Abstract
Introduction
Studies have shown that systemic levels of branched-chain amino acids (BCAAs) and aromatic amino acids (AAAs) are elevated in cardiometabolic diseases (CMDs) in populations resident in high income countries. However, little is known about the association of BCAAs and AAAs with metabolic syndrome and its components in Asian Indian (AI) and Black African (BA) populations.
Objective
The aim of this study was to describe the association of BCAAs and AAAs with the metabolic syndrome, its individual components and insulin resistance in AI and BA populations.
Methods
Serum samples collected from AI (n = 349) and BA (n = 369) subjects were used to measure levels of BCAAs and AAAs by ultra-pressure liquid chromatography tandem mass spectrometry (UPLC-MS/MS). Anthropometric, demographic and cardiometabolic variables were measured in all subjects.
Results
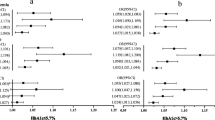
The sum of BCAAs and AAAs was higher in AIs compared to BAs. The BCAAs and AAAs were positively associated with insulin resistance, metabolic syndrome and its individual components. This was particularly the case for AI subjects, in unadjusted regression models. However, these associations were non-significant after adjusting for co-variates, particularly visceral adipose tissue (VAT). Triglyceride levels were significantly associated with valine and leucine levels in BAs even after adjustment for co-variates. Lastly, we found that fasting circulatory BCAA and AAA levels are strongly correlated with VAT in both populations.
Conclusion
This study identified specific associations of serum valine and leucine levels with triglycerides in BAs. The association of amino acids with CMDs was observed in AIs, but was found to be the result of confounding by VAT. Further studies are required to determine whether BCAAs and AAAs are aetiological factors in CMDs and how VAT modulates their serum levels.

Similar content being viewed by others
References
Alberti, K. G. M. M., Eckel, R. H., Grundy, S. M., Zimmet, P. Z., Cleeman, J. I., Donato, K. A., et al. (2009). Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute. Circulation, 120(16), 1640–1645. https://doi.org/10.1161/CIRCULATIONAHA.109.192644
Banerji, M. A., Faridi, N., Atluri, R., Chaiken, R. L., & Lebovitz, H. E. (1999). Body composition, visceral fat, leptin, and insulin resistance in Asian Indian men. The Journal of Clinical Endocrinology & Metabolism, 84(1), 137–144. https://doi.org/10.1210/jcem.84.1.5371
Batch, B. C., Shah, S. H., Newgard, C. B., Turer, C. B., Haynes, C., Bain, J. R., et al. (2013). Branched chain amino acids are novel biomarkers for discrimination of metabolic wellness. Metabolism: Clinical and Experimental, 62(7), 961–969. https://doi.org/10.1016/j.metabol.2013.01.007
Bonds, D. E., Zaccaro, D. J., Karter, A. J., Selby, J. V., Saad, M., & Goff, D. C. (2003). Ethnic and racial differences in diabetes care: The insulin resistance atherosclerosis study. Diabetes Care, 26(4), 1040–1046.
Cappuccio, F. P., & Miller, M. A. (2016). Cardiovascular disease and hypertension in sub-Saharan Africa: Burden, risk and interventions. Internal and Emergency Medicine, 11(3), 299–305. https://doi.org/10.1007/s11739-016-1423-9
Cardiovascular diseases (CVDs). (n.d.). Retrieved May 8, 2020 from https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds).
