Abstract
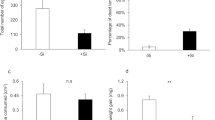
The transition from a perennial to an annual life cycle, as well as domestication, are expected to increase plant growth and reproduction at the same time that anti-herbivore defences are reduced. Here, we investigated the effects of the life-history transition (the perennial teosinte Zea diploperennis to the annual teosinte Z. mays ssp. mexicana) and domestication of Zea (annual teosinte to the modern maize Z. mays ssp. mays) on direct and indirect defences against the fall armyworm Spodoptera frugiperda. The direct defence of Zea was assessed by larval survival and nutritional indices based on food intake and utilisation. Indirect defence was measured in terms of the olfactory preference of the night-active predatory earwig Doru luteipes for nocturnal herbivore-induced plant volatiles (HIPVs) from the teosintes and maize. Larval growth and survival were reduced on teosintes relative to maize. Whilst larvae fed on perennial teosinte had lower food intake indices, those on annual teosinte showed lower food utilisation indices relative to maize. The earwig preferred HIPVs emitted by teosintes over those by maize, but it did not discriminate between odours of herbivore-damaged annual and perennial teosinte. The nocturnal HIPV blend from maize contained the lowest total amount of fatty acid derivatives, while it had higher total amounts of terpenes compared to teosintes. Our study shows that the teosintes are better defended than maize in terms of direct and indirect defences; however, the perennial teosinte have stronger direct defences against the fall armyworm than the annual teosinte.



Similar content being viewed by others
Data availability
The manuscript has not been published elsewhere and is not under consideration by another journal.
References
Aartsma Y, Leroy B, van der Werf W, Dicke M, Poelman EH, Bianchi FJ (2019) Intraspecific variation in herbivore-induced plant volatiles influences the spatial range of plant–parasitoid interactions. Oikos 128:77–86. https://doi.org/10.1111/oik.05151
Allaby RG (2014) Domestication syndrome in plants. In: Smith C (ed) Encyclopedia of global archaeology. Springer, New York, pp 2182–2184. https://doi.org/10.1007/978-1-4419-0465-2_2416
Alvarenga CD, Vendramim JD, Cruz I (1996) Efeito do predador Doru luteipes (Scud.) sobre o crescimento populacional de Schizaphis graminum (Rond.) em diferentes genótipos de sorgo. An Soc Entomol 25:137–140. https://doi.org/10.1590/S1519-566X2002000100018
Bazzaz FA, Chiariello NR, Coley PD, Pitelka LF (1987) Allocating resources to reproduction and defense. Bioscience 37:58–67. https://doi.org/10.2307/1310178
Bellota E, Medina RF, Bernal JS (2013) Physical leaf defenses—altered by Zea life-history evolution, domestication, and breeding—mediate oviposition preference of a specialist leafhopper. Entomol Exp Appl 149:185–195. https://doi.org/10.1111/eea.12122
Boos S, Meunier J, Pichon S, Kölliker M (2014) Maternal care provides antifungal protection to eggs in the European earwig. Behav Ecol 25:754–761. https://doi.org/10.1093/beheco/aru046
Chen MS (2008) Inducible direct plant defense against insect herbivores: a review. Insect Sci 15:101–114. https://doi.org/10.1006/bcon.1997.0590
