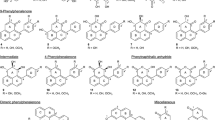
Abstract
Pseudomonas syringae pv. actinidiae (Psa) has caused severe damage to kiwifruit plantations worldwide. It is still crucial to find compounds that are able to control the disease. In this work we developed a time and space-saving leaf disk assay using highly susceptible yellow-fleshed cultivars. The assay enabled 29 molecules/compounds to be screened in relation to their capacity to condition the development of necrosis on leaf disks by treating them before the inoculation. A necrosis index was thereby created. Fosetyl-aluminum, DL-β-Amino-n-butyric acid (BABA) and saccharin (BIT) reduced symptoms significantly, and Fosetyl-aluminum was the most effective. Maxim®, triclopyr and Methyl jasmonate enhanced the necrosis index significantly. S-Methyl 1,2,3-benzothiadiazole-7-carbothioate (BTH) also enhanced symptom severity, though there was no statistical support confirming this. Salicylic acid did not condition symptom development. In vitro investigations into the direct effect of the compounds on Psa highlighted that Fosetyl-aluminum and the polyamines, spermidine and cadaverine, inhibited growth and also had a biocidal effect in a dose-dependent fashion. Salicylic acid and spermine also strongly reduced bacterial growth. Long-term trials with potted plants in the greenhouse confirmed the protective effect of Fosetyl-aluminum and clearly showed that BTH enhances the susceptibility of kiwifruit to Psa. Fosetyl-aluminum significantly decreased symptom expression in kiwifruit plantations (cv. Hayward) subjected to natural Psa infection. Monitoring Psa growth dynamics in the leaf disks by time-point-based Real-Time PCR, showed that treatment with Fosetyl-aluminum restricts bacterial multiplication within the host plant.








Similar content being viewed by others
References
Achary, V. M. M., Ram, B., Manna, M., Datta, D., Bhatt, A., Reddy, M. K., & Agrawal, P. K. (2017). Phosphite: A novel P fertilizer for weed management and pathogen control. Plant Biotechnology Journal, 15, 493–1508. https://doi.org/10.1111/pbi.12803.
Antoniacci, L., Bugiani, R., Rossi, R., Cavazza, F., Franceschelli, F., & Scannavini, M. (2014). Impiego di prodotti di sintesi e naturali nella difesa dal cancro batterico del kiwi (Pseudomonas syringae pv. actinidiae). Atti Giornate Fitopatologiche, 2, 173–180.
Baccelli, I., & Mauch-Mani, B. (2017). When the story proceeds backward: The discovery of endogenous β-aminobutyric acid as the missing link for a potential new plant hormone. Communucative & Integrative Biology, 10(2). https://doi.org/10.1080/19420889.2017.1290019.
Balestra, G. M., Mazzaglia, A., Spinelli, R., Graziani, S., Quattrucci, A., & Rossetti, A. (2008). Cancro batterico su Actinidia chinensis. L’Informatore Agrario, 38, 75–76.
Balmer, A., Glauser, G., Mauch-Mani, B., & Baccelli, I. (2019). Accumulation patterns of endogenous β-aminobutyric acid during plant development and defence in Arabidopsis thaliana. Plant Biology, 21(2), 318–325. https://doi.org/10.1111/plb.12940.
Burra, D. D., Berkowitz, O., Hedley, P. E., Morris, J., Resjo, S., Levander, F., Liljeroth, E., Andreasson, E., & Alexandersson, E. (2014). Phosphite-induced changes of the transcriptome and secretome in Solanum tuberosum leading to resistance against Phytophthora infestans. BMC Plant Biology, 14, 254. https://doi.org/10.1186/s12870-014-0254-y.
Butler, M. I., Stockwell, P. A., Black, M. A., Day, R. C., Lamont, I. L., & Poulter, R. T. M. (2013). Pseudomonas syringae pv. actinidiae from recent outbreaks of kiwifruit bacterial canker belong to different clones that originated in China. PLoS ONE, 8(2), e57464. https://doi.org/10.1371/journal.pone.0057464.
