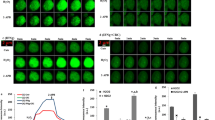
Abstract
Microglia as the primary immune cells of brain act protective effects against injuries and infections in the central nervous system. Inflammation via excessive Ca2+ influx and oxygen radical species (ROS) generation is a known factor in many neurodegenerative disorders. Importantly, the Ca2+ permeable TRPM2 channel is activated by oxidative stress. Thus, TRPM2 could provide the excessive Ca2+ influx in the microglia. Although TRPM2 expression level is high in inflammatory cells, the interplay between mouse microglia and TRPM2 channel during inflammation is not fully identified. Thus, it is important to understand the mechanisms and factors involved in order to enhance neuronal regeneration and repair. The data presented here indicate that TRPM2 channels were activated in microglia cells by interferon-gamma (IFNγ). The IFNγ treatment further increased apoptosis (early and late) and cytokine productions (TNF-α, IL-1β, and IL-6) which were due to increased lipid peroxidation and ROS generations as well as increased activations of caspase −3 (Casp-3) and − 9 (Casp-9). However, selenium treatment diminished activations of TRPM2, cytokine, Casp-3, and Casp-9, and levels of lipid peroxidation and mitochondrial ROS production in the microglia that were treated with IFNγ. Moreover, addition of either PARP1 inhibitors (PJ34 or DPQ) or TRPM2 blockers (2-APB or ACA) potentiated the modulator effects of selenium. These results clearly suggest that IFNγ leads to TRPM2 activation in microglia cells; whereas, selenium prevents IFNγ-mediated TRPM2 activation and cytokine generation. Together the interplay between IFNγ released from microglia cells is importance in brain inflammation and may affect oxidative cytotoxicity in the microglia.

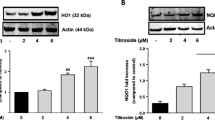
Summary of pathways involved in IFNγ-induced TRPM2 activation and microglia death through excessive reactive oxygen species (ROS): Modulator role of selenium (Se). The IFNγ causes the microglia activation. Nudix box domain of TRPM2 is sensitive to ROS. The ROS induces DNA damage and ADPR-ribose (ADPR) production in the nucleus via PARP1 enzyme activation. ADPR and ROS-induced TRPM2 activation stimulates excessive Ca2+ influx. ROS are produced in the mitochondria through the increase of free cytosolic Ca2+ (via TRPM2 activation) by the IFNγ treatment, although they are diminished by the TRPM2 channel blocker (ACA and 2-APB) and PARP1 inhibitor treatments. The main mechanism in the cell death and inflammatory effects of IFNγ is mediated by stimulation of ROS-mediated caspase (caspase −3 and − 9) activations and cytokine production (TNF-α, IL-1β, and IL-6) via TRPM2 activation, respectively. The apoptotic, inflammatory, and oxidant actions of IFNγ are modulated through TRPM2 inhibition by the Se treatment








Similar content being viewed by others
References
Aminzadeh M, Roghani M, Sarfallah A, Riazi GH (2018) TRPM2 dependence of ROS-induced NLRP3 activation in Alzheimer's disease. Int Immunopharmacol 54:78–85. https://doi.org/10.1016/j.intimp.2017.10.024
Arioz BI, Tastan B, Tarakcioglu E, Tufekci KU, Olcum M, Ersoy N, Bagriyanik A, Genc K, Genc S (2019) Melatonin attenuates LPS-induced acute depressive-like behaviors and microglial NLRP3 inflammasome activation through the SIRT1/Nrf2 pathway. Front Immunol 10:1511. https://doi.org/10.3389/fimmu.2019.01511