Chen YH, Welter SC (2005) Crop domestication disrupts a native tritrophic interaction associated with the sunflower, Helianthus annuus (Asterales: Asteraceae). Ecol Entomol 30:673–683. https://doi.org/10.1111/j.0307-6946.2005.00737.x
Chen YH, Gols R, Benrey B (2015) Crop domestication and its impact on naturally selected trophic interactions. Ecol Entomol 30:673–683. https://doi.org/10.1111/j.0307-6946.2005.00737.x
Cipollini D, Purrington CB, Bergelson J (2003) Costs of induced responses in plants. Basic Appl Ecol 4:79–89. https://doi.org/10.1078/1439-1791-00134
Coley PD, Bryant JP, Chapin FS (1985) Resource availability and plant antiherbivore defense. Science 230:895–899. https://doi.org/10.1126/science.230.4728.895
Cruz I, Alvarenga CD, Figueiredo PEF (1995) Biologia de Doru luteipes (Scudder) e sua capacidade predatória de ovos de Helicoverpa zea (Boddie). An Soc Entomol Bras 24:273–278. https://doi.org/10.4025/actasciagron.v31i1.6602
D’Alessandro M, Turlings TCJ (2005) In situ modification of herbivore-induced plant odors: a novel approach to study the attractiveness of volatile organic compounds to parasitic wasps. Chem Senses 30:739–753. https://doi.org/10.1093/chemse/bji066
Dávila-Flores AM, Dewitt TJ, Bernal JS (2013) Facilitated by nature and agriculture: performance of a specialist herbivore improves with host-plant life history evolution, domestication, and breeding. Oecologia 173:1425–1437. https://doi.org/10.1007/s00442-013-2728-2
de Lange ES, Balmer D, Mauch-Mani B, Turlings TC (2014) Insect and pathogen attack and resistance in maize and its wild ancestors, the teosintes. New Phytol 204:329–341. https://doi.org/10.1111/nph.13005
de Lange ES, Farnier K, Gaudillat B, Turlings TC (2016) Comparing the attraction of two parasitoids to herbivore-induced volatiles of maize and its wild ancestors, the teosintes. Chemoecology 26:33–44. https://doi.org/10.1007/s00049-015-0205-6
de Lange ES, Farnier K, Degen T, Gaudillat B, Aguilar-Romero R, Bahena-Juárez F, Oyama K, Turlings TC (2018) Parasitic wasps can reduce mortality of teosinte plants infested with fall armyworm: support for a defensive function of herbivore-induced plant volatiles. Front Ecol Evol 6:55. https://doi.org/10.3389/fevo.2018.00055
De Moraes CM, Lewis WJ, Pare PW, Alborn HT, Tumlinson JH (1998) Herbivore-infested plants selectively attract parasitoids. Nature 393:570–573. https://doi.org/10.1038/31219
De Moraes CM, Mescher MC, Tumlinson JH (2001) Caterpillar-induced nocturnal plant volatiles repel nonspecific females. Nature 410:577–580. https://doi.org/10.1038/35069058
Degen T, Dillmann C, Marion-Poll F, Turlings TC (2004) High genetic variability of herbivore-induced volatile emission within a broad range of maize inbred lines. Plant Physiol 135:1928–1938. https://doi.org/10.1104/pp.104.039891
Dicke M, van Loon JJA (2000) Multitrophic effects of herbivore-induced plant volatiles in an evolutionary context. Entomol Exp Appl 97:237–249. https://doi.org/10.1046/j.1570-7458.2000.00736.x
Doebley J (2004) The genetics of maize evolution. Annu Rev Genet 38:37–59. https://doi.org/10.1146/annurev.genet.38.072902.092425
Evans LT (1993) Crop evolution, adaptation, and yield. Cambridge University Press, Cambridge
FAOSTAT (2014) Most Produced Commodities in Selected Country (World) Average 1993–2013. Available at: https://faostat3.fao.org/faostat-gateway/go/to/browse/Q/QC/E. Accessed 20 June 2019
