Abstract
Introduced species face numerous biological barriers before they can establish in a new environment. Understanding how they overcome these obstacles is crucial for the development of effective risk assessment and regulation. Reproductive biology is known to influence establishment capacity in plants and is widely used for risk assessment. This biological field should receive more attention, and particularly in the case of insects, as they display a wide range of reproductive traits and have a great impact on the economy and environment. Among insects, the order Hymenoptera is of interest for its diversity, both in terms of reproductive traits and introduction history, as invasive species and biological control agents. We review the main reproductive strategies of Hymenoptera, spanning parthenogenesis, sex determination, reproductive parasites and mating strategies, and evaluate their effect on invasive potential. For instance, thelytoky could decrease the strength of Allee effects while Arrhenotoky could increase adaptive potential. A species with complementary sex determination could be more affected by inbreeding than other species, while paternal genome elimination could lead to high levels of homozygosity. Finally, some reproductive behaviours could decrease inbreeding, facilitate mate location or adaptation by encouraging admixture. The two invasive species Apis mellifera scutellata and Leptocybe invasa and the biocontrol agent Aphidius ervi serve as case studies to illustrate the effect of reproductive traits on species capacities to become established in a new area.


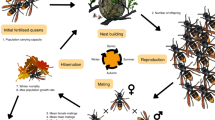
(Adapted from (Rabeling and Kronauer 2013))
Similar content being viewed by others
References
Arca M, Mougel F, Guillemaud T et al (2015) Reconstructing the invasion and the demographic history of the yellow-legged hornet, Vespa velutina, in Europe. Biol Invas 17:2357–2371. https://doi.org/10.1007/s10530-015-0880-9
Aron S (2001) Reproductive strategy: an essential component in the success of incipient colonies of the invasive Argentine ant. Insectes Soc 48:25–27. https://doi.org/10.1007/PL00001740
Austin A, Dowton M (2000) Hymenoptera: evolution, biodiversity and biological control. Csiro Publishing, London
Avtzis DN, Matoševi D (2013) Taking Europe by storm: a first insight in the introduction and expansion of Dryocosmus kuriphilus in central Europe by mtDNA. Šumar List 8
Baker H (1955) Self-compatibility and establishment after “long-distance” dispersal. Evolution 9:347–349
Baker H (1965) Characteristics and modes of origin of weeds. Genetics of colonizing species. Academic Press, New York, pp 137–172
Baker H (1967) Support for Baker’s law as a rule. Evolution 21:853–856
Baudry E, Kryger P, Allsopp M et al (2004) Whole-genome scan in thelytokous-laying workers of the cape honeybee (Apis mellifera capensis): central fusion, reduced recombination rates and centromere mapping using half-tetrad analysis. Genetics 167:243–252. https://doi.org/10.1534/genetics.167.1.243
Boissin E, Hurley B, Wingfield MJ et al (2012) Retracing the routes of introduction of invasive species: the case of the Sirex noctilio woodwasp. Mol Ecol 21:5728–5744. https://doi.org/10.1111/mec.12065
Bompard A, Amat I, Fauvergue X, Spataro T (2013) Host-parasitoid dynamics and the success of biological control when parasitoids are prone to Allee effects. PLoS ONE 8:e76768. https://doi.org/10.1371/journal.pone.0076768
Bono JM, Herbers JM (2003) Proximate and ultimate control of sex ratios in Myrmica brevispinosa colonies. Proc R Soc Lond B Biol Sci 270:811–817
Brown JL (1997) A theory of mate choice based on heterozygosity. Behav Ecol 8:60–65. https://doi.org/10.1093/beheco/8.1.60