Chevalier, S., Marliss, E. B., Morais, J. A., Lamarche, M., & Gougeon, R. (2005). Whole-body protein anabolic response is resistant to the action of insulin in obese women. The American Journal of Clinical Nutrition, 82(2), 355–365. https://doi.org/10.1093/ajcn.82.2.355
Cummings, N. E., Williams, E. M., Kasza, I., Konon, E. N., Schaid, M. D., Schmidt, B. A., et al. (2018). Restoration of metabolic health by decreased consumption of branched-chain amino acids. The Journal of Physiology, 596(4), 623–645. https://doi.org/10.1113/JP275075
Dugas, L. R., Chorell, E., Plange-Rhule, J., Lambert, E. V., Cao, G., Cooper, R. S., et al. (2016). Obesity-related metabolite profiles of black women spanning the epidemiologic transition. Metabolomics, 12(3), 45. https://doi.org/10.1007/s11306-016-0960-6
Ferris, W. F., Naran, N. H., Crowther, N. J., Rheeder, P., van der Merwe, L., & Chetty, N. (2005). The relationship between insulin sensitivity and serum adiponectin levels in three population groups. Hormone and Metabolic Research, 37(11), 695–701. https://doi.org/10.1055/s-2005-870580
Fiehn, O., Garvey, W. T., Newman, J. W., Lok, K. H., Hoppel, C. L., & Adams, S. H. (2010). Plasma metabolomic profiles reflective of glucose homeostasis in non-diabetic and type 2 diabetic obese African-American women. PLoS ONE, 5(12), e15234. https://doi.org/10.1371/journal.pone.0015234
Flores-Guerrero, J. L., Groothof, D., Connelly, M. A., Otvos, J. D., Bakker, S. J. L., & Dullaart, R. P. F. (2019). Concentration of branched-chain amino acids is a strong risk marker for incident hypertension. Hypertension, 74(6), 1428–1435. https://doi.org/10.1161/HYPERTENSIONAHA.119.13735
Gängler, S., Waldenberger, M., Artati, A., Adamski, J., van Bolhuis, J. N., Sørgjerd, E. P., et al. (2019). Exposure to disinfection byproducts and risk of type 2 diabetes: A nested case–control study in the HUNT and Lifelines cohorts. Metabolomics. https://doi.org/10.1007/s11306-019-1519-0.
George, J. A., Norris, S. A., Toman, M., Snyman, T., & Crowther, N. J. (2016). Visceral adiposity is a predictor of parathyroid hormone levels in healthy adults. Journal of Endocrinological Investigation, 39(4), 447–453. https://doi.org/10.1007/s40618-015-0400-x
George, J. A., Norris, S. A., van Deventer, H. E., & Crowther, N. J. (2013). The association of 25 hydroxyvitamin D and parathyroid hormone with metabolic syndrome in two ethnic groups in South Africa. PLoS ONE, 8(4), e61282. https://doi.org/10.1371/journal.pone.0061282
George, J. A., Norris, S. A., van Deventer, H. E., Pettifor, J. M., & Crowther, N. J. (2014). Effect of adiposity, season, diet and calcium or vitamin D supplementation on the vitamin D status of healthy urban African and Asian-Indian adults. The British Journal of Nutrition, 112(4), 590–599. https://doi.org/10.1017/S0007114514001202
Gogna, N., Krishna, M., Oommen, A. M., & Dorai, K. (2015). Investigating correlations in the altered metabolic profiles of obese and diabetic subjects in a South Indian Asian population using an NMR-based metabolomic approach. Molecular BioSystems, 11(2), 595–606. https://doi.org/10.1039/c4mb00507d
Gómez-Olivé, F. X., Ali, S. A., Made, F., Kyobutungi, C., Nonterah, E., Micklesfield, L., et al. (2017). Regional and sex differences in the prevalence and awareness of hypertension: An H3Africa AWI-gen study across 6 sites in sub-saharan Africa. Global Heart, 12(2), 81–90. https://doi.org/10.1016/j.gheart.2017.01.007
Hsu, W. C., Araneta, M. R. G., Kanaya, A. M., Chiang, J. L., & Fujimoto, W. (2015). BMI cut points to identify at-risk Asian Americans for type 2 diabetes screening. Diabetes Care, 38(1), 150–158. https://doi.org/10.2337/dc14-2391
Huffman, K. M., Shah, S. H., Stevens, R. D., Bain, J. R., Muehlbauer, M., Slentz, C. A., et al. (2009). Relationships between circulating metabolic intermediates and insulin action in overweight to obese, inactive men and women. Diabetes Care, 32(9), 1678–1683. https://doi.org/10.2337/dc08-2075
Hulshof, KFa. M., Brussaard, J. H., Kruizinga, A. G., Telman, J., & Löwik, M. R. H. (2003). Socio-economic status, dietary intake and 10 y trends: The Dutch national food consumption survey. European Journal of Clinical Nutrition, 57(1), 128–137. https://doi.org/10.1038/sj.ejcn.1601503
