Abstract
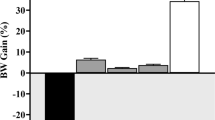
Diabetes mellitus is associated with increased oxidative stresses, kidney damage, ocular neuropathy, and atherosclerosis. In the present study, the protective effect of Rubus fruticosus hydroethanolic extract on the biochemical parameters in streptozotocin (STZ)-induced diabetic rats were investigated. In this experimental study, 40 male Wistar rats were divided into five groups (n = 8) as control, diabetic (STZ-injection 55 mg kg−1 of body weight, intraperitoneally (i.p.)), and treatment 1, 2, and 3 which treated by R. fruticosus extract (50, 100 and 200 mg kg−1, i.p.). After 4 weeks, the animals were deeply anesthetized and their blood samples were collected from the heart directly for the measurement of biochemical parameters and inflammatory cytokines. The findings indicate that the serum level of alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), gamma-glutamyl transferase (GGT), ceruloplasmin (Cp), copper (Cu), tumor necrosis factor (TNF)-α, and interleukin (IL)-6 increased significantly after the onset of diabetes (P < 0.001 for all assayed parameters). Treatment by R. fruticosus extract at high dose (200 mg kg−1) reduced the serum levels of ALT, AST, ALP, GGT, Cp, Cu, TNF-α, and IL-6 (P < 0.001, for all parameters) significantly compared to the diabetic group. Overall, the administration of the R. fruticosus extract can ameliorate some of the adverse effects of STZ-induced diabetes among the male rats that received this treatment.


Similar content being viewed by others
References
Abdu D, Majeed SN (2012) Identification of antioxidant compounds in red raspberry (Rubus idaeus) fruit in Kurdistan region (North Iraq). J Appl Chem 2:6–10
Abdulwahab RA, Allaith AA, Shinwari Z, Alaiya A, Giha HA (2019) Association of TATA box-binding protein-associated factor RNA polymerase I subunit C (TAF1C) with T2DM. Gene 706:43–51. https://doi.org/10.1016/j.gene.2019.04.076
Ahmadpour SH, Sadeghi YO, Hami J, Haghir H (2008) Effects of insulin and ascorbic acid therapy on plasma cu level in streptozotocin-induced diabetic rats. J Birjand Univ Med Sci 15:26–31
Alonso R, Cadavid I, Calleja JM (1980) A preliminary study of hypoglycemic activity of Rubus fruticosus. Planta Med 40:102–106. https://doi.org/10.1055/s-2008-1075012
Al-Shaaibi SN, Waly MI, Al-Subhi L, Tageldin MH, Al-Balushi NM, Rahman MS (2016) Ameliorative effects of pomegranate peel extract against dietary-induced nonalcoholic fatty liver in rats. Prev Nutr Food Sci 21:14–23. https://doi.org/10.3746/pnf.2016.21.1.14
AmouoghliTabrizi B, Mohajeri D (2014) Preventive effects of green tea extract from hepatic steatosis in the rats fed with high fat diet. JRUMS 13:125–140
Ashraf H, Zare S, Farnad N (2014) The effect of aqueous extract of barberry fruit on liver damage in streptozotocin-induced diabetic rats. J Shahrekord Univ Med Sci 15:1–9
Azizi V, Allahyari F, Hosseini A (2018) The antiepileptic and neuroprotective effect of the Buxus hyrcana Pojark hydroethanolic extract against the pentylentetrazol induced model of the seizures in the male rats. Int J Neurosci 128:913–919. https://doi.org/10.1080/00207454.2018.1443925
Bloch KO, Zemel R, Bloch OV, Grief H, Vardi P (2000) Streptozotocin and alloxan-based selection improves toxin resistance of insulin-producing RINm cells. Int J Exp Diabetes Res 1:211–219
Boivin D, Blanchette M, Barrette S, Moghrabi A, Beliveau R (2007) Inhibition of cancer cell proliferation and suppression of TNF-induced activation of NFkappaB by edible berry juice. Anticancer Res 27:937–948
Bonnefont-Rousselot D (2004) The role of antioxidant micronutrients in the prevention of diabetic complications. Treat Endocrinol 3:41–52
Brownlee M (2001) Biochemistry and molecular cell biology of diabetic complications. Nature 414:813–820. https://doi.org/10.1038/414813a
Cheraghi J, Krishchi P, Nasri S, Boorboor MJ (2016) The effect of ethanolic extracts of Petroselinum crispum leaves on histopathological and activity of liver enzymes in streptozotocin-induced diabetic rats. SJIMU 23:190–202