Ataizi ZS, Ertilav K, Nazıroğlu M (2019) Mitochondrial oxidative stress-induced brain and hippocampus apoptosis decrease through modulation of caspase activity, Ca(2+) influx and inflammatory cytokine molecular pathways in the docetaxel-treated mice by melatonin and selenium treatments. Metab Brain Dis 34(4):1077–1089. https://doi.org/10.1007/s11011-019-00428-x
Cardozo AK, Ortis F, Storling J, Feng YM, Rasschaert J, Tonnesen M, van Eylen F, Mandrup-Poulsen T, Herchuelz A, Eizirik DL (2005) Cytokines downregulate the sarcoendoplasmic reticulum pump Ca2+ ATPase 2b and deplete endoplasmic reticulum Ca2+, leading to induction of endoplasmic reticulum stress in pancreatic beta-cells. Diabetes 54(2):452–461. https://doi.org/10.2337/diabetes.54.2.452
Cotgreave IA, Duddy SK, Kass GE, Thompson D, Moldéus P (1989) Studies on the anti-inflammatory activity of ebselen. Ebselen interferes with granulocyte oxidative burst by dual inhibition of NADPH oxidase and protein kinase C? Biochem Pharmacol 38(4):649–656. https://doi.org/10.1016/0006-2952(89)90211-6
Demirci S, Kutluhan S, Nazıroğlu M, Uğuz AC, Yürekli VA, Demirci K (2013) Effects of selenium and topiramate on cytosolic Ca(2+) influx and oxidative stress in neuronal PC12 cells. Neurochem Res 38(1):90–97. https://doi.org/10.1007/s11064-012-0893-z
Ebner F, Brandt C, Thiele P, Richter D, Schliesser U, Siffrin V, Schueler J, Stubbe T, Ellinghaus A, Meisel C, Sawitzki B, Nitsch R (2013) Microglial activation milieu controls regulatory T cell responses. J Immunol 191(11):5594–5602. https://doi.org/10.4049/jimmunol.1203331
Egger F, Jakab M, Fuchs J, Oberascher K, Brachtl G, Ritter M, Kerschbaum HH, Gaisberger M. (2020) Effect of glycine on BV-2 microglial cells treated with interferon-γ and lipopolysaccharide. Int J Mol Sci. 26;21(3). pii: E804. https://doi.org/10.3390/ijms21030804
Ertilav K (2019) Pregabalin protected cisplatin-induced oxidative neurotoxicity in neuronal cell line. J Cell Neurosci Oxid Stress 11(1):815–824. https://doi.org/10.37212/jcnos.653500
Ertilav K, Nazıroğlu M, Ataizi ZS, Braidy N (2019) Selenium enhances the apoptotic efficacy of docetaxel through activation of TRPM2 channel in DBTRG glioblastoma cells. Neurotox Res 35(4):797–808. https://doi.org/10.1007/s12640-019-0009-5
Espino J, Bejarano I, Redondo PC, Rosado JA, Barriga C, Reiter RJ, Pariente JA, Rodríguez AB (2010) Melatonin reduces apoptosis induced by calcium signaling in human leukocytes: evidence for the involvement of mitochondria and Bax activation. J Membr Biol 233(1–3):105–118. https://doi.org/10.1007/s00232-010-9230-0
Espino J, Bejarano I, Paredes SD, Barriga C, Rodríguez AB, Pariente JA (2011a) Protective effect of melatonin against human leukocyte apoptosis induced by intracellular calcium overload: relation with its antioxidant actions. J Pineal Res 51(2):195–206. https://doi.org/10.1111/j.1600-079X.2011.00876.x
Espino J, Bejarano I, Paredes SD, Barriga C, Reiter RJ, Pariente JA, Rodríguez AB (2011b) Melatonin is able to delay endoplasmic reticulum stress-induced apoptosis in leukocytes from elderly humans. Age (Dordr) 33(4):497–507. https://doi.org/10.1007/s11357-010-9194-0
Garcia JA, Cardona SM, Cardona AE (2014) Isolation and analysis of mouse microglial cells. Curr Protoc Immunol 104:14.35.1–14.35.15. https://doi.org/10.1002/0471142735.im1435s104