CABI (2019a) Apis mellifera scutellata (africanized bee). Invasive Species Compendium. CAB International, Wallingford, UK
CABI (2019b) Leptocybe invasa (blue gum chalcid). Invasive Species Compendium. CAB International, Wallingford, UK
CABI (2019c) Aphidius ervi. Invasive Species Compendium. CAB International, Wallingford, UK
Cameron PJ, Walker GP (1989) Release and establishment of Aphidius spp. (Hymenoptera: Aphidiidae), parasitoids of pea aphid and blue green aphid in New Zealand. N Z J Agric Res 32:281–290. https://doi.org/10.1080/00288233.1989.10423463
Chapman TW, Stewart SC (1996) Extremely high levels of inbreeding in a natural population of the free-living wasp Ancistrocerus antliope (Hymenoptera: Vespidae: Eumeninae). Heredity 76:65–69
Charnov EL (1982) The theory of sex allocation. Princeton University Press, Princeton
Charnov EL, Skinner SW (1984) Evolution of host selection and clutch size in parasitoid wasps. Fla Entomol 67:5. https://doi.org/10.2307/3494101
Chen HY, Yao JM, Xu ZF (2009) First description of the male of Leptocybe invasa Fisher & La Salle (Hymenoptera: Eulophidae) from China. J Environ Entomol 31:285–287
Cheptou P-O (2012) Clarifying Baker’s law. Ann Bot 109:633–641. https://doi.org/10.1093/aob/mcr127
Cordona C, Oatman E (1975) Biology and physical ecology of Apanteles subandinus Blanchard (Hymenoptera: Braconidae), with notes on temperature responses of Apanteles scutellaris Muesebeck and its host, the potato tuberworm. Hilgardia 43:1–51
Courchamp F, Clutton-Brock T, Grenfell B (1999) Inverse density dependence and the Allee effect. Trends Ecol Evol 14:405–410. https://doi.org/10.1016/S0169-5347(99)01683-3
Courtois P, Figuieres C, Mulier C, Weill J (2018) A cost-benefit approach for prioritizing invasive species. Ecol Econ 146:607–620. https://doi.org/10.1016/j.ecolecon.2017.11.037
Craig SF, Slobodkin LB, Wray GA, Biermann CH (1997) The ‘paradox’of polyembryony: a review of the cases and a hypothesis for its evolution. Evol Ecol 11:127–143
Craig TP, Price PW, Itami JK (1992) Facultative sex ratio shifts by a herbivorous insect in response to variation in host plant quality. Oecologia 92:153–161
Darrouzet E, Gévar J, Guignard Q, Aron S (2015) Production of early diploid males by European colonies of the invasive hornet Vespa velutina nigrithorax. PLoS ONE 10:e0136680. https://doi.org/10.1371/journal.pone.0136680
Darvill B, Lye GC, Goulson D (2007) Aggregations of male Bombus muscorum (Hymenoptera: Apidae) at mature nests. Incestuous brothers or amorous suitors? Apidologie 38:518–524. https://doi.org/10.1051/apido:2007032
Deredec A, Courchamp F (2003) Extinction thresholds in host—parasite dynamics. Ann Zool Fenn 40:115–130
Dittrich-Schröder G, Hoareau TB, Hurley BP et al (2018) Population genetic analyses of complex global insect invasions in managed landscapes: a Leptocybe invasa (Hymenoptera) case study. Biol Invasions 20:2395–2420. https://doi.org/10.1007/s10530-018-1709-0
Dittrich-Schröder G, Wingfield MJ, Hurley BP, Slippers B (2012) Diversity in Eucalyptus susceptibility to the gall-forming wasp Leptocybe invasa. Agric For Entomol 14:419–427. https://doi.org/10.1111/j.1461-9563.2012.00583.x
Dyson EA, Hurst GDD (2004) Persistence of an extreme sex-ratio bias in a natural population. Proc Natl Acad Sci 101:6520–6523. https://doi.org/10.1073/pnas.0304068101
Engelstädter J, Telschow A (2009) Cytoplasmic incompatibility and host population structure. Heredity 103:196–207. https://doi.org/10.1038/hdy.2009.53
Eschen R, Britton K, Brockerhoff E et al (2015) International variation in phytosanitary legislation and regulations governing importation of plants for planting. Environ Sci Policy 51:228–237. https://doi.org/10.1016/j.envsci.2015.04.021
