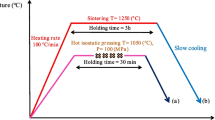
Abstract
In the present study, the microstructure, mechanical properties, corrosion behavior, wettability, haemocompatibility, and cytocompatibility of the as-cast and as-rolled biodegradable Zn-1Mg-0.1Ca and Zn-1Mg-0.5Ca have been systematically investigated to evaluate their feasibility as potential biodegradable materials. The results demonstrated that the Zn-1Mg-0.1Ca have significantly improved mechanical properties, with the yield strength (YS), ultimate tensile strength (UTS), and elongation of as-rolled Zn-1Mg-0.1Ca are (209.04 ± 28.31) MPa, (331.51 ± 40.06) MPa, and (35.43 ± 3.53)%, respectively. Wettability test results demonstrated that the Zn-1Mg-0.1Ca and Zn-1Mg-0.5Ca have hydrophilic surfaces that can enhance cell responses and tissue-implant interactions. The haemocompatibility evaluation showed that the hemolysis ratio of Zn-1Mg-0.1Ca have a low hemolysis ratio of 0.6%; the platelets remain sphere morphology and are not activated. High cell viability indicates the cytocompatibility of the as-rolled Zn-1Mg-0.1Ca alloy. The Zn-1Mg-0.1Ca alloy can be considered as new suitable biodegradable Zn-based alloys for further biomedical applications.












Similar content being viewed by others
References
Han H-S, Loffredo S, Jun I, Edwards J, Kim Y-C, Seok H-K. et al. Current status and outlook on the clinical translation of biodegradable metals. Mater Today. 2019;23:57–71. https://doi.org/10.1016/j.mattod.2018.05.018.
Cho DH, Nam JH, Lee BW, Cho KM, Park IM. Effect of Mn addition on grain refinement of biodegradable Mg 4Zn 0.5Ca alloy. J Alloy Compd. 2016;676:461–8. https://doi.org/10.1016/j.jallcom.2016.03.182.
Gui Z, Kang Z, Li Y. Mechanical and corrosion properties of Mg-Gd-Zn-Zr-Mn biodegradable alloy by hot extrusion. J Alloy Compd. 2016;685:222–30. https://doi.org/10.1016/j.jallcom.2016.05.241.
Salleh EM, Zuhailawati H, Ramakrishnan S, Gepreel MA-H. A statistical prediction of density and hardness of biodegradable mechanically alloyed Mg–Zn alloy using fractional factorial design. J Alloy Compd. 2015;644:476–84. https://doi.org/10.1016/j.jallcom.2015.04.090.
Roche V, Koga GY, Matias TB, Kiminami CS, Bolfarini C, Botta WJ, et al. Degradation of biodegradable implants: the influence of microstructure and composition of Mg-Zn-Ca alloys. J Alloy Compd. 2019;774:168–81. https://doi.org/10.1016/j.jallcom.2018.09.346.
Sotoudeh Bagha P, Khakbiz M, Sheibani S, Hermawan H. Design and characterization of nano and bimodal structured biodegradable Fe-Mn-Ag alloy with accelerated corrosion rate. J Alloy Compd. 2018;767:955–65. https://doi.org/10.1016/j.jallcom.2018.07.206.
Kraus T, Moszner F, Fischerauer S, Fiedler M, Martinelli E, Eichler J, et al. Biodegradable Fe-based alloys for use in osteosynthesis: outcome of an in vivo study after 52 weeks. Acta Biomater. 2014;10:3346–53. https://doi.org/10.1016/j.actbio.2014.04.007.
Sotoudeh Bagha P, Khakbiz M, Safaie N, Sheibani S, Ebrahimi-Barough S. Effect of high energy ball milling on the properties of biodegradable nanostructured Fe-35 wt.%Mn alloy. J Alloy Compd. 2018;768:166–75. https://doi.org/10.1016/j.jallcom.2018.07.261.
Yang H, Wang C, Liu C, Chen H, Wu Y, Han J, et al. Evolution of the degradation mechanism of pure zinc stent in the one-year study of rabbit abdominal aorta model. Biomaterials 2017;145:92–105. https://doi.org/10.1016/j.biomaterials.2017.08.022.
Saghiri MA, Asatourian A, Orangi J, Sorenson CM, Sheibani N. Functional role of inorganic trace elements in angiogenesis-Part II: Cr, Si, Zn, Cu, and S. Crit Rev Oncol Hematol. 2015;96:143–55.