González D, Espino J, Bejarano I, López JJ, Rodríguez AB, Pariente JA (2010) Caspase-3 and -9 are activated in human myeloid HL-60 cells by calcium signal. Mol Cell Biochem 333(1–2):151–157. https://doi.org/10.1007/s11010-009-0215-1
Graeber MB (2010) Changing face of microglia. Science. 330(6005):783–788. https://doi.org/10.1126/science.1190929
Grieco FA, Schiavo AA, Brozzi F, Juan-Mateu J, Bugliani M, Marchetti P, Eizirik DL (2019) The miRNAs miR-211-5p and miR-204-5p modulate ER stress in human beta cells. J Mol Endocrinol 63(2):139–149. https://doi.org/10.1530/JME-19-0066
Guadagno J, Swan P, Shaikh R, Cregan SP (2015) Microglia-derived IL-1β triggers p53-mediated cell cycle arrest and apoptosis in neural precursor cells. Cell Death Dis 6:e1779. https://doi.org/10.1038/cddis.2015.151
Hara Y, Wakamori M, Ishii M, Maeno E, Nishida M, Yoshida T, Yamada H, Shimizu S, Mori E, Kudoh J, Shimizu N, Kurose H, Okada Y, Imoto K, Mori Y (2002) LTRPC2 Ca2+−permeable channel activated by changes in redox status confers susceptibility to cell death. Mol Cell 9(1):163–173. https://doi.org/10.1016/S1097-2765(01)00438-5
Heiner I, Eisfeld J, Lückhoff A (2003) Role and regulation of TRP channels in neutrophil granulocytes. Cell Calcium 33(5–6):533–540. https://doi.org/10.1016/S0143-4160(03)00058-7
Joshi DC, Bakowska JC (2011) Determination of mitochondrial membrane potential and reactive oxygen species in live rat cortical neurons. J Vis Exp 51:pii: 2704. https://doi.org/10.3791/2704
Kahya MC, Nazıroğlu M, Övey İS (2017) Modulation of diabetes-induced oxidative stress, apoptosis, and Ca(2+) entry through TRPM2 and TRPV1 channels in dorsal root ganglion and hippocampus of diabetic rats by melatonin and selenium. Mol Neurobiol 54(3):2345–2360. https://doi.org/10.1007/s12035-016-9727-3
Kakae M, Miyanohara J, Morishima M, Nagayasu K, Mori Y, Shirakawa H, Kaneko S (2019) Pathophysiological role of TRPM2 in age-related cognitive impairment in mice. Neuroscience. 408:204–213. https://doi.org/10.1016/j.neuroscience.2019.04.012
Kato T, Mizoguchi Y, Monji A, Horikawa H, Suzuki SO, Seki Y, Iwaki T, Hashioka S, Kanba S (2008) Inhibitory effects of aripiprazole on interferon-gamma-induced microglial activation via intracellular Ca2+ regulation in vitro. J Neurochem 106(2):815–825. https://doi.org/10.1111/j.1471-4159.2008.05435.x
Keil VC, Funke F, Zeug A, Schild D, Müller M (2011) Ratiometric high-resolution imaging of JC-1 fluorescence reveals the subcellular heterogeneity of astrocytic mitochondria. Pflugers Arch 462:693–708. https://doi.org/10.1007/s00424-011-1012-8
Kierdorf K, Prinz M (2013) Factors regulating microglia activation. Front Cell Neurosci 7:44. https://doi.org/10.3389/fncel.2013.00044
Kraft R, Grimm C, Frenzel H, Harteneck C (2006) Inhibition of TRPM2 cation channels by N-(p-amylcinnamoyl)anthranilic acid. Br J Pharmacol 148(3):264–273
Lennicke C, Rahn J, Bukur J, Hochgräfe F, Wessjohann LA, Lichtenfels R, Seliger B (2016) Modulation of MHC class I surface expression in B16F10 melanoma cells by methylseleninic acid. Oncoimmunology 6(6):e1259049
Malko P, Syed Mortadza SA, McWilliam J, Jiang LH (2019) TRPM2 channel in microglia as a new player in neuroinflammation associated with a spectrum of central nervous system pathologies. Front Pharmacol 10:239. https://doi.org/10.3389/fphar.2019.00239
McHugh D, Flemming R, Xu SZ, Perraud AL, Beech DJ (2003) Critical intracellular Ca2+ dependence of transient receptor potential melastatin 2 (TRPM2) cation channel activation. J Biol Chem 278(13):11002–11006