Cameron, A., & Sarojini, V. (2014). Pseudomonas syringae pv. actinidiae: Chemical control, resistance mechanisms and possible alternatives. Plant Pathology, 63, 1–11. https://doi.org/10.1111/ppa.12066.
Cellini, A., Fiorentini, L., Buriani, G., Yu, J., Donati, I., Cornish, D. A., Novak, B., Costa, G., Vanneste, J. L., & Spinelli, F. (2014). Elicitors of the salicylic acid pathway reduce incidence of bacterial canker of kiwifruit caused by Pseudomonas syringae pv. actinidae. Annals of Applied Biology, 165, 441–453. https://doi.org/10.1111/aab.12150.
Chapman, J. R., Taylor, R. K., Weir, B. S., Romberg, M. K., Vanneste, J. L., Luck, J., & Alexander, B. J. R. (2012). Phylogenetic relationships among global populations of Pseudomonas syringae pv. actinidiae. Phytopathology, 102, 1034–1044. https://doi.org/10.1094/PHYTO-03-12-0064-R.
Chen, Z., Agnew, J. L., Cohen, J. D., He, P., Shan, L., Sheen, J., & Kunkel, B. N. (2007). Pseudomonas syringae type III effector AvrRpt2 alters Arabidopsis thaliana auxin physiology. Proceedings of the National Academy of Sciences, 104, 20131–20136. https://doi.org/10.1073/pnas.0704901104.
Colombi, E., Straub, C., Künzel, S., Templeton, M. D., McCann, H. C., & Rainey, P. B. (2017). Evolution of copper resistance in the kiwifruit pathogen Pseudomonas syringae pv. actinidiae through acquisition of integrative conjugative elements and plasmids. Environmental Microbiology, 19(2), 819–832. https://doi.org/10.1111/1462-2920.13662.
Corsi, B., Linthorst, J. M. H., Forni, C., & Riccioni, L. (2017). Enhancement of PR1 and PR5 gene expressions by chitosan treatment in kiwifruit plants inoculated with Pseudomonas syringae pv. actinidiae. European Journal of Plant Pathology, 148, 163–179. https://doi.org/10.1007/s10658-016-1080-x.
Cui, F., Wu, S., Sun, W., Coaker, G., Kunkel, B., He, P., & Shan, L. (2013). The Pseudomonas syringae type III effector AvrRpt2 promotes pathogen virulence via stimulating arabidopsis auxin/indole acetic acid protein turnover. Plant Physiology, 162, 1018–1029. https://doi.org/10.1104/pp.113.219659.
Dalio, R. J. D., Fleischmann, F., Humez, M., & Osswald, W. (2014). Phosphite protects Fagus sylvatica seedlings towards Phytophthora plurivora via local toxicity, priming and facilitation of pathogen recognition. PLoS One, 9(1), e87860. https://doi.org/10.1371/journal.pone.0087860.
De Jong, H., Reglinski, T., Elmer, P. A. G., Wurms, K., Joel, L., Vanneste, J. L., Guo, L. F., & Alavi, M. (2019). Integrated use of Aureobasidium pullulans strain CG163 and Acibenzolar-S-methyl for management of bacterial canker in kiwifruit. Plants, 8(8), 287. https://doi.org/10.3390/plants8080287.
Eshragi, L., Anderson, J., Aryamanesh, N., Shearer, B., Mc Comb, J., Hardy, G. E. S. J., & O'Brien, P. A. (2011). Phosphite primed defence responses and enhanced expression of defence genes in Arabidopsis thaliana infected with Phytophthora cinnamomi. Plant Pathology, 60, 1086–1095. https://doi.org/10.1111/j.1365-3059.2011.02471.x.
Faize, L., & Faize, M. (2018). Functional analogues of salicylic acid and their use in crop protection. Agronomy, 8, 5. https://doi.org/10.3390/agronomy8010005.
Ferrante, P., & Scortichini, M. (2009). Identification of Pseudomonas syringae pv. actinidiae as causal agent of bacterial canker of yellow kiwifruit (Actinidia chinensis Planchon) in Central Italy. Journal of Phytopathology, 157, 768–770. https://doi.org/10.1111/j.1439-0434.2009.01550.x.