Cho MJ, Howard LR, Prior RL, Clark JR (2004) Flavonoid glycosides and antioxidant capacity of various blackberry, blueberry and red grape genotypes determined by high-performance liquid chromatography/mass spectrometry. J Sci Food Agric 84:1771–1782. https://doi.org/10.1002/jsfa.1885
Finley EB, Cerklewski FL (1983) Influence of ascorbic acid supplementation on copper status in young adult men. Am J Clin Nutr 37:553–556. https://doi.org/10.1093/ajcn/37.4.553
Ghalayini IF, Al-Ghazo MA, Harfeil MN (2011) Prophylaxis and therapeutic effects of raspberry (Rubus idaeus) on renal stone formation in Balb/c mice. Int Braz J Urol 37:259–266
Golizeh M et al (2017) Increased serotransferrin and ceruloplasmin turnover in diet-controlled patients with type 2 diabetes. Free Radic Biol Med 113:461–469. https://doi.org/10.1016/j.freeradbiomed.2017.10.373
Gomar A, Hosseini A, Mirazi N (2014) Preventive effect of Rubus fruticosus on learning and memory impairment in an experimental model of diabetic neuropathy in male rats. PharmaNutrition 2:155–160
Harada S et al (2017) Influence of hyperglycemia on liver inflammatory conditions in the early phase of non-alcoholic fatty liver disease in mice. J Pharm Pharmacol 69:698–705. https://doi.org/10.1111/jphp.12705
Harris EH (2005) Elevated liver function tests in type 2 diabetes. Clin Diabetes 23:115–119
Jean-Gilles D, Li L, Ma H, Yuan T, Chichester CO III, Seeram NP (2012) Anti-inflammatory effects of polyphenolic-enriched red raspberry extract in an antigen-induced arthritis rat model. J Agric Food Chem 60:5755–5762. https://doi.org/10.1021/jf203456w
Jelodar G, Mohsen M, Shahram S (2007) Effect of walnut leaf, coriander and pomegranate on blood glucose and histopathology of pancreas of alloxan induced diabetic rats. Afr J Tradit Complem 4:299–305
Karganov MY, Alchinova IB, Tinkov AA et al (2020) Streptozotocin (STZ)-induced diabetes affects tissue trace element content in rats in a dose-dependent manner. Biol Trace Elem Res. https://doi.org/10.1007/s12011-020-02090-2
Kazi TG, Afridi HI, Kazi N, Jamali MK, Arain MB, Jalbani N, Kandhro GA (2008) Copper, chromium, manganese, iron, nickel, and zinc levels in biological samples of diabetes mellitus patients. Biol Trace Elem Res 122:1–18. https://doi.org/10.1007/s12011-007-8062-y
Keshavarz M, Hasanain P, Parviz M, Mansoori M, Soltani N, Mirazi N (2006) Oral magnesium sulfate in prevention of diabetic neuropathy in mice. Tehran Univ Med J 64:37–46
King GL (2008) The role of inflammatory cytokines in diabetes and its complications. J Periodontol 79:1527–1534. https://doi.org/10.1902/jop.2008.080246
Koca I, Karadeniz B (2009) Antioxidant properties of blackberry and blueberry fruits grown in the Black Sea Region of Turkey. Sci Hortic 121:447–450
Lee TH, Kim WR, Poterucha JJ (2012) Evaluation of elevated liver enzymes. Clin Liver Dis 16:183–198. https://doi.org/10.1016/j.cld.2012.03.006
Mansour HA, Newairy AS, Yousef MI, Sheweita SA (2002) Biochemical study on the effects of some Egyptian herbs in alloxan-induced diabetic rats. Toxicology 170:221–228
Milne DB, Klevay LM, Hunt JR (1988) Effects of ascorbic acid supplements and a diet marginal in copper on indices of copper nutriture in women. Nutr Res 8:865–873
Mishra KP, Ganju L, Sairam M, Banerjee PK, Sawhney RC (2008) A review of high throughput technology for the screening of natural products. Biomed Pharmacother 62:94–98. https://doi.org/10.1016/j.biopha.2007.06.012
Mishu FA, Boral N, Ferdous N, Nahar S, Sultana GS, Yesmin MS, Khan NZ (2019) Estimation of serum zinc, copper and magnesium levels in bangladeshi women with gestational diabetes mellitus attending in a tertiary care hospital. Mymensingh Med J 28:157–162
Neha K, Haider MR, Pathak A, Yar MS (2019) Medicinal prospects of antioxidants: a review. Eur J Med Chem 178:687–704. https://doi.org/10.1016/j.ejmech.2019.06.010
