Abstract
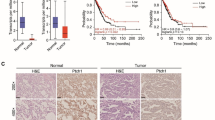
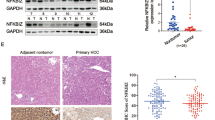
Brain metastasis (BM) is associated with poor prognosis in patients with advanced non-small cell lung cancer (NSCLC). Epidermal growth factor receptor (EGFR) mutation reportedly enhances the development of BM. However, the exact mechanism of how EGFR-mutant NSCLC contributes to BM remains unknown. Herein, we found the protein WNT5A, was significantly downregulated in BM tissues and EGFR-mutant samples. In addition, the overexpression of WNT5A inhibited the growth, migration, and invasion of EGFR-mutant cells in vitro and retarded tumor growth and metastasis in vivo compared with the EGFR wide-type cells. We demonstrated a molecular mechanism whereby WNT5A be negatively regulated by transcription factor E2F1, and ERK1/2 inhibitor (U0126) suppressed E2F1’s regulation of WNT5A expression in EGFR-mutant cells. Furthermore, WNT5A inhibited β-catenin activity and the transcriptional levels of its downstream genes in cancer progression. Our research revealed the role of WNT5A in NSCLC BM with EGFR mutation, and proved that E2F1-mediated repression of WNT5A was dependent on the ERK1/2 pathway, supporting the notion that targeting the ERK1/2-E2F1-WNT5A pathway could be an effective strategy for treating BM in EGFR-mutant NSCLC.






Similar content being viewed by others
Abbreviations
- BM:
-
Brain metastasis
- NSCLC:
-
Non-small cell lung cancer
- EGFR:
-
Epidermal growth factor receptor
- TKI:
-
Tyrosine kinase inhibitor
- ERK1/2:
-
Extracellular signal-regulated kinase ½
- WNT5A:
-
Wnt family member 5A
- ROR2:
-
Receptor tyrosine kinase-like orphan receptor 2
- β-Catenin:
-
Catenin beta 1
- E2F1:
-
E2F transcription factor 1
- PI3K:
-
Phosphoinositide 3-kinase
- HDAC:
-
Histone deacetylase
- cDNA:
-
Complementary DNA
- qRT-PCR:
-
Quantitative real-time PCR
- ChIP:
-
Chromatin immunoprecipitation
- HR:
-
Hazard ratio
- mut:
-
Mutation
- wt:
-
Wild-type
- GEO:
-
Gene Expression Omnibus
- TFs:
-
Transcriptional factors
- OS:
-
Overall survival
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424. https://doi.org/10.3322/caac.21492
Siegel RL, Miller KD, Jemal A (2017) Cancer statistics, 2017. CA Cancer J Clin 67(1):7–30. https://doi.org/10.3322/caac.21387
Langer CJ, Mehta MP (2005) Current management of brain metastases, with a focus on systemic options. J Clin Oncol 23(25):6207–6219. https://doi.org/10.1200/jco.2005.03.145
Yang J, Gong W (2019) Lorlatinib for the treatment of anaplastic lymphoma kinase-positive non-small cell lung cancer. Expert Rev Clin Pharmacol 12(3):1–6. https://doi.org/10.1080/17512433.2019.1570846
Yang Z, Guo Q, Wang Y, Chen K, Zhang L, Cheng Z, Xu Y, Yin X, Bai Y, Rabbie S, Kim DW, Ahn MJ, Yang JC, Zhang X (2016) AZD3759, a BBB-penetrating EGFR inhibitor for the treatment of EGFR mutant NSCLC with CNS metastases. Sci Transl Med 8(368):368ra172. https://doi.org/10.1126/scitranslmed.aag0976
Moscatello DK, Holgado-Madruga M, Emlet DR, Montgomery RB, Wong AJ (1998) Constitutive activation of phosphatidylinositol 3-kinase by a naturally occurring mutant epidermal growth factor receptor. J Biol Chem 273(1):200–206. https://doi.org/10.1074/jbc.273.1.200
Lorimer IA, Lavictoire SJ (2001) Activation of extracellular-regulated kinases by normal and mutant EGF receptors. Biochim Biophys Acta 1538(1):1–9. https://doi.org/10.1016/s0167-4889(00)00129-4
Tan L, Wu Y, Ma X, Yan Y, Shao S, Liu J, Ma H, Liu R, Chai L, Ren J (2019) A comprehensive meta-analysis of association between EGFR mutation status and brain metastases in NSCLC. Pathol Oncol Res POR 25(2):791–799. https://doi.org/10.1007/s12253-019-00598-0