Ferrante, P., & Scortichini, M. (2010). Molecular and phenotypic features of Pseudomonas syringae pv. actinidiae isolated during recent epidemics of bacterial canker on yellow kiwifruit (Actinidia chinensis) in Central Italy. Plant Pathology, 59(5), 954–962. https://doi.org/10.1111/j.1365-3059.2010.02304.x.
Ferrante, P., & Scortichini, M. (2015). Redefining the global populations of Pseudomonas syringae pv. actinidiae based on pathogenic, molecular and phenotypic characteristics. Plant Pathology, 64, 51–62. https://doi.org/10.1111/ppa.12236.
Flores, O., Prince, C., Nuñez, M., Vallejos, A., Mardones, C., Yañez, C., Besoain, X., & Bastías, R. (2018). Genetic and phenotypic characterization of indole-producing isolates of Pseudomonas syringae pv. actinidiae obtained from chilean kiwifruit orchards. Frontiers in Microbiology, 9, 1907. https://doi.org/10.3389/fmicb.2018.01907.
Fratarcangeli, L., Rossetti, A., Mazzaglia, A., & Balestra, G. M. (2010). Il ruolo del rame nella lotta al cancro batterico del kiwi. L'Informatore Agrario, 8, 52–56.
Fujikawa, T., & Sawada, H. (2016). Genome analysis of the kiwifruit canker pathogen Pseudomonas syringae pv. actinidiae biovar 5. Scientific Reports, 6, 21399. https://doi.org/10.1038/srep21399.
Fujikawa, T., & Sawada, H. (2019). Genome analysis of Pseudomonas syringae pv. actinidiae biovar 6, which produces the phytotoxins, phaseolotoxin and coronatine. Scientific Reports, 9, 3836. https://doi.org/10.1038/s41598-019-40754-9.
Gallelli, A., L’Aurora, A., & Loreti, S. (2011). Gene sequence analysis for the molecular detection of Pseudomonas syringae pv. actinidiae: Developing diagnostic protocols. Journal of Plant Pathology, 93(2), 425–435. https://doi.org/10.4454/jpp.v93i2.1198.
Gallelli, A., Talocci, S., Pilotti, M., & Loreti, S. (2014). Real-time and qualitative PCR for detecting Pseudomonas syringae pv. actinidiae isolates causing recent outbreaks of kiwifruit bacterial canker. Plant Pathology, 63(2), 264–276. https://doi.org/10.1111/ppa.12082.
Gómez-Merino, F. C., & Trejo-Téllez, L. I. (2015). Biostimulant activity of phosphite in horticulture. Scientia Horticulturae, 196, 82–90. https://doi.org/10.1016/j.scienta.2015.09.035.
Gonzalez-Lamothe, R., El Oirdi, M., Normand Brisson, N., & Kamal, B. K. (2012). The conjugated auxin indole-3-acetic acid–aspartic acid promotes plant disease development. The Plant Cell, 24, 762–777. https://doi.org/10.1105/tpc.111.095190.
Han, H. S., Nam, H. Y., Koh, Y. J., Hur, J. S., & Jung, J. S. (2003). Molecular bases of high-level streptomycin resistance in Pseudomonas marginalis and Pseudomonas syringae pv. actinidiae. The Journal of Microbiology, 41(1), 16–21.
Kazan, K., & Lyons, R. (2014). Intervention of Phytohormone pathways by pathogen effectors. The Plant Cell, 26(6), 2285–2309. https://doi.org/10.1105/tpc.114.125419.
Koh, Y. J., Cha, J. B., Chung, J. H., & Lee, H. D. (1994). Outbreak and spread of bacterial canker in kiwifruit. Korean Journal of Plant Pathology, 10, 68–72.
Kunkel, B. N., & Harper, C. P. (2018). The roles of auxin during interactions between bacterial plant pathogens and their hosts. Journal of Experimental Botany, 69(2), 245–254. https://doi.org/10.1093/jxb/erx447.
Livak, K. J., & Schmittgen, T. D. (2001). Analysis of relative expression data using real-time quantitative PCR and the 2-∆∆CT method. Methods, 25(4), 402–408. https://doi.org/10.1006/meth.2001.1262.