Feeny PP (1976) Plant apparency and chemical defense. In: Wallace JW, Mansell RL (eds) Biochemical interaction between plants and insects. Recent advances in phytochemistry. Plenum Publishing, New York, pp 1–40
Gaillard MD, Glauser G, Robert CA, Turlings TC (2018) Fine-tuning the ‘plant domestication-reduced defense’ hypothesis: specialist vs generalist herbivores. New Phytol 217:355–366. https://doi.org/10.1111/nph.14757
Gols R, Wagenaar R, Bukovinszky T, Dam NMV, Dicke M, Bullock JM, Harvey JA (2008) Genetic variation in defense chemistry in wild cabbages affects herbivores and their endoparasitoids. Ecology 89:1616–1626. https://doi.org/10.1890/07-0873.1
Gols R, Bullock JM, Dicke M, Bukovinszky T, Harvey JA (2011) Smelling the wood from the trees: non-linear parasitoid responses to volatile attractants produced by wild and cultivated cabbage. J Chem Ecol 37:795–807. https://doi.org/10.1007/s10886-011-9993-5
Gonzalez-Segovia E, Pérez-Limon S, Cíntora-Martínez GC, Guerrero-Zavala A, Janzen GM, Hufford MB, Ross-Ibarra J, Sawers RJ (2019) Characterization of introgression from the teosinte Zea mays ssp. mexicana to Mexican highland maize. PeerJ 7:e6815. https://doi.org/10.7717/peerj.6815
Gouin A, Bretaudeau A, Nam K, Gimenez S, Aury JM, Duvic B, Kuwar S (2017) Two genomes of highly polyphagous lepidopteran pests (Spodoptera frugiperda, Noctuidae) with different host-plant ranges. Sci Rep 7:11816. https://doi.org/10.1038/s41598-017-10461-4
Gouinguené S, Degen T, Turlings TCJ (2001) Variability in herbivore-induced odour emissions among maize cultivars and their wild ancestors (teosinte). Chemoecology 11:9–16. https://doi.org/10.1007/PL00001832
Gutiérrez SLP, González JJS, Corral JAR, Parra JR, Medrano RM, Larios LLC, Ildefonso RL (2009) Diversidad de especies insectiles en maíz y teocintle en Mexico. Folia Entomol Mex 48:103–118
Hagenbucher S, Birgersson G, Chattington S, Anderson P (2016) Domestication influences choice behavior and performance of a generalist herbivore. Perspect Plant Ecol Evol Syst 23:63–72. https://doi.org/10.1016/j.ppees.2016.09.001
Haile A, Hofsvang T (2001) Survey of lepidopterous stem borer pests of sorghum, maize and pearl millet in Eritrea. Crop Prot 20:151–157. https://doi.org/10.1016/S0261-2194(00)00125-3
Hoballah ME, Turlings TCJ (2005) The role of fresh versus old leaf damage in the attraction of parasitic wasps to herbivore-induced maize volatiles. J Chem Ecol 31:2003–2018. https://doi.org/10.1007/s10886-005-6074-7
Hoballah ME, Degen T, Bergvinson D, Savidan A, Tamo C, Turlings TC (2004) Occurrence and direct control potential of parasitoids and predators of the fall armyworm (Lepidoptera: Noctuidae) on maize in the subtropical lowlands of Mexico. Agr Forest Entomol 6:83–88. https://doi.org/10.1111/j.1461-9555.2004.00207.x
Howe GA, Jander G (2008) Plant immunity to insect herbivores. Annu Rev Plant Biol 59:41–66. https://doi.org/10.1146/annurev.arplant.59.032607.092825
Kessler A, Baldwin IT (2001) Defensive function of herbivore-induced plant volatile emissions in nature. Science 291:2141–2144. https://doi.org/10.1126/science.291.5511.2141
Kessler A, Baldwin IT (2002) Plant responses to insect herbivory: the emerging molecular analysis. Annu Rev Plant Biol 53:299–328. https://doi.org/10.1146/annurev.arplant.53.100301.135207
Kharboutli MS, Mack TP (1993) Effect of temperature, humidity, and prey density on feeding rate of the striped earwig (Dermaptera: Labiduridae). Environ Entomol 22:1134–1139. https://doi.org/10.1093/ee/22.5.1134