Kobayashi, R., Shimomura, Y., Murakami, T., Nakai, N., Fujitsuka, N., Otsuka, M., et al. (1997). Gender difference in regulation of branched-chain amino acid catabolism. The Biochemical Journal, 327(Pt 2), 449–453. https://doi.org/10.1042/bj3270449
Lackey, D. E., Lynch, C. J., Olson, K. C., Mostaedi, R., Ali, M., Smith, W. H., et al. (2013). Regulation of adipose branched-chain amino acid catabolism enzyme expression and cross-adipose amino acid flux in human obesity. American Journal of Physiology Endocrinology and Metabolism, 304(11), E1175–E1187. https://doi.org/10.1152/ajpendo.00630.2012
Lamont, L. S., McCullough, A. J., & Kalhan, S. C. (2003). Gender differences in the regulation of amino acid metabolism. Journal of Applied Physiology, 95(3), 1259–1265. https://doi.org/10.1152/japplphysiol.01028.2002
Lee, C. C., Watkins, S. M., Lorenzo, C., Wagenknecht, L. E., Il’yasova, D., Chen, Y.-D. I., et al. (2016). Branched-chain amino acids and insulin metabolism: The insulin resistance atherosclerosis study (IRAS). Diabetes Care, 39(4), 582–588. https://doi.org/10.2337/dc15-2284
Lloyd-Sherlock, P., Beard, J., Minicuci, N., Ebrahim, S., & Chatterji, S. (2014). Hypertension among older adults in low- and middle-income countries: Prevalence, awareness and control. International Journal of Epidemiology, 43(1), 116–128. https://doi.org/10.1093/ije/dyt215
Matthews, D. R., Hosker, J. P., Rudenski, A. S., Naylor, B. A., Treacher, D. F., & Turner, R. C. (1985). Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia, 28(7), 412–419. https://doi.org/10.1007/bf00280883
Mook-Kanamori, D. O., Römisch-Margl, W., Kastenmüller, G., Prehn, C., Petersen, A. K., Illig, T., et al. (2014). Increased amino acids levels and the risk of developing of hypertriglyceridemia in a 7-year follow-up. Journal of Endocrinological Investigation, 37(4), 369–374. https://doi.org/10.1007/s40618-013-0044-7
Naran, N. H., Haagensen, M., & Crowther, N. J. (2018). Steatosis in South African women: How much and why? PLoS ONE, 13(1), e0191388. https://doi.org/10.1371/journal.pone.0191388
Newgard, C. B. (2012). Interplay between lipids and branched-chain amino acids in development of insulin resistance. Cell Metabolism, 15(5), 606–614. https://doi.org/10.1016/j.cmet.2012.01.024
Newgard, C. B., An, J., Bain, J. R., Muehlbauer, M. J., Stevens, R. D., Lien, L. F., et al. (2009). A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metabolism, 9(4), 311–326. https://doi.org/10.1016/j.cmet.2009.02.002
Ng, M., Fleming, T., Robinson, M., Thomson, B., Graetz, N., Margono, C., et al. (2014). Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: A systematic analysis for the global burden of disease study 2013. The Lancet, 384(9945), 766–781. https://doi.org/10.1016/S0140-6736(14)60460-8
Pakiet, A., Wilczynski, M., Rostkowska, O., Korczynska, J., Jabłonska, P., Kaska, L., et al. (2019). The effect of one anastomosis gastric bypass on branched-chain fatty acid and branched-chain amino acid metabolism in subjects with morbid obesity. Obesity Surgery. https://doi.org/10.1007/s11695-019-04157-z
Peer, N., Balakrishna, Y., de Villiers, A., Naidoo, P., & Pucci, G. (2018). Differential associations of cardio-metabolic diseases by population group, gender and adiposity in South Africa. PLOS ONE, 13(9), e0202899.
Peer, N., Steyn, K., Lombard, C., Gwebushe, N., & Levitt, N. (2013). A high burden of hypertension in the urban black population of Cape Town: The cardiovascular risk in black South Africans (CRIBSA) study. PLoS ONE, 8(11), e78567. https://doi.org/10.1371/journal.pone.0078567
Pietiläinen, K. H., Naukkarinen, J., Rissanen, A., Saharinen, J., Ellonen, P., Keränen, H., et al. (2008). Global transcript profiles of fat in monozygotic twins discordant for BMI: Pathways behind acquired obesity. PLoS Medicine, 5(3), e51. https://doi.org/10.1371/journal.pmed.0050051
Reddy, K. S., & Yusuf, S. (1998). Emerging epidemic of cardiovascular disease in developing countries. Circulation, 97(6), 596–601.