Ludwig-Müller, J. (2015). Bacteria and fungi controlling plant growth by manipulating auxin: Balance between development and defense. Journal of Plant Physiology, 172, 4–12. https://doi.org/10.1016/j.jplph.2014.01.002.
Marcelletti, S., Ferrante, P., Petriccione, M., Firrao, G., & Scortichini, M. (2011). Pseudomonas syringae pv. actinidiae draft genomes comparison reveal strain-specific features involved in adaptation and virulence to Actinidia species. PLoS ONE, 6(11), e27297. https://doi.org/10.1371/journal.pone.0027297.
Massoud, K., Barchietto, T., Le Rudulier, T., Pallandre, L., Didierlaurent, L., Garmier, M., Ambard-Bretteville, F., Seng, J. M., & Saindrenan, P. (2012). Dissecting phosphite-induced priming in arabidopsis infected with Hyaloperonospora arabidopsidis. Plant Physiology, 159, 286–298. https://doi.org/10.1104/pp.112.194647.
Mazzaglia, A., Studholme, D. J., Taratufolo, M. C., Cai, R., Almeida, N. F., Goodman, T., Guttman, D. S., Vinatzer, B. A., & Balestra, G. M. (2012). Pseudomonas syringae pv. actinidiae (PSA) isolates from recent bacterial canker of kiwifruit outbreaks belong to the same genetic lineage. PLoS ONE, 7(5), e36518. https://doi.org/10.1371/journal.pone.0036518.
McCann, H. C., Rikkerink, E. H. A., Bertels, F., Fiers, M., Lu, A., Rees-George, J., Andersen, M. T., Gleave, A. P., Haubold, B., Wohlers, M. W., Guttman, D. S., Wang, P. W., Straub, C., Vanneste, J., Rainey, P. B., & Templeton, M. D. (2013). Genomic analysis of the kiwifruit pathogen Pseudomonas syringae pv actinidiae provides insight into the origins of an emergent plant disease. PLoS Pathogens, 9(7), e1003503. https://doi.org/10.1371/journal.ppat.1003503.
McCann, H. C., Li, L., Liu, Y., Li, D., Pan, H., Zhong, C., Rikkerink, E. H. A., Templeton, M. D., Straub, C., Colombi, E., Rainey, P. B., & Huang, H. (2017). Origin and evolution of the kiwifruit canker pandemic. Genome Biology and Evolution, 9(4), 932–944. https://doi.org/10.1093/gbe/evx055.
McDonald, A. E., Grant, B. R., & Plaxton, W. C. (2001). Phosphite (phosphorous acid): Its relevance in the environment and agriculture and influence on plant phosphate starvation response. Journal of Plant Nutrition, 24(10), 1505–1519. https://doi.org/10.1081/PLN-100106017.
McKinney, H. H. (1923). Influence of soil temperature and moisture on infection of wheat seedlings by Helminthosporium sativum. Journal of Agricultural Research, 26, 195–225.
Michelotti, V., Lamontanara, A., Buriani, G., Orrù, L., Cellini, A., Donati, I., Vanneste, J. L., Cattivelli, L., Tacconi, G., & Spinelli, F. (2018). Comparative transcriptome analysis of the interaction between Actinidia chinensis var. chinensis and Pseudomonas syringae pv. actinidiae in absence and presence of acibenzolar-S-methyl. BMC Genomics, 19(1), 585. https://doi.org/10.1186/s12864-018-4967-4.
Monchiero, M., Gullino, M. L., Pugliese, M., Spadaro, D., & Garibaldi, A. (2015). Efficacy of different chemical and biological products in the control of Pseudomonas syringae pv. actinidiae on kiwifruit. Australasian Plant Pathology, 44, 13–23. https://doi.org/10.1007/s13313-014-0328-1.
Navarro, L., Dunoyer, P., Jay, F., Arnold, B., Dharmasiri, N., Estelle, M., Voinnet, O., & Jones, J. D. G. (2006). A plant miRNA contributes to antibacterial resistance by repressing Auxin signaling. Science, 312(5772), 436–439. https://doi.org/10.1126/science.1126088.