Eyer P, Matsuura K, Vargo EL et al (2018) Inbreeding tolerance as a pre-adapted trait for invasion success in the invasive ant Brachyponera chinensis. Mol Ecol. https://doi.org/10.1111/mec.14910
FAO (2012) Forest pest species profiles - Leptocybe invasa, blue gum chalcid. Food and Agriculture Organization, Rome, Italy
Faria LRR, Soares EDG, do Carmo E, de Oliveira PMC (2016) Diploid male dynamics under different numbers of sexual alleles and male dispersal abilities. Theory Biosci 135:111–119. https://doi.org/10.1007/s12064-016-0226-x
Fauvergue X, Hopper KR (2009) French wasps in the New World: experimental biological control introductions reveal a demographic Allee effect. Popul Ecol 51:385–397. https://doi.org/10.1007/s10144-009-0147-3
Ferguson C, Moeed A, Barratt B, et al (2007) BCANZ-Biological control agents introduced to New Zealand. http://www.b3nz.org/bcanz. Accessed 3 Jul 2019
Fialho RF, Stevens L (2000) Male-killing Wolbachia in a flour beetle. Proc R Soc Lond B Biol Sci 267:1469–1473. https://doi.org/10.1098/rspb.2000.1166
Folliot R (1964) Contribution à l’étude de la biologie des cynipides gallicoles (Hymenoptera : Cynipoidea). Ann Sci Nat-Zool 6:407–564
Frankham R (2005) Resolving the genetic paradox in invasive species. Heredity 94:385–385. https://doi.org/10.1038/sj.hdy.6800634
Galindo-Cardona A, Acevedo-Gonzalez JP, Rivera-Marchand B, Giray T (2013) Genetic structure of the gentle Africanized honey bee population (gAHB) in Puerto Rico. BMC Genet 14:65
Garnas JR, Auger-Rozenberg M-A, Roques A et al (2016) Complex patterns of global spread in invasive insects: eco-evolutionary and management consequences. Biol Invasions 18:935–952. https://doi.org/10.1007/s10530-016-1082-9
Gebiola M, Kelly SE, Hammerstein P et al (2016) “Darwin’s corollary” and cytoplasmic incompatibility induced by Cardinium may contribute to speciation in Encarsia wasps (Hymenoptera: Aphelinidae). Evolution 70:2447–2458. https://doi.org/10.1111/evo.13037
Gerrish PJ, Lenski RE (1998) The fate of competing beneficial mutations in an asexual population. Genetica 102:127
Gillespie J (1998) Population genetics: a concise guide. Johns Hopkins University Press, Baltimore
Giorgini M, Bernardo U, Monti MM et al (2010) Rickettsia symbionts cause parthenogenetic reproduction in the parasitoid wasp Pnigalio soemius (Hymenoptera: Eulophidae). Appl Environ Microbiol 76:2589–2599. https://doi.org/10.1128/AEM.03154-09
Giorgini M, Monti MM, Caprio E et al (2009) Feminization and the collapse of haplodiploidy in an asexual parasitoid wasp harboring the bacterial symbiont Cardinium. Heredity 102:365–371. https://doi.org/10.1038/hdy.2008.135
Gloag R, Ding G, Christie JR et al (2016) An invasive social insect overcomes genetic load at the sex locus. Nat Ecol Evol 1:0011. https://doi.org/10.1038/s41559-016-0011
Godfray HCJ (1990) The causes and consequences of constrained sex allocation in haplodiploid animals. J Evol Biol 3:3–17
Goud KB, Kumari HK, Vastrad AS et al (2010) Screening of eucalyptus genotypes against gall wasp, Leptocybe invasa Fisher and La Salle (Hymenoptera: Eulophidae). Karnataka J Agric Sci 23:213–214
Hails RS, Crawley MJ (1991) The population dynamics of an alien insect: Andricus quercuscalicis (Hymenoptera: Cynipidae). J Anim Ecol 60:545–561. https://doi.org/10.2307/5297
Hajek AE (2018) Natural enemies: an introduction to biological control, 2nd edn. Cambridge University Press, Cambridge
Hall HG (1990) Parental analysis of introgressive hybridization between African and European honeybees using nuclear DNA RFLPs. Genetics 125:611–621
