Abstract
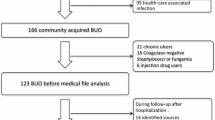
Burkholderia cepacia complex (BCC) is an emerging pathogen of nosocomial infection in chronic or critically ill patients without cystic fibrosis (CF). The objective was to evaluate the management and outcomes of BCC bacteremia in patients without CF. We conducted a retrospective study of non-CF adult patients with BCC bacteremia between January 1997 and December 2016 at 4 tertiary hospitals in South Korea. A total of 216 non-CF patients with BCC bacteremia were identified. Most cases were hospital-acquired (79.2%), and the most common source was a central venous catheter (CVC) (42.1%). The rates of susceptibility to trimethoprim-sulfamethoxazole and piperacillin-tazobactam of BCC isolates were high as 92.8% and 90.3%, respectively. The rates of susceptibility to ceftazidime, meropenem, and levofloxacin were 75.5%, 72.3%, and 64.1%, respectively. The 14-day, 30-day, and in-hospital mortality rate was 19.4%, 23.1%, and 31.0%, respectively. Female (OR = 3.1; 95% CI, 1.4–6.8), liver cirrhosis (OR = 6.2; 95% CI, 1.6–16.6), septic shock (OR = 11.2; 95% CI, 5.1–24.8), and catheter-related infection (OR = 2.6, 95% CI, 1.2–5.8) were the independent risk factors for 30-day mortality. The outcome did not differ according to type of antibiotics used. Among 91 patients with CVC-related BCC bacteremia, delayed CVC removal (> 3 days) had a higher rate of persistent bacteremia (54.5 vs. 26.1%; P = 0.03) and lower rate of clinical response (49.0 vs. 71.9%; P = 0.04), compared with early CVC removal (within 3 days). BCC bacteremia occurring in non-CF patients was mostly hospital-acquired and CVC-related. Early removal of the catheter is crucial in treatment of CVC-related BCC bacteremia.


Similar content being viewed by others
References
Kalish LA, Waltz DA, Dovey M, Potter-Bynoe G, McAdam AJ, Lipuma JJ, Gerard C, Goldmann D (2006) Impact of Burkholderia dolosa on lung function and survival in cystic fibrosis. Am J Respir Crit Care Med 173:421–425
Nasser RM, Rahi AC, Haddad MF, Daoud Z, Irani-Hakime N, Almawi WY (2004) Outbreak of Burkholderia cepacia bacteremia traced to contaminated hospital water used for dilution of an alcohol skin antiseptic. Infect Control Hosp Epidemiol 25:231–239
Lipuma JJ (2005) Update on the Burkholderia cepacia complex. Curr Opin Pulm Med 11:528–533
Liao CH, Chang HT, Lai CC, Huang YT, Hsu MS, Liu CY, Yang CJ, Hsueh PR (2011) Clinical characteristics and outcomes of patients with Burkholderia cepacia bacteremia in an intensive care unit. Diagn Microbiol Infect Dis 70:260–266
Huang CH, Jang TN, Liu CY, Fung CP, Yu KW, Wong WW (2001) Characteristics of patients with Burkholderia cepacia bacteremia. J Microbiol Immunol Infect 34:215–219
Woods CW, Bressler AM, LiPuma JJ, Alexander BD, Clements DA, Weber DJ, Moore CM, Reller LB, Kaye KS (2004) Virulence associated with outbreak-related strains of Burkholderia cepacia complex among a cohort of patients with bacteremia. Clin Infect Dis 38:1243–1250
El Chakhtoura NG, Saade E, Wilson BM, Perez F, Papp-Wallace KM, Bonomo RA (2017) A 17-year nationwide study of Burkholderia cepacia complex bloodstream infections among patients in the United States Veterans Health Administration. Clin Infect Dis 65:1253–1259
von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, Vandenbroucke JP, Initiative S (2007) The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet 370:1453–1457
Clinical and Laboratory Standards Institute (2011) Performance standards for antimicrobial susceptibility testing; 21th Infomational supplment. CLSI document M100-S21. Clinical and Laboratory Standards Institue, Wayne, PA, USA