Naseem, M., Srivastava, M., Tehseen, M., & Ahmed, N. (2015). Auxin crosstalk to plant immune networks: A plant-pathogen interaction perspective. Current Protein and Peptide Science, 16(5), 389–394. https://doi.org/10.2174/1389203716666150330124911.
OEPP/EPPO. (2014). PM 7/120 (1) Pseudomonas syringae pv. actinidiae. OEPP/EPPO Bulletin, 44(3), 360–375. https://doi.org/10.1111/epp.12171.
Petriccione, M., Mastrobuoni, F., Zampella, L., & Scortichini, M. (2015). Reference gene selection for normalization of RT-qPCR gene expression data from Actinidia deliciosa leaves infected with Pseudomonas syringae pv. actinidiae. Scientific Reports, 5, 16961. https://doi.org/10.1038/srep16961.
Pizzinat, A., Giordani, L., Asteggiano, L., Nari, L., Giraudo, M., Pavarino, A., Bevilacqua, A., Spinelli, F., Morone, C., & Vittone, G. (2014). Contenimento della batteriosi dell’actinidia in Piemonte. Atti Giornate Fitopatologiche, 2, 163–172.
Prencipe, S., Gullino, M. L., & Spadaro, D. (2018). Pseudomonas syringae pv. actinidiae isolated from Actinidia chinensis Var. deliciosa in northern Italy: Genetic diversity and virulence. European Journal of Plant Pathology, 150, 191–204. https://doi.org/10.1007/s10658-017-1267-9.
Pucci, N., Orzali, L., Modesti, V., Lumia, V., Brunetti, A., Pilotti, M., & Loreti, S. (2018). Essential oils with inhibitory capacities on Pseudomonas syringae pv. actinidiae, the causal agent of kiwifruit bacterial canker. Asian Journal of Plant Pathology, 12(1), 16–26. https://doi.org/10.3923/ajppaj.2018.16.26.
Quattrucci, A., Renzi, M., Rossetti, A., Ricci, L., Taratufolo, C., Mazzaglia, A., & Balestra, G. M. (2010). Cancro batterico del kiwi verde: nuove strategie di controllo. L'Informatore Agrario, 16, 53–58.
Robert-Seilaniantz, A., Grant, M., & Jones, J. D. G. (2011). Hormone crosstalk in plant disease and defense: More than just JASMONATE-SALICYLATE antagonism. Annual Review of Phytopathology, 49, 317–343. https://doi.org/10.1146/annurev-phyto-073009-114447.
Scortichini, M. (1994). Occurrence of Pseudomonas syringae pv. actinidiae in Italy. Plant Pathology, 43, 1035–1038. https://doi.org/10.1111/j.1365-3059.1994.tb01654.x.
Scortichini, M., Marcelletti, S., Ferrante, P., Petriccione, M., & Firrao, G. (2012). Pseudomonas syringae pv. actinidiae: A reemerging, multi-faceted, pandemic pathogen. Molecular Plant Pathology, 13(7), 631–640. https://doi.org/10.1111/j.1364-3703.2012.00788.x.
Scortichini, M. (2014). Field efficacy of chitosan to control Pseudomonas syringae pv. actinidiae, the causal agent of kiwifruit bacterial canker. European Journal of Plant Pathology, 140, 887–892. https://doi.org/10.1007/s10658-014-0515-5.
Simonetti, G., Pucci, N., Brasili, E., Valletta, A., Sammarco, I., Carnevale, E., Pasqua, G., & Loreti, S. (2019). In vitro antimicrobial activity of plant extracts against Pseudomonas syringae pv. actinidiae causal agent of bacterial canker in kiwifruit. Plant Biosystems, 154(1), 100–106. https://doi.org/10.1080/11263504.2019.1699194.
Takikawa, Y., Serizawa, S., Ichikawa, T., Tsuyumu, S., & Goto, M. (1989). Pseudomonas syringae pv. actinidiae pv. Nov.: The causal bacterium of canker of kiwifruit in Japan. Japan Journal of Phytopathology, 55(4), 437–444. https://doi.org/10.3186/jjphytopath.55.437.