Xiong X, Shi Q, Yang X, Wang W, Tao J (2019) LINC00052 functions as a tumor suppressor through negatively modulating miR-330-3p in pancreatic cancer. J Cell Physiol 234(9):15619–15626. https://doi.org/10.1002/jcp.28209
Asem MS, Buechler S, Wates RB, Miller DL, Stack MS (2016) Wnt5a signaling in cancer. Cancers (Basel) 8(9):79. https://doi.org/10.3390/cancers8090079
Borcherding N, Kusner D, Kolb R, Xie Q, Li W, Yuan F, Velez G, Askeland R, Weigel RJ, Zhang W (2015) Paracrine WNT5A signaling inhibits expansion of tumor-initiating cells. Can Res 75(10):1972–1982. https://doi.org/10.1158/0008-5472.CAN-14-2761
Prasad CP, Manchanda M, Mohapatra P, Andersson T (2018) WNT5A as a therapeutic target in breast cancer. Cancer Metas Rev 37(4):767–778. https://doi.org/10.1007/s10555-018-9760-y
Bauer M, Benard J, Gaasterland T, Willert K, Cappellen D (2013) WNT5A encodes two isoforms with distinct functions in cancers. PLoS ONE 8(11):e80526. https://doi.org/10.1371/journal.pone.0080526
Mikels AJ, Nusse R (2006) Purified Wnt5a protein activates or inhibits beta-catenin-TCF signaling depending on receptor context. PLoS Biol 4(4):e115. https://doi.org/10.1371/journal.pbio.0040115
Mikels A, Minami Y, Nusse R (2009) Ror2 receptor requires tyrosine kinase activity to mediate Wnt5A signaling. J Biol Chem 284(44):30167–30176. https://doi.org/10.1074/jbc.M109.041715
Schambony A, Wedlich D (2007) Wnt-5A/Ror2 regulate expression of XPAPC through an alternative noncanonical signaling pathway. Dev Cell 12(5):779–792. https://doi.org/10.1016/j.devcel.2007.02.016
Yoon S, Choi MH, Chang MS, Baik JH (2011) Wnt5a-dopamine D2 receptor interactions regulate dopamine neuron development via extracellular signal-regulated kinase (ERK) activation. J Biol Chem 286(18):15641–15651. https://doi.org/10.1074/jbc.M110.188078
Chen T, Yang Z, Liu C, Wang L, Yang J, Chen L, Li W (2019) Circ_0078767 suppresses non-small-cell lung cancer by protecting RASSF1A expression via sponging miR-330-3p. Cell Prolif 52(2):e12548. https://doi.org/10.1111/cpr.12548
Jin Z, Jia B, Tan L, Liu Y (2019) miR-330-3p suppresses liver cancer cell migration by targeting MAP2K1. Oncol Lett 18(1):314–320. https://doi.org/10.3892/ol.2019.10280
Li D, Song H, Mei H, Fang E, Wang X, Yang F, Li H, Chen Y, Huang K, Zheng L, Tong Q (2018) Armadillo repeat containing 12 promotes neuroblastoma progression through interaction with retinoblastoma binding protein 4. Nat Commun 9(1):2829. https://doi.org/10.1038/s41467-018-05286-2
Li D, Chen Y, Mei H, Jiao W, Song H, Ye L, Fang E, Wang X, Yang F, Huang K, Zheng L, Tong Q (2018) Ets-1 promoter-associated noncoding RNA regulates the NONO/ERG/Ets-1 axis to drive gastric cancer progression. Oncogene 37(35):4871–4886. https://doi.org/10.1038/s41388-018-0302-4
Slotta-Huspenina J, Drecoll E, Feith M, Habermehl D, Combs S, Weichert W, Bettstetter M, Becker K, Langer R (2018) MicroRNA expression profiling for the prediction of resistance to neoadjuvant radiochemotherapy in squamous cell carcinoma of the esophagus. J Transl Med 16(1):109. https://doi.org/10.1186/s12967-018-1492-9
Nagy A, Lanczky A, Menyhart O, Gyorffy B (2018) Validation of miRNA prognostic power in hepatocellular carcinoma using expression data of independent datasets. Sci Rep 8(1):9227. https://doi.org/10.1038/s41598-018-27521-y
Katoh M, Katoh M (2009) Transcriptional mechanisms of WNT5A based on NF-kappaB, Hedgehog, TGFbeta, and Notch signaling cascades. Int J Mol Med 23(6):763–769. https://doi.org/10.3892/ijmm_00000190
Roskoski R Jr (2014) The ErbB/HER family of protein-tyrosine kinases and cancer. Pharmacol Res 79:34–74. https://doi.org/10.1016/j.phrs.2013.11.002
