Abstract
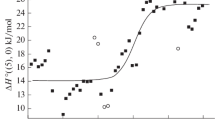
Anionic nonstoichiometry in fluorides (replacement of F1– with O2–) deteriorates the quality of optical materials. Two reviews have been devoted to the initial stage of anionic nonstoichiometry: fluorides MF2 (M = Ca, Sr, Ba) are considered in this paper, and fluorides RF3 (R are 16 rare-earth elements) will be considered in the next one. These 19 fluorides play an important role in the fluoride materials science, comprising more than 70% of 27 MFm that are used to design two-component fluoride crystalline materials. The initial stage of anionic nonstoichiometry in MF2 is the only one in which oxofluorides MF2 – 2xOx with low oxygen content are formed. Partial replacement of F1– with O2– in the fluorite structure is accompanied by thermal stabilization of the structure type when moving up the temperature scale with a maximum in the melting curves of the oxofluoride phase, which decomposes upon cooling. No other intermediate phases containing fluorine and oxygen were found in the MF2–MO systems studied.



Similar content being viewed by others
REFERENCES
B. P. Sobolev, Crystallogr. Rep. 57 (3), 434 (2012).
B. P. Sobolev and N. I. Sorokin, Crystallogr. Rep. 59 (6), 807 (2014).
B. P. Sobolev, The Rare Earth Trifluorides, Part 1: The High Temperature Chemistry of Rare Earth Trifluorides (Ed. by Institut d’Estudis Catalons, Barcelona, 2000).
B. P. Sobolev, The Rare Earth Trifluorides, Part 2: Introduction to Materials Science of Multicomponent Metal Fluoride Crystals (Ed. by Institut d’Estudis Catalans, Barcelona, 2001).
V. Osiko and I. Shcherbakov, Fotonika, No. 3(39), 14 (2013).
B. P. Sobolev, A. M. Golubev, and P. Herrero, Crystallogr. Rep. 48 (1), 141 (2003).
B. P. Sobolev, E. G. Ippolitov, B. M. Zhigarnovskii, and L. S. Garashina, Izv. Akad. Nauk SSSR, Neorg. Mater. 1 (3), 362 (1965).
B. P. Sobolev, Doctoral Dissertation in Chemistry (Institute of Crystallography, Russian Academy of Sciences, Moscow, 1978).
J. S. Anderson, Problems of Nonstoichiometry, Ed. by A. Rabenau (North Holland, Amsterdam, 1970).
R. D. Shannon, Acta Crystallogr. A 32 (5), 75 (1976).
V. V. Osiko, A. A. Sobol’, and M. I. Timoshechkin, Trudy FIAN (Nauka, Moscow, 1972), Vol. 60, p. 71.
D. B. Schinn and H. A. Eick, Inorg. Chem. 8 (2), 232 (1969).
D. S. Stockbarger, J. Opt. Soc. Am. 39 (9), 31 (1949).
B. P. Sobolev, Crystallogr. Rep. 47 (Suppl. 1), 63 (2002).
I. V. Stepanov and P. P. Feofilov, Crystal Growth (Izd-vo AN SSSR. Moscow, 1957) [in Russian], p. 229.
M. E. Fremy, Annal. Chim. Phys. 47, Ser. 3, 5 (1856).
M. A. Mikhailov, Izv. Vost. Fil. Akad. Nauk SSSR, No. 9, 64 (1957).
C. V. Banks, K. E. Burke, et al., Anal. Chim. Acta 19, 239 (1958).
W. Bontinck, Physica 22 (8), 650 (1958).
W. L. Phillips and J. E. Hanlon, J. Am. Ceram. Soc. 46 (9), 447 (1963).
A. Molchanov, J. Friedrich, G. Wehrhan, et al., J. Cryst. Growth 273 (3–4), 629 (2005).
D.-G. Kim, C. van Hoek, C. Liebeske, et al., ISIJ Int. 52 (11), 1945 (2012).
V. I. Nikolaichik, B. P. Sobolev, M. A. Zaporozhets, and A. S. Avilov, Crystallogr. Rep. 57 (2), 299 (2012).
J. T. Mouhovski, Prog. Cryst. Growth Charact. Mater. 53, 79 (2007).
O. G. Polyachenok, Izv. Akad. Nauk SSSR, Neorg. Mater. 2 (6), 958 (1966).
C. R. A. Catlow, J. Phys. Chem. Solids 38, 1131 (1977).
K. T. Jacob, V. S. Saji, and Y. Waseda, Int. J. Appl. Ceram. Technol. 3 (4), 312 (2006).
M. Robinson and D. M. Cripe, J. Appl. Phys. 37 (5), 2072 (1966).
M. Robinson, G. Hills, and D. M. Cripe, U.S. Patent No. 3 649 552 (4 March 1972).
P. P. Budnikov and S. G. Tresvetskii, Dokl. Akad. Nauk SSSR, 89 (3), 479 (1953).
S. K. Maksimov, F. S. Avilov, B. P. Sobolev, and P. Herrero, Proc. IX Ross. Conf. on Electron Microscopy, Chernogolovka, May 28–31,2002, p. 162.
S. K. Maksimov, A. S. Avilov, B. P. Sobolev, and P. Herrero, Zavod. Lab. Diagn. Mater. 69 (10), 24 (2004).
ACKNOWLEDGMENTS
I am grateful to N.I. Sorokin and D.N. Karimov for fruitful discussions and to E.A. Krivandina and Z.I. Zhmurova for supplying crystals.
Funding
This study was supported by the Ministry of Science and Higher Education of the Russian Federation within a State assignment for the Federal Scientific Research Centre “Crystallography and Photonics” of the Russian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by Yu. Sin’kov
Rights and permissions
About this article
Cite this article
Sobolev, B.P. Nonstoichiometry in Inorganic Fluorides. III: Anionic Nonstoichiometry in MF2 (M = Ca, Sr, Ba). Crystallogr. Rep. 65, 678–686 (2020). https://doi.org/10.1134/S106377452005020X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S106377452005020X