Tosi, L., Tacconi, G., Spinelli, F., Posenato, G., Bertaiola, F., & Giacopini, A. (2014). Efficacia di alcuni formulati nei confronti del cancro batterico dell’actinidia causato da Pseudomonas syringae pv. actinidiae. Atti Giornate Fitopatologiche, 2, 157–162.
Valente, M., Ortugno, C., Tosi, L., Scannavini, M., Pelliconi, F., Fagioli, L., Scortichini, M., Vittone, G., Fiorillo, E., Pradolesi, G., & Donati, G. (2014). Bion®50WG (acibenzolar-S-methyl), induttore delle autodifese della pianta: efficacia nella prevenzione di Pseudomonas syringae pv. actinidiae su Actinidia. Atti Giornate Fitopatologiche, 2, 147–156.
Vanneste, J. L. (2017). The scientific, economic, and social impacts of the New Zealand outbreak of bacterial canker of kiwifruit (Pseudomonas syringae pv. actinidiae). Annual Review of Phytopathology, 55, 377–399. https://doi.org/10.1146/annurev-phyto-080516-035530.
Vavala, E., Passariello, C., Pepi, F., Colone, M., Garzoli, S., Ragno, R., Pirolli, A., Stringaro, A., & Angiolella, L. (2016). Antibacterial activity of essential oils mixture against PSA. Natural Product Research, 30, 412–418. https://doi.org/10.1080/14786419.2015.1022543.
Wasternack, C., & Miroslav Strnad, M. (2016). Jasmonate signaling in plant stress responses and development – Active and inactive compounds. New Biotechnology, 33(5), 604–613. https://doi.org/10.1016/j.nbt.2015.11.001.
Zhang, Y., & Li, X. (2019). Salicylic acid: Biosynthesis, perception, and contributions to plant immunity. Current Opinion in Plant Biology, 50, 29–36. https://doi.org/10.1016/j.pbi.2019.02.004.
Acknowledgements
This research was supported by: i) a Mipaaf (the Italian Ministry of Agricultural and Forestry Policies) project, “INTERACT/ARDICA, Interventi di coordinamento ed implementazione alle azioni di ricerca, lotta e difesa al cancro batterico dell’Actinidia”; ii) a Latium Region PSR (Programma di Sviluppo Rurale) project, “PRO.ACTI.BA.RE, Protezione dell’actinidia dal cancro batterico (Pseudomonas syringae pv. actinidiae) mediante l’uso di induttori di resistenza”.
Availability of data and material (data transparency)
Raw data can be viewed upon request.
Code availability (software application or custom code)
Not applicable.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest/competing interests
The authors have no conflict of interest or competing interests to declare.
Ethics approval
All principles of ethical and professional conduct have been followed during this research and the drafting of this manuscript.
Consent to participate
All the authors gave consent to participate in the authorship.
Consent for publication
All the authors agreed with the content of the article and gave explicit consent to submit and publish the article.
Electronic supplementary material
Online Resource 1
Details on the preparation of compounds to be tested for the anti-bacterial activity and for their effect on the infection of Pseudomonas syringae pv. actinidiae (Psa) in the leaf disk assays (PDF 110 kb)
Online Resource 2
Symptom scoring for the inference of the disease severity index of the greenhouse trials (McKinney 1923) and data on the outcome of the trials. A. Classes of symptoms observed on potted plants treated with Fosetyl-aluminum, BTH and water and inoculated with Pseudomonas syringae pv. actinidiae (Psa), and the attributed scores. The score derived from the combination of different classes was obtained from the sum of the values of each class. Class combinations including F are valued as F. B. Score assigned to each plant within each treatment and trial (A, B1 and B2) based on symptom class ranking. (PDF 68 kb)
Online Resource 3
Optical density (OD) of Pseudomonas syringae pv. actinidiae (Psa) after growing in NSB supplemented with compounds/molecules, which were also used in the leaf disk assay. All measurements were made with a spectrophotometer at 660 nm after 20–22 h incubation, when non- compound-supplemented Psa cultures (called Control 1, C1) reached approximately the value of 1.0. A: OD measurements for each compound/molecule at different concentration. The comparison is with C1. Control 2 (C2) is the absorbance of non-inoculated NSB. Different letters indicate significant differences between groups (p < 0.01). The vertical bars indicate standard deviations of the means. Asterisks indicate the bactericidal concentrations. B: compounds/molecules and concentrations that caused a growth reduction ≥40% compared with C1. Differences are expressed as growth percentage compared with C1 (= 100%) and as percentage growth reduction (PDF 161 kb)