Ribeiro, R. V., Solon-Biet, S. M., Pulpitel, T., Senior, A. M., Cogger, V. C., Clark, X., et al. (2019). Of older mice and men: Branched-chain amino acids and body composition. Nutrients, 11(8), 1882. https://doi.org/10.3390/nu11081882
Richter, L. M., Norris, S. A., & De Wet, T. (2004). Transition from birth to ten to birth to twenty: the South African cohort reaches 13 years of age. Paediatric and Perinatal Epidemiology, 18(4), 290–301. https://doi.org/10.1111/j.1365-3016.2004.00572.x
Ruiz-Canela, M., Toledo, E., Clish, C. B., Hruby, A., Liang, L., Salas-Salvadó, J., et al. (2016). Plasma branched-chain amino acids and incident cardiovascular disease in the PREDIMED trial. Clinical Chemistry, 62(4), 582–592. https://doi.org/10.1373/clinchem.2015.251710
Schutte, A. E., Schutte, R., Huisman, H. W., van Rooyen, J. M., Fourie, C. M. T., Malan, N. T., et al. (2012). Are behavioural risk factors to be blamed for the conversion from optimal blood pressure to hypertensive status in Black South Africans? A 5-year prospective study. International Journal of Epidemiology, 41(4), 1114–1123. https://doi.org/10.1093/ije/dys106
Sesso, H. D., Cook, N. R., Buring, J. E., Manson, J. E., & Gaziano, J. M. (2008). Alcohol consumption and the risk of hypertension in women and men. Hypertension, 51(4), 1080–1087. https://doi.org/10.1161/HYPERTENSIONAHA.107.104968
Shah, S. H., Crosslin, D. R., Haynes, C. S., Nelson, S., Turer, C. B., Stevens, R. D., et al. (2012). Branched-chain amino acid levels are associated with improvement in insulin resistance with weight loss. Diabetologia, 55(2), 321–330. https://doi.org/10.1007/s00125-011-2356-5
Solon-Biet, S. M., Cogger, V. C., Pulpitel, T., Wahl, D., Clark, X., Bagley, E. E., et al. (2019). Branched-chain amino acids impact health and lifespan indirectly via amino acid balance and appetite control. Nature Metabolism, 1(5), 532–545. https://doi.org/10.1038/s42255-019-0059-2
Tai, E. S., Tan, M. L. S., Stevens, R. D., Low, Y. L., Muehlbauer, M. J., Goh, D. L. M., et al. (2010). Insulin resistance is associated with a metabolic profile of altered protein metabolism in Chinese and Asian-Indian men. Diabetologia, 53(4), 757–767. https://doi.org/10.1007/s00125-009-1637-8
Takashina, C., Tsujino, I., Watanabe, T., Sakaue, S., Ikeda, D., Yamada, A., et al. (2016). Associations among the plasma amino acid profile, obesity, and glucose metabolism in Japanese adults with normal glucose tolerance. Nutrition & Metabolism, 13, 5. https://doi.org/10.1186/s12986-015-0059-5
Tillin, T., Hughes, A. D., Wang, Q., Würtz, P., Ala-Korpela, M., Sattar, N., et al. (2015). Diabetes risk and amino acid profiles: Cross-sectional and prospective analyses of ethnicity, amino acids and diabetes in a South Asian and European cohort from the SABRE (Southall And Brent REvisited) study. Diabetologia, 58(5), 968–979. https://doi.org/10.1007/s00125-015-3517-8
Tomoda, K., Yoshikawa, M., Kubo, K., Koyama, N., Yamamoto, Y., & Kimura, H. (2012). Effects of cigarettes smoke on branched chain amino acids (BCAA) levels in plasma and skeletal muscles in rats. In A64. Local and systemic effects of smoke exposure (Vols. 1–315, pp. A2014–A2014). American Thoracic Society.