Friedman ND, Kaye KS, Stout JE, McGarry SA, Trivette SL, Briggs JP, Lamm W, Clark C, MacFarquhar J, Walton AL, Reller LB, Sexton DJ (2002) Health care-associated bloodstream infections in adults: a reason to change the accepted definition of community-acquired infections. Ann Intern Med 137:791–797
Mermel LA, Allon M, Bouza E, Craven DE, Flynn P, O’Grady NP, Raad II, Rijnders BJ, Sherertz RJ, Warren DK (2009) Clinical practice guidelines for the diagnosis and management of intravascular catheter-related infection: 2009 update by the Infectious Diseases Society of America. Clin Infect Dis 49:1–45
Boktour M, Hanna H, Ansari S, Bahna B, Hachem R, Tarrand J, Rolston K, Safdar A, Raad I (2006) Central venous catheter and Stenotrophomonas maltophilia bacteremia in cancer patients. Cancer 106:1967–1973
Ku NS, Han SH, Kim CO, Baek JH, Jeong SJ, Jin SJ, Choi JY, Song YG, Kim JM (2011) Risk factors for mortality in patients with Burkholderia cepacia complex bacteraemia. Scand J Infect Dis 43:792–797
Lee Y, Lee YT, Wang YC, Chen CT, Sun JR, Liu CP, Liu YM, Kuo SC, Chiu CH, Yang YS, Lin JC, Chen TL (2018) Risk of mortality of catheter-related bloodstream infections caused by Acinetobacter species: is early removal of the catheters associated with a better survival outcome? J Intensive Care Med 33:361–369
Friedman ND, Korman TM, Fairley CK, Franklin JC, Spelman DW (2002) Bacteraemia due to Stenotrophomonas maltophilia: an analysis of 45 episodes. J Inf Secur 45:47–53
Lee YM, Moon C, Kim YJ, Lee HJ, Lee MS, Park KH (2018) Clinical impact of delayed catheter removal for patients with central-venous-catheter-related Gram-negative bacteraemia. J Hosp Infect 99:106–113
Caraher E, Reynolds G, Murphy P, McClean S, Callaghan M (2007) Comparison of antibiotic susceptibility of Burkholderia cepacia complex organisms when grown planktonically or as biofilm in vitro. Eur J Clin Microbiol Infect Dis 26:213–216
Mahenthiralingam E, Urban TA, Goldberg JB (2005) The multifarious, multireplicon Burkholderia cepacia complex. Nat Rev Microbiol 3:144–156
Chien YC, Liao CH, Sheng WH, Chien JY, Huang YT, Yu CJ, Hsueh PR (2018) Clinical characteristics of bacteraemia caused by Burkholderia cepacia complex species and antimicrobial susceptibility of the isolates in a medical centre in Taiwan. Int J Antimicrob Agents 51:357–364
Avgeri SG, Matthaiou DK, Dimopoulos G, Grammatikos AP, Falagas ME (2009) Therapeutic options for Burkholderia cepacia infections beyond co-trimoxazole: a systematic review of the clinical evidence. Int J Antimicrob Agents 33:394–404
Yu WL, Wang DY, Lin CW, Tsou MF (1999) Endemic Burkholderia cepacia bacteraemia: clinical features and antimicrobial susceptibilities of isolates. Scand J Infect Dis 31:293–298
Caverly LJ, Spilker T, Kalikin LM, Stillwell T, Young C, Huang DB, LiPuma JJ (2020) In vitro activities of β-lactam-β-lactamase inhibitor antimicrobial agents against cystic fibrosis respiratory pathogens. Antimicrob Agents Chemother 64:e01595–e01519
Funding
This work has supported by a National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (No. NRF-2018R1C1B5043292).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
This study was approved by the Institutional Review Board of Kyung Hee University Hospital (2018-10-038).
Informed consent
Informed consent was not required because of the retrospective nature of the present study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lee, YM., Park, KH., Moon, C. et al. Management and outcomes of Burkholderia cepacia complex bacteremia in patients without cystic fibrosis: a retrospective observational study. Eur J Clin Microbiol Infect Dis 39, 2057–2064 (2020). https://doi.org/10.1007/s10096-020-03960-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-020-03960-2