Remon J, Ahn MJ, Girard N, Johnson M, Kim DW, Lopes G, Pillai RN, Solomon B, Villacampa G, Zhou Q (2019) Advanced-stage non-small cell lung cancer: advances in thoracic oncology 2018. J Thorac Oncol 14(7):1134–1155. https://doi.org/10.1016/j.jtho.2019.03.022
Ponce S, Bruna J, Juan O, López R, Navarro A, Ortega AL, Puente J, Verger E, Bartolomé A, Nadal E (2019) Multidisciplinary expert opinion on the treatment consensus for patients with EGFR mutated NSCLC with brain metastases. Crit Rev Oncol Hematol 138:190–206. https://doi.org/10.1016/j.critrevonc.2019.03.017
Ramalingam SS, Yang JC, Lee CK, Kurata T, Kim DW, John T, Nogami N, Ohe Y, Mann H, Rukazenkov Y, Ghiorghiu S, Stetson D, Markovets A, Barrett JC, Thress KS, Jänne PA (2018) Osimertinib as first-line treatment of EGFR mutation-positive advanced non-small-cell lung cancer. J Clin Oncol 36(9):841–849. https://doi.org/10.1200/jco.2017.74.7576
Wang Z, Qu H, Gong W, Liu A (2018) Up-regulation and tumor-promoting role of SPHK1 were attenuated by miR-330-3p in gastric cancer. IUBMB Life 70(11):1164–1176. https://doi.org/10.1002/iub.1934
Arora S, Ranade AR, Tran NL, Nasser S, Sridhar S, Korn RL, Ross JT, Dhruv H, Foss KM, Sibenaller Z, Ryken T, Gotway MB, Kim S, Weiss GJ (2011) MicroRNA-328 is associated with (non-small) cell lung cancer (NSCLC) brain metastasis and mediates NSCLC migration. Int J Cancer 129(11):2621–2631. https://doi.org/10.1002/ijc.25939
Xiao F, Zhang P, Wang Y, Tian Y, James M, Huang CC, Wang L, Wang L (2020) Single-nucleotide polymorphism rs13426236 contributes to an increased prostate cancer risk via regulating MLPH splicing variant 4. Mol Carcinog 59(1):45–55. https://doi.org/10.1002/mc.23127
Zhan C, Yan L, Wang L, Sun Y, Wang X, Lin Z, Zhang Y, Shi Y, Jiang W, Wang Q (2015) Identification of immunohistochemical markers for distinguishing lung adenocarcinoma from squamous cell carcinoma. J Thorac Dis 7(8):1398–1405. https://doi.org/10.3978/j.issn.2072-1439.2015.07.25
Clevers H, Nusse R (2012) Wnt/beta-catenin signaling and disease. Cell 149(6):1192–1205. https://doi.org/10.1016/j.cell.2012.05.012
Kumawat K, Gosens R (2016) WNT-5A: signaling and functions in health and disease. Cell Mol Life Sci CMLS 73(3):567–587. https://doi.org/10.1007/s00018-015-2076-y
Huang TC, Lee PT, Wu MH, Huang CC, Ko CY, Lee YC, Lin DY, Cheng YW, Lee KH (2017) Distinct roles and differential expression levels of Wnt5a mRNA isoforms in colorectal cancer cells. PLoS ONE 12(8):e0181034. https://doi.org/10.1371/journal.pone.0181034
Ren D, Dai Y, Yang Q, Zhang X (2019) Wnt5a induces and maintains prostate cancer cells dormancy in bone. J Exp Med 216(2):428–449. https://doi.org/10.1084/jem.20180661
Li L, Ying J, Tong X, Zhong L, Su X, Xiang T, Shu X, Rong R, Xiong L, Li H, Chan AT, Ambinder RF, Guo Y, Tao Q (2014) Epigenetic identification of receptor tyrosine kinase-like orphan receptor 2 as a functional tumor suppressor inhibiting beta-catenin and AKT signaling but frequently methylated in common carcinomas. Cell Mol Life Sci CMLS 71(11):2179–2192. https://doi.org/10.1007/s00018-013-1485-z
McDonald SL, Silver A (2009) The opposing roles of Wnt-5a in cancer. Br J Cancer 101(2):209–214. https://doi.org/10.1038/sj.bjc.6605174
Bradner JE, Hnisz D, Young RA (2017) Transcriptional addiction in cancer. Cell 168(4):629–643. https://doi.org/10.1016/j.cell.2016.12.013
Dittmer J (2015) The role of the transcription factor Ets1 in carcinoma. Semin Cancer Biol 35(20–38):20–38. https://doi.org/10.1016/j.semcancer.2015.09.010