Kistler L, Yoshi Maezumi S, de Souza JG, Przelomska NAS, Costa FM, Smith O, Loiselle H, Ramos-Madrigal J, Wales N, Ribeiro ER, Morrison RR, Grimaldo C, Prous AP, Arriaza B, Gilbert MTP, de Oliveira Freitas F, Allaby RG (2018) Multiproxy evidence highlights a complex evolutionary legacy of maize in South America. Science 362(6420):1309–1313
Lauter N, Gustus C, Westerbergh A, Doebley J (2004) The inheritance and evolution of leaf pigmentation and pubescence in teosinte. Genetics 167:1949–1959. https://doi.org/10.1534/genetics.104.026997
Li X, Garvey M, Kaplan I, Li B, Carrillo J (2018) Domestication of tomato has reduced the attraction of herbivore natural enemies to pest-damaged plants. Agr Forest Entomol 20:390–401. https://doi.org/10.1111/een.12438
Maag D, Erb M, Bernal JS, Wolfender JL, Turlings TC, Glauser G (2015) Maize domestication and anti-herbivore defences: leaf-specific dynamics during early ontogeny of maize and its wild ancestors. PLoS ONE 10:e0135722. https://doi.org/10.1371/journal.pone.0135722
Mas F, Haynes KF, Kölliker M (2009) A chemical signal of offspring quality affects maternal care in a social insect. Proc R Soc B Biol Sci 276:2847–2853. https://doi.org/10.1098/rspb.2009.0498
Matsuoka Y, Vigouroux Y, Goodman MM, Sanchez J, Buckler E, Doebley J (2002) A single domestication for maize shown by multilocus microsatellite genotyping. Proc Natl Acad Sci USA 99:6080–6084. https://doi.org/10.1073/pnas.052125199
Meyer RS, DuVal AE, Jensen HR (2012) Patterns and processes in crop domestication: an historical review and quantitative analysis of 203 global food crops. New Phytol 196:29–48. https://doi.org/10.1111/j.1469-8137.2012.04253.x
Morishima H, Sano Y, Oka HI (1984) Differentiation of perennial and annual types due to habitat conditions in the wild riceOryza perennis. Plant Syst Evol 144(2):119–135
Moya-Raygoza G (2016) Early development of leaf trichomes is associated with decreased damage in teosinte, compared with maize, by Spodoptera frugiperda (Lepidoptera: Noctuidae). Ann Entomol Soc Am 109:737–743. https://doi.org/10.1093/aesa/saw049
Munyiri SW, Mugo SN, Otim M, Mwololo JK, Okori P (2013) Mechanisms and sources of resistance in tropical maize inbred lines to Chilo partellus stem borers. J Agric Sci 5:51–60. https://doi.org/10.5539/jas.v5n7p51
Mutyambai DM, Bruce TJ, Midega CA, Woodcock CM, Caulfield JC, van den Berg J, Pickett JA, Khan ZR (2015) Responses of parasitoids to volatiles induced by Chilo partellus oviposition on teosinte, a wild ancestor of maize. J Chem Ecol 41:323–329. https://doi.org/10.1007/s10886-015-0570-1
Naranjo-Guevara N, Peñaflor MFG, Cabezas-Guerrero MF, Bento JMS (2017) Nocturnal herbivore-induced plant volatiles attract the generalist predatory earwig Doru luteipes Scudder. Sci Nat 104:77. https://doi.org/10.1007/s00114-017-1498-9
Nault LR, Madden LV (1985) Ecological strategies of Dalbulus leafhoppers. Ecol Entomol 10:57–63. https://doi.org/10.1111/j.1365-2311.1985.tb00534.x
Niazi IAK, Rafique A, Rauf S, da Silva JAT, Afzal M (2014) Simultaneous selection for stem borer resistance and forage related traits in maize (Zea mays ssp. mays L.) × teosinte (Zea mays ssp. mexicana L.) derived populations. Crop Prot 57:27–34. https://doi.org/10.1016/j.cropro.2013.10.026