Meng XL, Chen CL, Liu YY, Su SJ, Gou JM, Huan FN, Wang D, Liu HS, Ben SB, Lu J (2019) Selenoprotein SELENOK enhances the migration and phagocytosis of microglial cells by increasing the cytosolic free Ca(2+) level resulted from the up-regulation of IP(3)R. Neuroscience 406:38–49. https://doi.org/10.1016/j.neuroscience.2019.02.029
Milenkovic VM, Slim D, Bader S, Koch V, Heinl ES, Alvarez-Carbonell D et al (2019) CRISPR-Cas9 mediated TSPO gene knockout alters respiration and cellular metabolism in human primary microglia cells. Int J Mol Sci 20(13):pii: E3359. https://doi.org/10.3390/ijms20133359
Miyake T, Shirakawa H, Kusano A, Sakimoto S, Konno M, Nakagawa T, Mori Y, Kaneko S (2014) TRPM2 contributes to LPS/IFNγ-induced production of nitric oxide via the p38/JNK pathway in microglia. Biochem Biophys Res Commun 444(2):212–217. https://doi.org/10.1016/j.bbrc.2014.01.022
Mizuno T, Zhang G, Takeuchi H, Kawanokuchi J, Wang J, Sonobe Y, Jin S, Takada N, Komatsu Y, Suzumura A (2008) Interferon-gamma directly induces neurotoxicity through a neuron specific, calcium-permeable complex of IFN-gamma receptor and AMPA GluR1 receptor. FASEB J 22(6):1797–1806. https://doi.org/10.1096/fj.07-099499
Mortadza SS, Sim JA, Stacey M, Jiang LH (2017) Signalling mechanisms mediating Zn(2+)-induced TRPM2 channel activation and cell death in microglial cells. Sci Rep 7:45032. https://doi.org/10.1038/srep45032
Nam KN, Koketsu M, Lee EH (2008) 5-Chloroacetyl-2-amino-1,3-selenazoles attenuate microglial inflammatory responses through NF-kappaB inhibition. Eur J Pharmacol 589(1–3):53–57. https://doi.org/10.1016/j.ejphar.2008.03.034
Nazıroğlu M (2007) New molecular mechanisms on the activation of TRPM2 channels by oxidative stress and ADP-ribose. Neurochem Res 32(11):1990–2001. https://doi.org/10.1007/s11064-007-9386-x
Nazıroğlu M (2009) Role of selenium on calcium signaling and oxidative stress-induced molecular pathways in epilepsy. Neurochem Res 34(12):2181–2191. https://doi.org/10.1007/s11064-009-0015-8
Nazıroğlu M (2012) Molecular role of catalase on oxidative stress-induced Ca(2+) signaling and TRP cation channel activation in nervous system. J Recept Signal Transduct Res 32(3):134–141. https://doi.org/10.3109/10799893.2012.672994
Nazıroğlu M (2017) Activation of TRPM2 and TRPV1 channels in dorsal root ganglion by NADPH oxidase and protein kinase c molecular pathways: a patch clamp study. J Mol Neurosci 61(3):425–435. https://doi.org/10.1007/s12031-017-0882-4
Nazıroğlu M, Lückhoff A (2008) Effects of antioxidants on calcium influx through TRPM2 channels in transfected cells activated by hydrogen peroxide. J Neurol Sci 270(1–2):152–158. https://doi.org/10.1016/j.jns.2008.03.003
Nazıroğlu M, Özgül C, Küçükayaz M, Çiğ B, Hebeisen S, Bal R (2013) Selenium modulates oxidative stress-induced TRPM2 cation channel currents in transfected Chinese hamster ovary cells. Basic Clin Pharmacol Toxicol 112(2):96–102. https://doi.org/10.1111/j.1742-7843.2012.00934.x
Nazıroğlu M, Kutluhan S, Ovey IS, Aykur M, Yurekli VA (2014) Modulation of oxidative stress, apoptosis, and calcium entry in leukocytes of patients with multiple sclerosis by Hypericum perforatum. Nutr Neurosci 17(5):214–221. https://doi.org/10.1179/1476830513Y.0000000083
Nazıroğlu M, Öz A, Yıldızhan K (2020) Selenium and neurological diseases: focus on peripheral pain and TRP channels. Curr Neuropharmacol 18(6):501–517. https://doi.org/10.2174/1570159X18666200106152631
