Abstract
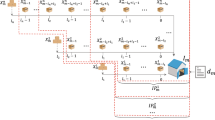
We study a new class of inventory control problems, the recruitment stocking problem (RSP), applicable to general recruitment systems and products with limited supplies. Recruitment stocking occurs routinely in many organizations with the goal of identifying qualified candidates rapidly and cost effectively. This activity may take place at multiple sites simultaneously to shorten the recruitment time. Examples include recruiting patients in clinical trials, enlisting personnel in the military, and recruiting customers for product sampling in market testing and promotion. RSP differs from the extant inventory management literature in that it stipulates a finite recruitment target, so that recruitment is terminated as soon as the total number of recruits across all locations reaches that prescribed target. This distinctive feature of RSP calls for the development of new stochastic models to evaluate and optimize system performance. Thus, we present a novel methodology of relaxation and decomposition to characterize the probability distribution of rejections in RSP (number of arrivals to an empty inventory). This method provides a basis for efficient and accurate evaluation of the Type 2 service level and expected recruitment time. We also leverage the attendant computational efficiency to develop optimization algorithms to compute the optimal stocking quantities.



Similar content being viewed by others
References
Barnett International. (2011). Good clinical practice Q&A: Focus on study drug. Journal of Clinical Research Best Practices, 7(7), 11.
Dentcheva, D., Prékopa, A., & Ruszczynski, A. (2000). Concavity and efficient points of discrete distributions in probabilistic programming. Mathematical Programming, 89(1), 55–77.
Dupuis, P., Nuzman, C., & Whiting, P. (2004). Large deviation asymptotics for occupancy problems. Annals of Probability, 32, 2765–2818.
Eramo, V., Listanti, M., Nuzman, C., & Whiting, P. (2002). Optical switch dimensioning and the classical occupancy problem. International Journal of Communication Systems, 15(2–3), 127–147.
Commission, European. (2005). Commission directive of April 2005 laying down principles and detailed guidelines for good clinical practice as regards investigational medicinal products for human use, as well as the requirements for authorisation of the manufacturing or importation of such products. Official Journal of the European Union, 91, 13–19.
Fleischhacker, A., Ninh, A., & Zhao, Y. (2015). Positioning inventory in clinical trial supply chains. Production and Operations Management, 24(6), 991–1011.
Fox, B. (1966). Discrete optimization via marginal analysis. Management Science, 13(3), 210–216.
Frey, J. (2009). An algorithm for computing rectangular multinomial probabilities. Journal of Statistical Computation and Simulation, 79(12), 1483–1489.
Glickman, S. W., McHutchison, J. G., Peterson, E. D., Cairns, C. B., Harrington, R. A., Califf, R. M., et al. (2009). Ethical and scientific implications of the globalization of clinical research. New England Journal of Medicine, 360(8), 816–823.
Graves, S. C., & Willems, S. P. (2003). Supply chain design: Safety stock placement and supply chain configuration. A.G. de Kok and S.C. Graves, eds. Handbooks in Operations Research and Management Science, 11, 95–132.
Han, Q., Fleischhacker, A. & Fok, P. (2015). First passage times in clinical trial supply chains with stockouts. In: Working paper, Alfred Lener College of Business and Economics. University of Delaware, DE.
Holst, L. (1986). On birthday, collectors’, occupancy and other classical urn problems. International Statistical Review/Revue Internationale de Statistique, 54(1), 15–27.
Jain, D., Mahajan, V., & Muller, E. (1995). An approach for determining optimal product sampling for the diffusion of a new product. Journal of Product Innovation Management, 12(2), 124–135.
Johnson, N. L., & Kotz, S. (1977). Urn models and their application: An approach to modern discrete probability theory (Vol. 77). New York: Wiley.
Lachin, J. M. (1981). Introduction to sample size determination and power analysis for clinical trials. Controlled Clinical Trials, 2(2), 93–113.
Lebrun, R. (2013). Efficient time/space algorithm to compute rectangular probabilities of multinomial, multivariate hypergeometric and multivariate Pólya distributions. Statistics and Computing, 23(5), 1–9.
Lefew, M., Ninh, A., & Anisimov, V. (2020). End-to-end drug supply management in multicenter trials. Methodology and Computing in Applied Probability. https://doi.org/10.1007/s11009-020-09776-z.
Levin, B. (1981). A representation for multinomial cumulative distribution functions. The Annals of Statistics, 9(5), 1123–1126.
Murty, V. N. (1981). Counting the integer solutions of a linear equation with unit coefficients. Mathematics Magazine, 54(2), 79–81.
Ninh, A., LeFew, M. & Anisimov, V. (2019). Clinical trial simulation: Modeling and practical considerations. In: 2019 winter simulation conference (WSC) (pp. 118–132). IEEE.
Oncolytics Biotech. (2003). Oncolytics biotech inc. announces completion of manufacturing process development for REOLYSIN. New York: PR Newswire.
Porteus, E. L. (2002). Foundations of stochastic inventory theory. Stanford: Stanford University Press.
Powell, M. (2010). Presentation by senior vice president, pharmaceutical development, Bristol-Myers Squibb. In: Argyle executive forum’s 2010 leadership in pharmaceuticals & biotechnology conference. Argyle executive forum, June 22 2010.
Prékopa, A. (1988). Boole–Bonferroni inequalities and linear programming. Operations Research, 36(1), 145–162.
Prékopa, A. (1990). Sharp bounds on probabilities using linear programming. Operations Research, 38(2), 227–239.
Prékopa, A., Ninh, A., & Alexe, G. (2016). On the relationship between the discrete and continuous bounding moment problems and their numerical solutions. Annals of Operations Research, 238(1–2), 521–575.
Ramakrishna, M. V. & Mukhopadhyay, P. (1988). Analysis of bounded disorder file organization. In: Proceedings of the 7th ACM SIGACT-SIGMOD-SIGART symposium on Principles of database systems (pp. 117–125). ACM.
Rowland, C. (2003). Clinical trials seen shifting overseas. International Journal of Health Services, 34(3), 555–556.
Sakpal, T. V. (2010). Sample size estimation in clinical trial. Perspectives in Clinical Research, 1(2), 67.
Simchi-Levi, D., & Zhao, Y. (2011). Performance evaluation of stochastic multi-echelon inventory systems: A survey. Advances in Operations Research. https://doi.org/10.1155/2012/126254.
Thiers, F. A., Sinskey, A. J., & Berndt, E. R. (2008). Trends in the globalization of clinical trials. Nature Reviews Drug Discovery, 7(1), 13–14.
Usanmaz, G. (2000). End-of-life cycle product management. Master’s thesis, Diss. Massachusetts Institute of Technology.
Vallance, P. (2011). Presentation at the Cowen and company 31st annual healthcare conference in Boston. In: Cowen and Company Healthcare Conference, March 17 2011.
Zipkin, P. H. (2000). Foundations of inventory management. New York: McGraw-Hill.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ninh, A., Melamed, B. & Zhao, Y. Analysis and optimization of recruitment stocking problems. Ann Oper Res 295, 747–767 (2020). https://doi.org/10.1007/s10479-020-03822-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10479-020-03822-2