Niinemets Ü, Kännaste A, Copolovici L (2013) Quantitative patterns between plant volatile emissions induced by biotic stresses and the degree of damage. Front Plant Sci 4:262. https://doi.org/10.3389/fpls.2013.00262
Painter RH (1955) Insects on corn and teosinte in guatemala1. J Econ Entomol 48(1):36–42
Parra JRP, Carvalho SM (1984) Biologia e nutrição quantitativa de Spodoptera frugiperda (JE Smith, 1797) em meios artificiais compostos de diferentes variedades de feijão. An Soc Entomol Bras 13:306–319
Parra JRP, Panizzi AR, Haddad ML (2012) Nutritional indices for measuring insect food intake and utilization. In: Panizzi AR, Parra JR (eds) Insect bioecology and nutrition for integrated pest management. CRC Press, Boca Raton, pp 13–50
Peñaflor MFGV (2019) Use of semiochemical-based strategies to enhance biological control. In: Souza B, Vázquez LL, Marucci RC (eds) Natural enemies of insect pests in neotropical agroecosystems, 1st edn. Springer, Cham, pp 509–522
Peñaflor MFGV, Erb M, Miranda LA, Werneburg AG, Bento JMS (2011) Herbivore-induced plant volatiles can serve as host location cues for a generalist and a specialist egg parasitoid. J Chem Ecol 37:1304–1313. https://doi.org/10.1007/s10886-011-0047-9
Rasmann S, Köllner TG, Degenhardt J, Hiltpold I, Toepfer S, Kuhlmann U, Gershenzon J, Turlings TCJ (2005) Recruitment of entomopathogenic nematodes by insect-damaged maize roots. Nature 434:732–737. https://doi.org/10.1038/nature03451
Reis LL, Oliveira LJ, Cruz I (1988) Biologia e potencial de Doru luteipes no controle de Spodoptera frugiperda. Pesqui Agropecu Bras 23:333–342
Rodriguez-Saona C, Vorsa N, Singh AP, Johnson-Cicalese J, Szendrei Z, Mescher MC, Frost CJ (2011) Tracing the history of plant traits under domestication in cranberries: potential consequences on anti-herbivore defences. J Exp Bot 62:2633–2644. https://doi.org/10.1093/jxb/erq466
Romero-Sueldo GM, Virla EG (2009) Datos biológicos de Doru luteipes (Dermaptera: Forficulidae) en plantaciones de caña de azúcar y consumo de huevos de Diatraea saccharalis (Lepidoptera: Crambidae) en condiciones de laboratorio. Rev Soc Entomol Argent 68:359–363
Rosenthal JP, Dirzo R (1997) Effects of life history, domestication and agronomic selection on plant defence against insects: evidence from maizes and wild relatives. Evol Ecol 11:337–355. https://doi.org/10.1023/A:1018420504439
Rosenthal JP, Welter SC (1995) Tolerance to herbivory by a stemboring caterpillar in architecturally distinct maize and wild relatives. Oecologia 102:146–155. https://doi.org/10.1007/BF00333245
Rowen E, Kaplan I (2016) Eco-evolutionary factors drive induced plant volatiles: a meta-analysis. New Phytol 210(1):284–294
Schoonhoven LM, van Loon JA, Dicke M (2005) Insect-plant biology. Oxford University Press, Oxford
Stebbins GL (1974) Flowering plants: evolution above the species level. Belknap Press of Harvard University Press, Cambridge
Sueldo MR, Bruzzone OA, Virla EG (2010) Characterization of the earwig, Doru lineare, as a predator of larvae of the fall armyworm, Spodoptera frugiperda: a functional response study. J Insect Sci 10:38. https://doi.org/10.1673/031.010.3801
Szczepaniec A, Widney SE, Bernal JS, Eubanks MD (2013) Higher expression of induced defenses in teosintes (Zea spp.) is correlated with greater resistance to fall armyworm, Spodoptera frugiperda. Entomol Exp Appl 146:242–251. https://doi.org/10.1111/eea.12014