Neumann H (2001) Control of glial immune function by neurons. Glia. 36(2):191–199. https://doi.org/10.1002/glia.1108
Ogawa N, Kurokawa T, Mori Y (2016) Sensing of redox status by TRP channels. Cell Calcium 60(2):115–122. https://doi.org/10.1016/j.ceca.2016.02.009
Ohsawa K, Imai Y, Sasaki Y, Kohsaka S (2004) Microglia/macrophage-specific protein Iba1 binds to fimbrin and enhances its actin-bundling activity. J Neurochem 88(4):844–856. https://doi.org/10.1046/j.1471-4159.2003.02213.x
Özkaya D, Nazıroğlu M (2020) Curcumin diminishes cisplatin-induced apoptosis and mitochondrial oxidative stress through inhibition of TRPM2 channel signaling pathway in mouse optic nerve. J Recept Signal Transduct Res 40(2):97–108. https://doi.org/10.1080/10799893.2020.1720240
Pariente R, Pariente JA, Rodríguez AB, Espino J (2016) Melatonin sensitizes human cervical cancer HeLa cells to cisplatin-induced cytotoxicity and apoptosis: effects on oxidative stress and DNA fragmentation. J Pineal Res 60(1):55–64. https://doi.org/10.1111/jpi.12288
Peng Z, Luchtman DW, Wang X, Zhang Y, Song C (2019) Activation of microglia synergistically enhances neurodegeneration caused by MPP+ in human SH-SY5Y cells. Eur J Pharmacol 850:64–74. https://doi.org/10.1016/j.ejphar.2019.01.024
Ray SK, Wilford GG, Crosby CV, Hogan EL, Banik NL (1999) Diverse stimuli induce calpain overexpression and apoptosis in C6 glioma cells. Brain Res 829(1–2):18–27. https://doi.org/10.1016/S0006-8993(99)01290-1
Roychowdhury S, Wolf G, Keilhoff G, Horn TF (2003) Cytosolic and mitochondrial glutathione in microglial cells are differentially affected by oxidative/nitrosative stress. Nitric Oxide 8(1):39–47. https://doi.org/10.1016/S1089-8603(02)00146-5
Ruiz A, Matute C, Alberdi E (2010) Intracellular Ca2+ release through ryanodine receptors contributes to AMPA receptor-mediated mitochondrial dysfunction and ER stress in oligodendrocytes. Cell Death Dis 1:e54. https://doi.org/10.1038/cddis.2010.31
Scalia M, Satriano C, Greca R, Stella AM, Rizzarelli E, Spina-Purrello V (2013) PARP-1 inhibitors DPQ and PJ-34 negatively modulate proinflammatory commitment of human glioblastoma cells. Neurochem Res 38(1):50–58. https://doi.org/10.1007/s11064-012-0887-x
Socodato R, Portugal CC, Rodrigues A, Henriques J, Rodrigues C, Figueira C, Relvas JB (2018) Redox tuning of Ca(2+) signaling in microglia drives glutamate release during hypoxia. Free Radic Biol Med 118:137–149. https://doi.org/10.1016/j.freeradbiomed.2018.02.036
Sun Y, Sukumaran P, Selvaraj S, Cilz NI, Schaar A, Lei S, Singh BB (2018) TRPM2 promotes neurotoxin MPP(+)/MPTP-induced cell death. Mol Neurobiol 55(1):409–420. https://doi.org/10.1007/s12035-016-0338-9
Tian LP, Zhang S, Xu L, Li W, Wang Y, Chen SD, Ding JQ (2012) Selenite benefits embryonic stem cells therapy in Parkinson's disease. Curr Mol Med 12(8):1005–1014. https://doi.org/10.2174/156652412802480880
Togashi K, Inada H, Tominaga M (2008) Inhibition of the transient receptor potential cation channel TRPM2 by 2-aminoethoxydiphenyl borate (2-APB). Br J Pharmacol 153(6):1324–1330. https://doi.org/10.1038/sj.bjp.0707675
Uguz AC, Cig B, Espino J, Bejarano I, Naziroglu M, Rodríguez AB, Pariente JA (2012) (2012). Melatonin potentiates chemotherapy-induced cytotoxicity and apoptosis in rat pancreatic tumor cells. J Pineal Res 53(1):91–98. https://doi.org/10.1111/j.1600-079X.2012.00974.x
