphological adaptations of the muscles and bones of the hind limb in three African mole-rat species. J Anat 235: 811–824
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9: 671–675
Smith NC, Wilson AM, Jespers KJ, Payne RC (2006) Muscle architecture and functional anatomy of the pelvic limb of the ostrich (Struthio camelus). J Anat 209:765–779
Van Sandwyk JHdT, Bennett NC (2005) Do solitary seismic signalling Cape mole-rats (Georychus capensis) demonstrate spontaneous or induced ovulation? J Zool 267:75–80
Wickiewicz TL, Roy RR, Powell PL, Edgerton VR (1983) Muscle architecture of the human lower limb. Clin Orthop Rel Res 179:275–283
Williams SB, Payne RC, Wilson AM (2007) Functional specialization of the pelvic limb of the hare (Lepus europeus). J Anat 210: 472–490
Wilson A, Lichtwark G (2011) The anatomical arrangement of muscle and tendon enhances limb versatility and locomotor performance. Philos Trans R Soc B 366: 1540–1553
Zajac FE (1989) Muscle and tendon: properties, models, scaling and application to biomechanics and motor control. Crit Rev Biomed Engng 17:359–411
Zajac FE (1992) How musculotendon architecture and joint geometry affect the capacity of muscles to move and exert force on objects: a review with application t arm and forearm tendon transfer design. J Hand Surg Am 17: 799-804
Acknowledgements
The authors have no conflict of interest to declare. The authors would like to thank Prof Martin Kidd for assistance with the statistical analysis. The financial assistance of the National Research Foundation (NRF) and SARChi Mammal Behavioural Ecology and Physiology towards this research is hereby acknowledged. Opinions expressed and conclusions arrived at, are those of the authors and are not necessarily to be attributed to the NRF.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Funding
The financial assistance of the National Research Foundation (NRF) and SARChi Mammal Behavioural Ecology and Physiology towards this research is hereby acknowledged. Opinions expressed and conclusions arrived at, are those of the authors and are not necessarily to be attributed to the NRF.
Author information
Authors and Affiliations
Contributions
Lauren Sahd performed the analysis and drafted the manuscript. Nigel Bennett provided the samples and edited the manuscript. Sanet Kotzé was the principle investigator, designed the project and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors have no conflict of interest to declare.
Ethical Clearance
Ethical approval to use the specimens was obtained from the Stellenbosch University Research Ethics Committee: Animal Care and Use (REC: ACU; SU-ACUM 16–00005)
Electronic supplementary material
ESM 1
(DOCX 78 kb)
Rights and permissions
About this article
Cite this article
Sahd, L., Bennett, N.C. & Kotzé, S.H. Hind Foot Drumming: Muscle Architecture of the Hind Limb in Three Bathyergidae Species. J Mammal Evol 28, 511–525 (2021). https://doi.org/10.1007/s10914-020-09527-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10914-020-09527-4