Takahashi CG, Kalns LL, Bernal JS (2012) Plant defense against fall armyworm mini micro-sympatric maize (Zea mays ssp. mays) and Balsas teosinte (Zea mays ssp. parviglumis). Entomol Exp Appl 145:191–200. https://doi.org/10.1111/eea.12004
Tamiru A, Bruce TJA, Woodcock CM, Caulfield JC, Midega CAO, Ogol CKPO, Mayon P, Birkett MA, Pickett JA, Khan ZR (2011) Maize landraces recruit egg and larval parasitoids in response to egg deposition by a herbivore. Ecol Lett 14:1075–1083. https://doi.org/10.1111/j.1461-0248.2011.01674.x
Turlings TCJ, Tumlinson JH, Lewis WJ (1990) Exploitation of herbivore-induced plant odors by host-seeking parasitic wasps. Science 250:1251–1253. https://doi.org/10.1126/science.250.4985.1251
Varella AC, Menezes-Netto AC, Alonso JDS, Caixeta DF, Peterson RK, Fernandes OA (2015) Mortality dynamics of Spodoptera frugiperda (Lepidoptera: Noctuidae) immatures in maize. PLoS ONE 10:e0130437. https://doi.org/10.1371/journal.pone.0130437
Weiss MJ, McDonald G (1998) European earwig, Forficula auricularia L (Dermaptera: Forficulidae), as a predator of the redlegged earth mite, Halotydeus destructor (Tucker) (Acarina: Penthaleidae). Austr Entomol 37:183–185. https://doi.org/10.1111/j.1440-6055.1998.tb01569.x
Wiseman BR, Snook ME, Isenhour DJ, Mihm JA, Widstrom NW (1992) Relationship between growth of corn earworm and fall armyworm larvae (Lepidoptera: Noctuidae) and maysin concentration in corn silks. J Econ Entomol 85:2473–2477. https://doi.org/10.1093/jee/85.6.2473
Acknowledgements
We thank Arodi Prado for technical assistance, Dr. Flavia Franca Teixeira (Embrapa Milho e Sorgo, Germplasm Bank, Sete Lagoas, Minas Gerais, Brazil) for providing teosinte seeds, and Koen Merkus and Janet W. Reid for English-language editing. We also thank the two reviewers (Dr. Yolanda Chen and an anonymous reviewer) for their valuable comments and suggestions, which improved the quality of our manuscript. This study was supported by the National Institute of Science and Technology (INCT) Semiochemicals in Agriculture (FAPESP and CNPq, grant numbers 2014/50871-0 and 465511/2014-7, respectively). NNG was funded by Estudantes-Convênio de Pós-Graduação (PEC-PG) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), and DBS by FAPESP (Process 2016/12771-0).
Funding
This research was funded by the National Institute of Science and Technology (INCT) Semiochemicals in Agriculture (FAPESP and CNPq, Grant Numbers 2014/50871-0 and 465511/2014-7, respectively). NNG was funded by Estudantes Convênio de Pós-Graduação (PEC-PG) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
Author information
Authors and Affiliations
Contributions
Conceptualization, NNG and MFGVP; methodology, NNG and MFGVP; performed behavioural experiments and statistical analysis, NNG and DBS; writing, reviewing and editing, NNG, DBS and MFGVP; supervision, JMSB. All authors reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
Not applicable.
Additional information
Communicated by Marko Rohlfs.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Naranjo-Guevara, N., Peñaflor, M.F.G.V., Silva, D.B. et al. A comparison of the direct and indirect defence abilities of cultivated maize versus perennial and annual teosintes. Chemoecology 31, 63–74 (2021). https://doi.org/10.1007/s00049-020-00329-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00049-020-00329-x