Newsholme P, Cruzat VF, Keane KN, Carlessi R, de Bittencourt PIH Jr. (2016) Molecular mechanisms of ROS production and oxidative stress in diabetes. Biochem J 473:4527–4550. https://doi.org/10.1042/BCJ20160503C
Patar AK, Sharma A, Syiem D, Bhan S (2018) Chlorophyllin supplementation modulates hyperglycemia-induced oxidative stress and apoptosis in liver of streptozotocin-administered mice. BioFactors (Oxford, England) 44:418–430. https://doi.org/10.1002/biof.1438
Qiu Q, Zhang F, Zhu W, Wu J, Liang M (2017) Copper in diabetes mellitus: a meta-analysis and systematic review of plasma and serum studies. Biol Trace Elem Res 177:53–63. https://doi.org/10.1007/s12011-016-0877-y
Reddy MK, Alexander-Lindo RL, Nair MG (2005) Relative inhibition of lipid peroxidation, cyclooxygenase enzymes, and human tumor cell proliferation by natural food colors. J Agric Food Chem 53:9268–9273. https://doi.org/10.1021/jf051399j
Sangiovanni E, Vrhovsek U, Rossoni G, Colombo E, Brunelli C, Brembati L, Trivulzio S, Gasperotti M, Mattivi F, Bosisio E, Dell’Agli M (2013) Ellagitannins from Rubus berries for the control of gastric inflammation: in vitro and in vivo studies. PLoS ONE 8:e71762. https://doi.org/10.1371/journal.pone.0071762
Sarkar A, Dash S, Barik BK, Muttigi MS, Kedage V, Shetty JK, Prakash M (2010) Copper and ceruloplasmin levels in relation to total thiols and GST in type 2 diabetes mellitus patients. Indian J Clin Biochem 25:74–76. https://doi.org/10.1007/s12291-010-0015-0
Skalnaya MG, Skalny AV, Tinkov AA (2017) Serum copper, zinc, and iron levels, and markers of carbohydrate metabolism in postmenopausal women with prediabetes and type 2 diabetes mellitus. J Trace Elem Med Biol 43:46–51. https://doi.org/10.1016/j.jtemb.2016.11.005
Sobczak AIS, Stefanowicz F, Pitt SJ, Ajjan RA, Stewart AJ (2019) Total plasma magnesium, zinc, copper and selenium concentrations in type-I and type-II diabetes. Biometals 32:123–138. https://doi.org/10.1007/s10534-018-00167-z
Uriu-Adams JY, Rucker RB, Commisso JF, Keen CL (2005) Diabetes and dietary copper alter 67Cu metabolism and oxidant defense in the rat. J Nutr Biochem 16:312–320. https://doi.org/10.1016/j.jnutbio.2005.01.007
Verma R, Gangrade T, Punasiya R, Ghulaxe C (2014) Rubus fruticosus (blackberry) use as an herbal medicine. Pharmacogn Rev 8:101–104. https://doi.org/10.4103/0973-7847.134239
Wada L, Ou B (2002) Antioxidant activity and phenolic content of Oregon caneberries. J Agric Food Chem 50:3495–3500
Wang LS et al (2009) Anthocyanins in black raspberries prevent esophageal tumors in rats. Cancer Prev Res 2:84–93. https://doi.org/10.1158/1940-6207.Capr-.08-0155
Zafra-Stone S, Yasmin T, Bagchi M, Chatterjee A, Vinson JA, Bagchi D (2007) Berry anthocyanins as novel antioxidants in human health and disease prevention. Mol Nutr Food Res 51:675–683. https://doi.org/10.1002/mnfr.200700002
Zia-Ul-Haq M, Riaz M, De Feo V, Jaafar HZ, Moga M (2014) Rubus fruticosus L.: constituents, biological activities and health related uses. Molecules 19:10998–11029. https://doi.org/10.3390/molecules190810998
Zikri NN, Riedl KM, Wang LS, Lechner J, Schwartz SJ, Stoner GD (2009) Black raspberry components inhibit proliferation, induce apoptosis, and modulate gene expression in rat esophageal epithelial cells. Nutr Cancer 61:816–826. https://doi.org/10.1080/01635580903285148
Acknowledgements
The authors of this article are aware of the gratitude and appreciation of the high-ranking officials of the Vice-Chancellor for Research and Technology, and the graduates of the Islamic Azad University of Damghan, as well as, the staff of the medical diagnostic laboratory of the city of Hamedan for their comprehensive assistance and cooperation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Soheilifar, M., Shiravi, A., Mirazi, N. et al. Modulatory Effect of Rubus fruticosus L. on Liver Biochemical Metabolism and Inflammatory Cytokines in the Diabetic Rats. Iran J Sci Technol Trans Sci 45, 103–110 (2021). https://doi.org/10.1007/s40995-020-01001-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40995-020-01001-3