Abstract
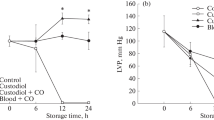
High organoprotective properties of a carbon monoxide (CO)–oxygen (O2) gas mixture were confirmed after prolonged (24-h) preservation of the papillary muscle and an isolated rat heart at 4°C. Hypothermic preservation in the high-pressure gas mixture (6 atm) provided efficient restoration of the contractile activity of the isolated rat heart after 24-h storage at 4°C. The isolated retrograde-perfused Langendorff heart performed physically relevant mechanical work, which was similar in duration to that of an intact control heart. Staining with triphenyltetrazolium chloride did not detect infarcted regions in the myocardium. After preservation, the heart tissue was highly capable of performing its function in a test for electrically stimulated contractile activity of papillary muscles. In the test group, The frequency–intensity relationship, the potentiation effect induced by a pause, and the response to stimulation with isoproterenol of test hearts generally corresponded to the parameters of a normal rat myocardium. A sheep heart, which is comparable in size and weight to a human heart, was for the first time successfully preserved using the gas mixture. Normal heartbeat was spontaneously restored after the start of perfusion in all experiments. Histology did not detect a significant difference between test and control sheep hearts. The normal tissue structure of the myocardium was preserved in the test hearts. The 24-h preservation achieved in the study was four times longer than the maximum allowable preservation time of standard static cold storage. The results obtained with the large laboratory animal heart model showed that the hypothermic preservation protocol is promising for prolonged storage of human hearts.





Similar content being viewed by others
REFERENCES
Global Observatory on Donation and Transplantation. http://www.transplant-observatory.org.
S. V. Gauthier and S. M. Khomyakov, Vestn. Transplantol. Iskusstv. Organov 21 (3), 7 (2019).
R. S. Mangus, et al., Transplantation 86, 298 (2008).
V. I. Shumakov, Heart Transplantation: A Manual forPhysicians (Moscow, 2006) [in Russian].
M. C. Ott, J. R. Scott, A. Bihari, et al., FASEB J. 19 (1), 106 (2005).
J. S. Neto, A. Nakao, K. Kimizuka, et al., Am. J. Physiol. Renal Physiol. 287 (5), 979 (2004).
A. Nakao, A. M. Choi, and N. Murase, J. Cell. Mol. Med. 10 (3), 650 (2006).
M. D. Musameh, C. J. Green, B. E. Mann, et al., J. Heart Lung Transplant. 26 (11), 1192 (2007).
L. E. Otterbein, R. Foresti, and R. Motterlini, Circ. Res. 118 (12), 1940 (2016).
A. Nakao, J. S. Neto, S. Kanno, et al., Am. J. Transplant. 5 (2), 282 (2005).
A. Nakao, G. Faleo, H. Shimizu, et al., Kidney Int. 74 (8), 1009 (2008).
N. Hatayama, M. Inubushi, M. Naito, et al., Sci. Rep. 6, 1 (2016).
N. Hatayama, S. Hirai, M. Naito, et al., Sci. Rep. 8 (1), 1 (2018).
E. E. Fesenko, N. E. Shvirst, E. L. Gagarinskii, et al., RF Patent No. RU 2 707 532 (2019).
N. V. Grudinin, V. K. Bogdanov, M. G. Sharapov, et al., Vestn. Transplantol. Iskusstv. Organov 22 (2), 20 (2020).
A. L. Szilagyi, P. Matrai, P. Hegyi, et al., World J. Gastroenterol. 24 (16), 1812 (2018).
Y. Li, S. Guo, G. Liu, et al., Artif. Organs 40 (5), 489 (2016).
J. M. O’Callaghan, S. R. Knight, R. D. Morgan, and P. J. Morris, Am. J. Transplant. 12 (4), 896 (2012).
M. Manzia, L. Toti, C. Quaranta, et al., Int. J. Surg. Rep. 57, 163 (2019).
J. N. Schroder, D. D’Alessandro, F. Esmailian, et al., J. Heart Lung Transplant. 38 (4), S42 (2019).
H. Mergental and G. R. Roll, Clin. Liver Dis. 10 (4), 97 (2017).
G. Loor, G. Warnecke, M. Villavicencio, et al., J. Heart Lung Transplant. 37 (Suppl. 4), S147 (2018).
OCS Heart System for Heart Transplant, Medtech Innovation Briefing (2016). https://www.nice.org.uk/advice/mib86/chapter/The-technology.
T. M. Suszynski, M. D. Rizzari, W. E. Scott, et al., J. Cardiothorac. Surg. 8, 105 (2013).
S. M. Minasyan, M. M. Galagudza, Yu. V. Dmitriev, et al., Region. Krovoobr. Mikrotsirk. 13 (3), 4 (2014).
ACKNOWLEDGMENTS
We are grateful to V.K. Bogdanov for help with surgery in large animals, N.P. Mozheiko for help with histological analysis of preparations, and the Sector of Optic Microscopy and Spectrophotometry (Pushchino Center of Biological Research, Russian Academy of Sciences).
Funding
This work was supported by the Ministry of Education and Science of the Russian Federation (Development of New Approaches to Cryopreservation of Biological Objects program, no. AAAA-A17-117032050021-0).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests. The authors declare that they have no conflict of interest.
Statement on the welfare of animals. The study was performed in compliance with the European Union Directive 2010/63/EU on the protection of animals used for scientific purposes. The experimental protocol was approved by the Ethics Committee at the Institute of Cell Biophysics (Russian Academy of Sciences).
Additional information
Translated by T. Tkacheva
Rights and permissions
About this article
Cite this article
Fesenko, E.E., Gagarinsky, E.L., Averin, A.S. et al. The Condition of the Rat Myocardium and Isolated Sheep Heart after Prolonged 24-Hour Hypothermic Preservation in a Pressurized Carbon Monoxide–Oxygen Gas Mixture. BIOPHYSICS 65, 666–675 (2020). https://doi.org/10.1134/S0006350920040065
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006350920040065