Purushothaman M, Gudrun R, Michal T, Bernhard H. Zinc modulates PPARγ signaling and activation of porcine endothelial cells. J Nutr. 2003;133:3058.
Kaji T, Fujiwara Y, Yamamoto C, Sakamoto M, Kozuka H. Stimulation by zinc of cultured vascular endothelial cell proliferation: possible involvement of endogenous basic fibroblast growth factor. Life Sci. 1994;55:1781.
Little PJ, Bhattacharya R, Moreyra AE, Korichneva IL. Zinc and cardiovascular disease. Nutrition 2010;26:1050–7.
Corbo MD, Lam J. Zinc deficiency and its management in the pediatric population: a literature review and proposed etiologic classification. J Am Acad Dermatol. 2013;69:616–24 e1. https://doi.org/10.1016/j.jaad.2013.04.028.
Yamaguchi M. Role of zinc in bone formation and bone resorption. J Trace Elem Exp Med Off Publ Int Soc Trace Elem Res Hum. 1998;11:119–35.
Mostaed E, Sikora-Jasinska M, Drelich JW, Vedani M. Zinc-based alloys for degradable vascular stent applications. Acta Biomater. 2018;71:1–23. https://doi.org/10.1016/j.actbio.2018.03.005.
Mostaed E, Sikora-Jasinska M, Mostaed A, Loffredo S, Demir AG, Previtali B, et al. Novel Zn-based alloys for biodegradable stent applications: design, development and in vitro degradation. J Mech Behav Biomed Mater. 2016;60:581–602. https://doi.org/10.1016/j.jmbbm.2016.03.018.
ASTM-E8-13. Standard test methods for tension testing of metallic materials. In: Annual book of ASTM standards. American Society for Testing and Materials. 2013.
ASTM G102-89. Standard practice for calculation of corrosion rates and related information from electrochemical measurements. In: Annual Book of ASTM Standards. American Society for Testing and Materials. 2010;89.
ISO 10993‐5. Biological evaluation of medical devices–Part 5: tests for in vitro cytotoxicity. Geneva: International Organization for Standardization; 2009.
Agrawal G, Negi YS, Pradhan S, Dash M, Samal SK. 3—Wettability and contact angle of polymeric biomaterials. In: Tanzi MC, Farè S, editors. Characterization of Polymeric Biomaterials. Woodhead Publishing; 2017. p. 57–81.
Spriano S, Sarath Chandra V, Cochis A, Uberti F, Rimondini L, Bertone E, et al. How do wettability, zeta potential and hydroxylation degree affect the biological response of biomaterials? Mater Sci Eng C Mater Biol Appl. 2017;74:542–55. https://doi.org/10.1016/j.msec.2016.12.107.
Zhao G, Schwartz Z, Wieland M, Rupp F, Geis-Gerstorfer J, Cochran DL, et al. High surface energy enhances cell response to titanium substrate microstructure. J Biomed Mater Res Part A. 2005;74:49–58. https://doi.org/10.1002/jbm.a.30320.
Seeger JM, Ingegno MD, Bigatan E, Klingman N, Amery D, Widenhouse C, et al. Hydrophilic surface modification of metallic endoluminal stents. J Vasc Surg. 1995;22:327–36. https://doi.org/10.1016/S0741-5214(95)70148-6.
Shim JW, Bae I-H, Park DS, Lee S-Y, Jang E-J, Lim K-S, et al. Hydrophilic surface modification of coronary stent using an atmospheric pressure plasma jet for endothelialization. J Biomater Appl. 2018;32:1083–9. https://doi.org/10.1177/0885328217748465.
ASTM F756-13. Standard practice for assessment of hemolytic properties of materials. In: Annual Book of ASTM Standards. 2013.
Acknowledgements
The present work was supported by the National Natural Science Foundation of China (No. 31700819), the Young Elite Scientists Sponsorship Program by CAST (YESS, No.2018QNRC001), the research funds of Lepu Medical Technology (Beijing) Co., Ltd and the Fundamental Research Funds for the Central Universities (No. 06500098).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, H., Shen, C., Ruan, D. et al. Microstructure, mechanical properties, and in vitro behavior of biodegradable Zn-1Mg-0.1Ca and Zn-1Mg-0.5Ca. J Mater Sci: Mater Med 31, 88 (2020). https://doi.org/10.1007/s10856-020-06444-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10856-020-06444-z