Online Resource 4
Example of non-inoculated leaf disks used as the control in order to assess any wound, time and compound-related senescence/phytotoxicity. The figure includes the non-treated leaf disks (NT) and leaf disks treated twice with BTH, following the protocol assay. Ten leaf disks were treated with BTH suspensions at different concentrations, and the experiment was repeated three times. We show close-ups of five disks for each concentration. The image was taken 20 days after BTH treatments. Belen was used as the source of the leaf disks (PDF 268 kb)
Online Resource 5
Confirmation of the outcome of the leaf disk assay on Actinidia chinensis var. deliciosa cv. Hayward: significant decrease in the necrosis index of leaf disks treated with Fosetyl-aluminum and BABA and subsequently inoculated with Pseudomonas syringae pv. actinidiae (Psa). The comparison is with the control non-treated and inoculated with Psa (NT-Psa). Necrosis indexes marked by different letters are significantly different from each other [p < 0.01 (Fosetyl-aluminum), p < 0.05 (BABA)]. The numbers indicate the independent statistical analyses conducted separately on data from two temporally-distinct recordings: the first in grey, the second black. The vertical bars indicate the standard error of the means (PDF 34 kb)
Online Resource 6
The IAE response (Increase and Anticipation of symptom Expression) obtained by treating the leaf disks with triclopyr and MeJA at various concentrations and then inoculating with Pseudomonas syringae pv. actinidiae (Psa). NT-Psa = non-treated and inoculated with Psa; NT = non-treated and non-inoculated (PDF 318 kb)
Online Resource 7
Examples of compounds/molecules whose treatment on leaf disks (cv. Belen) did not influence the outcome of Pseudomonas syringae pv. actinidiae (Psa) inoculation in terms of the necrosis index. The comparison is with the control non-treated and inoculated with Psa (NT-Psa). Necrosis index values marked by different letters are significantly different from each other (p < 0.05). The numbers indicate the independent statistical analyses conducted separately on data from two temporally-distinct recordings: the first in grey, the second black. The vertical bars indicate the standard error of the means (PDF 37.4 kb)
Online Resource 8
Compounds/molecules whose treatment on leaf disk (cv. Belen) did not statistically differ from the control non-treated and inoculated with Pseudomonas syringae pv. actinidiae (Psa) (NT-Psa) in terms of necrosis index, similarly to those depicted in Online resource 7. Unlike the effects reported in Online resource 7, the mean values of the necrosis index in this figure were repeatedly and evidently lower or higher than the NT-Psa in all the experiments. This prompts a hypothesis on a fine tuning by these compounds in the direction of resistance/susceptibility induction. Necrosis indexes marked by the different letters are significantly different from each other (p < 0.05). The numbers indicate the independent statistical analyses conducted separately on data from two temporally-distinct recordings: the first in grey, the second black. The vertical bars indicate the standard error of the means. Concentrations of BTH expressed in mM and g/l refer to the active principle and the commercial product (Bion) respectively (PDF 34.3 kb)
Online Resource 9
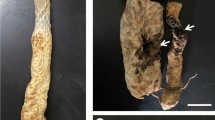
Healthy-looking Fosetyl-aluminum-treated/Psa-inoculated plants (a) compared with BTH-treated/Psa-inoculated plants (b), which were dead or severely stunted (B2 trial, during spring/summer 2016) (PDF 175 kb)
Online Resource 10
Rainfall (gray line) and average (green), maximum (red) and minimum (azure) temperature during the field trial. The trial was conducted from 9 April to 8 July (PDF 91 kb)
Rights and permissions
About this article
Cite this article
Brunetti, A., Pucci, N., Modesti, V. et al. In vitro and in planta screening of compounds for the control of Pseudomonas syringae pv. actinidiae in Actinidia chinensis var. chinensis. Eur J Plant Pathol 158, 829–848 (2020). https://doi.org/10.1007/s10658-020-02119-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-020-02119-1