Wang, T. J., Larson, M. G., Vasan, R. S., Cheng, S., Rhee, E. P., McCabe, E., et al. (2011). Metabolite profiles and the risk of developing diabetes. Nature Medicine, 17(4), 448–453. https://doi.org/10.1038/nm.2307
WHO Expert Consultation. (2004). Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet, 363(9403), 157–163. https://doi.org/10.1016/S0140-6736(03)15268-3
WHO Global status report on noncommunicable diseases 2010. (n.d.). Retrieved May 8, 2020 from https://www.who.int/nmh/publications/ncd_report2010/en/.
WHO Noncommunicable diseases country profiles 2014. (2014). WHO. Retrieved April 12, 2018 from http://www.who.int/nmh/publications/ncd-profiles-2014/en/.
Wild, S., Roglic, G., Green, A., Sicree, R., & King, H. (2004). Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care, 27(5), 1047–1053.
Würtz, P., Mäkinen, V.-P., Soininen, P., Kangas, A. J., Tukiainen, T., Kettunen, J., et al. (2012). Metabolic signatures of insulin resistance in 7098 young adults. Diabetes, 61(6), 1372–1380. https://doi.org/10.2337/db11-1355
Wyk, V. P., Msemburi, W., Laubscher, R., Dorrington, R. E., Groenewald, P., Glass, T., et al. (2016). Mortality trends and differentials in South Africa from 1997 to 2012: Second National Burden of Disease Study. The Lancet Global Health, 4(9), e642–e653. https://doi.org/10.1016/S2214-109X(16)30113-9
Xie, G., Ma, X., Zhao, A., Wang, C., Zhang, Y., Nieman, D., et al. (2014). The metabolite profiles of the obese population are gender-dependent. Journal of Proteome Research, 13(9), 4062–4073. https://doi.org/10.1021/pr500434s
Yamada, C., Kondo, M., Kishimoto, N., Shibata, T., Nagai, Y., Imanishi, T., et al. (2015). Association between insulin resistance and plasma amino acid profile in non-diabetic Japanese subjects. Journal of Diabetes Investigation, 6(4), 408–415. https://doi.org/10.1111/jdi.12323
Yamakado, M., Tanaka, T., Nagao, K., Ishizaka, Y., Mitushima, T., Tani, M., et al. (2012). Plasma amino acid profile is associated with visceral fat accumulation in obese Japanese subjects. Clinical Obesity, 2(1–2), 29–40. https://doi.org/10.1111/j.1758-8111.2012.00039.x
Yang, P., Hu, W., Fu, Z., Sun, L., Zhou, Y., Gong, Y., et al. (2016). The positive association of branched-chain amino acids and metabolic dyslipidemia in Chinese Han population. Lipids in Health and Disease, 15, 120. https://doi.org/10.1186/s12944-016-0291-7
Yang, R., Dong, J., Guo, H., Li, H., Wang, S., Zhao, H., et al. (2013). Rapid and precise measurement of serum branched-chain and aromatic amino acids by isotope dilution liquid chromatography tandem mass spectrometry. PLoS ONE, 8(12), e81144. https://doi.org/10.1371/journal.pone.0081144
Acknowledgements
This study was supported by the National Health Laboratory Services (NHLS) (94413) and the University of the Witwatersrand (00141084631015CHEMPATTS). The University of the Witwatersrand, the DST-NRF Centre of Excellence in Human Development, and the South African Medical Research Council supports the Birth to Twenty Cohort.
Author information
Authors and Affiliations
Contributions
L.K. performed the experiment, was involved in in experimental procedure, the analysis and interpretation of results and wrote the manuscript. T.S. was involved in the study design and contributed to the experimental procedure. S.A.N. contributed to the experimental procedure and study design. N.J.C. contributed to the data interpretation, statistical analysis of data and reviewed the manuscript. J.A.G. supervised the study, contributed to the design of the study, the data interpretation and reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
Ethical clearance for this study was obtained from the University of Witwatersrand Human Research Ethics Committee (Ethical clearance no: M150669). All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in this study.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Khambule, L., Snyman, T., Norris, S.A. et al. Branched-chain and aromatic amino acids and cardiometabolic risk in Black African and Asian Indian populations. Metabolomics 16, 108 (2020). https://doi.org/10.1007/s11306-020-01734-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-020-01734-7