Xie RX, Li DW, Liu XC, Yang MF, Fang J, Sun BL, Zhang ZY, Yang XY (2017) Carnosine attenuates brain oxidative stress and apoptosis after Intracerebral hemorrhage in rats. Neurochem Res 42(2):541–551. https://doi.org/10.1007/s11064-016-2104-9
Xu Z, Jin X, Pan T, Liu T, Wan N, Li S (2017) Antagonistic effects of selenium on cadmium-induced apoptosis by restoring the mitochondrial dynamic equilibrium and energy metabolism in chicken spleens. Oncotarget 8(32):52629–52641. https://doi.org/10.18632/oncotarget.17539
Yang D, Elner SG, Lin LR, Reddy VN, Petty HR, Elner VM (2009) Association of superoxide anions with retinal pigment epithelial cell apoptosis induced by mononuclear phagocytes. Invest Ophthalmol Vis Sci 50(10):4998–5005. https://doi.org/10.1167/iovs.09-3620
Yıldızhan K, Nazıroğlu M (2019) Microglia and its role in neurodegenerative diseases. J Cell Neurosci Oxid Stress 11:861–873. https://doi.org/10.37212/jcnos.683407
Yıldızhan K, Nazıroğlu M (2020) Glutathione depletion and Parkinsonian neurotoxin MPP+-induced TRPM2 channel activation play central roles in oxidative cytotoxicity and inflammation in microglia. Mol Neurobiol 57(8):3508–3525. https://doi.org/10.1007/s12035-020-01974-7
Yoshioka Y, Sugino Y, Shibagaki F, Yamamuro A, Ishimaru Y, Maeda S (2020) Dopamine attenuates lipopolysaccharide-induced expression of proinflammatory cytokines by inhibiting the nuclear translocation of NF-κB p65 through the formation of dopamine quinone in microglia. Eur J Pharmacol 866:172826. https://doi.org/10.1016/j.ejphar.2019.172826
Yüksel E, Nazıroğlu M, Şahin M, Çiğ B (2017) Involvement of TRPM2 and TRPV1 channels on hyperalgesia, apoptosis and oxidative stress in rat fibromyalgia model: protective role of selenium. Sci Rep 7(1):17543. https://doi.org/10.1038/s41598-017-17715-1
Zhang XY, Xu ZP, Wang W, Cao JB, Fu Q, Zhao WX, Li Y, Huo XL, Zhang LM, Li YF, Mi WD (2018) Vitamin C alleviates LPS-induced cognitive impairment in mice by suppressing neuroinflammation and oxidative stress. Int Immunopharmacol 65:438–447. https://doi.org/10.1016/j.intimp.2018.10.020
Zhu T, Zhao Y, Hu H, Zheng Q, Luo X, Ling Y, Ying Y, Shen Z, Jiang P, Shu Q (2019) TRPM2 channel regulates cytokines production in astrocytes and aggravates brain disorder during lipopolysaccharide-induced endotoxin sepsis. Int Immunopharmacol 75:105836. https://doi.org/10.1016/j.intimp.2019.105836
Funding
The study was supported by BSN Health, Analyses, Innovation, Consultancy, Organization, Agriculture and Industry Ltd., Goller Bolgesi Teknokenti, Isparta, Turkey (Project No: 2018–30). There is no financial disclosure of the current study.
Author information
Authors and Affiliations
Contributions
MN and YA formulated the present hypothesis and MN were responsible for writing the report. KY was responsible for isolating the microglia and analyzing the cytokine and patch-clamp. MN was responsible for the laser confocal microscope analyses. YA was also responsible from plate reader analyses. YA made critical revision for the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Akyuva, Y., Nazıroğlu, M. & Yıldızhan, K. Selenium prevents interferon-gamma induced activation of TRPM2 channel and inhibits inflammation, mitochondrial oxidative stress, and apoptosis in microglia. Metab Brain Dis 36, 285–298 (2021). https://doi.org/10.1007/s11011-020-00624-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-020-00624-0