Xu TP, Wang YF, Xiong WL, Ma P, Wang WY, Chen WM, Huang MD, Xia R, Wang R, Zhang EB, Liu YW, De W, Shu YQ (2017) E2F1 induces TINCR transcriptional activity and accelerates gastric cancer progression via activation of TINCR/STAU1/CDKN2B signaling axis. Cell Death Dis 8(6):e2837. https://doi.org/10.1038/cddis.2017.205
Lu Z, Luo RZ, Peng H, Huang M, Nishmoto A, Hunt KK, Helin K, Liao WS, Yu Y (2006) E2F-HDAC complexes negatively regulate the tumor suppressor gene ARHI in breast cancer. Oncogene 25(2):230–239. https://doi.org/10.1038/sj.onc.1209025
Engelmann D, Mayoli-Nussle D, Mayrhofer C, Furst K, Alla V, Stoll A, Spitschak A, Abshagen K, Vollmar B, Ran S, Putzer BM (2013) E2F1 promotes angiogenesis through the VEGF-C/VEGFR-3 axis in a feedback loop for cooperative induction of PDGF-B. J Mol Cell Biol 5(6):391–403. https://doi.org/10.1093/jmcb/mjt035
Gorgoulis VG, Zacharatos P, Mariatos G, Kotsinas A, Bouda M, Kletsas D, Asimacopoulos PJ, Agnantis N, Kittas C, Papavassiliou AG (2002) Transcription factor E2F-1 acts as a growth-promoting factor and is associated with adverse prognosis in non-small cell lung carcinomas. J Pathol 198(2):142–156. https://doi.org/10.1002/path.1121
Xiang J, Bandura J, Zhang P, Jin Y, Reuter H, Edgar BA (2017) EGFR-dependent TOR-independent endocycles support Drosophila gut epithelial regeneration. Nat Commun 8:15125. https://doi.org/10.1038/ncomms15125
Hu B, Wang Q, Wang YA, Hua S, Sauve CG, Ong D, Lan ZD, Chang Q, Ho YW, Monasterio MM, Lu X, Zhong Y, Zhang J, Deng P, Tan Z, Wang G, Liao WT, Corley LJ, Yan H, Zhang J, You Y, Liu N, Cai L, Finocchiaro G, Phillips JJ, Berger MS, Spring DJ, Hu J, Sulman EP, Fuller GN, Chin L, Verhaak RGW, DePinho RA (2016) Epigenetic activation of WNT5A drives glioblastoma stem cell differentiation and invasive growth. Cell 167(5):1281 e1218-1295 e1218. https://doi.org/10.1016/j.cell.2016.10.039
Sigismund S, Avanzato D, Lanzetti L (2018) Emerging functions of the EGFR in cancer. Mol Oncol 12(1):3–20. https://doi.org/10.1002/1878-0261.12155
Song X, Fan PD, Bantikassegn A, Guha U, Threadgill DW, Varmus H, Politi K (2015) ERBB3-independent activation of the PI3K pathway in EGFR-mutant lung adenocarcinomas. Cancer Res 75(6):1035–1045. https://doi.org/10.1158/0008-5472.can-13-1625
Truscott M, Islam AB, Lightfoot J, Lopez-Bigas N, Frolov MV (2014) An intronic microRNA links Rb/E2F and EGFR signaling. PLoS Genet 10(7):e1004493. https://doi.org/10.1371/journal.pgen.1004493
Funding
This work was supported by the National Natural Science Foundation of China (81573090, 81172595, 81703165).
Author information
Authors and Affiliations
Contributions
XRD conceived the study. HHL, LP, and JJW performed the experiments. HHL, FT, MR and RGZ analyzed the data. XRD and HHL wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethics approval and consent to participate
This study was approved by the Institutional Review Board of Huazhong University of Science and Technology. Written informed consent was obtained from all legal guardians of the patients. All animal experiments were conducted in agreement with the Guide for the Care and Use of Laboratory Animals and were approved by the Committee on Animal Handling of Huazhong University of Science and Technology.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.






Rights and permissions
About this article
Cite this article
Li, H., Tong, F., Meng, R. et al. E2F1-mediated repression of WNT5A expression promotes brain metastasis dependent on the ERK1/2 pathway in EGFR-mutant non-small cell lung cancer. Cell. Mol. Life Sci. 78, 2877–2891 (2021). https://doi.org/10.1007/s00018-020-03678-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-020-03678-6