Hamilton WD (1967) Extraordinary sex ratios. Science 156:477–488
Hardy ICW, Cook JM (1995) Brood sex ratio variance, developmental mortality and virginity in a gregarious parasitoid wasp. Oecologia 103:162–169. https://doi.org/10.1007/BF00329076
Harpur BA, Minaei S, Kent CF, Zayed A (2012) Management increases genetic diversity of honey bees via admixture. Mol Ecol 21:4414–4421. https://doi.org/10.1111/j.1365-294X.2012.05614.x
Hartfield M, Keightley PD (2012) Current hypotheses for the evolution of sex and recombination. Integr Zool 7:192–209. https://doi.org/10.1111/j.1749-4877.2012.00284.x
Hatcher MJ, Taneyhill DE, Dunn AM, Tofts C (1999) Population dynamics under parasitic sex ratio distortion. Theor Popul Biol 56:11–28. https://doi.org/10.1006/tpbi.1998.1410
He XZ, Wang Q, Teulon DAJ (2004) Emergence, sexual maturation and oviposition of Aphidius ervi (Hymenoptera: Aphidiidae). N Z Plant Prot 57:214
Henry LM, May N, Acheampong S et al (2010) Host-adapted parasitoids in biological control: Does source matter? Ecol Appl 20:242–250. https://doi.org/10.1890/08-1869.1
Henry LM, Roitberg BD, Gillespie DR (2008) Host-range evolution in Aphidius parasitoids: Fidelity, virulence and fitness trade-offs on an ancestral host. Evolution 62:689–699. https://doi.org/10.1111/j.1558-5646.2007.00316.x
Hofsvang T, Hågvar EB (1975) Duration of development and longevity in Aphidius ervi and Aphidius platensis [Hym.: Aphidiidae], two parasites of Myzus persicae [Hom.: Aphididae]. Entomophaga 20:11–22
Holway DA, Suarez AV (1999) Animal behavior: an essential component of invasion biology. Trends Ecol Evol 14:328–330
Hopper KR, Roush RT (1993) Mate finding, dispersal, number released, and the success of biological control introductions. Ecol Entomol 18:321–331
Huang Z-Y, Li J, Lu W et al (2018) Parasitoids of the eucalyptus gall wasp Leptocybe spp.: a global review. Environ Sci Pollut Res 25:29983–29995. https://doi.org/10.1007/s11356-018-3073-0
Hufbauer RA (2001) Pea aphid-parasitoid interactions: Have parasitoids adapted to differential resistance? Ecology 82:717. https://doi.org/10.2307/2680191
Hufbauer RA, Bogdanowicz SM, Harrison RG (2004) The population genetics of a biological control introduction: mitochondrial DNA and microsatellie variation in native and introduced populations of Aphidus ervi, a parisitoid wasp. Mol Ecol 13:337–348. https://doi.org/10.1046/j.1365-294X.2003.02084.x
Hurst G, Jiggins FM (2000) Male-killing bacteria in insects: mechanisms, incidence, and implications. Emerg Infect Dis 6:329–336. https://doi.org/10.3201/eid0604.000402
Kageyama D, Hoshizaki S, Ishikawa Y (1998) Female-biased sex ratio in the Asian corn borer. Ostrinia furnacalis: evidence for the occurrence of feminizing bacteria in an insect. 81:311–316
Kent RB (1988) The introduction and diffusion of the African honeybee in South America. Yearb Assoc Pac Coast Geogr 50:21–43. https://doi.org/10.1353/pcg.1988.0009
Kerr WE (1957) Introduçao de abelhas africanas no Brasil. Bras Apic 3:211–213
Kfir K (1981) Fertility of the polyembryonic parasite Copidosoma koehleri, effect of humidities on life length and relative abundance as compared with that of Apanteles subandinus in potato tuber moth. Ann Appl Biol 99:225–230
Koeniger N, Koeniger G (2000) Reproductive isolation among species of the genus Apis. Apidologie 31:313–339. https://doi.org/10.1051/apido:2000125
Kraus FB, Neumann P, van Praagh J, Moritz RFA (2004) Sperm limitation and the evolution of extreme polyandry in honeybees (Apis mellifera L.). Behav Ecol Sociobiol 55:494–501. https://doi.org/10.1007/s00265-003-0706-0
Kronauer DJC, Pierce NE, Keller L (2012) Asexual reproduction in introduced and native populations of the ant Cerapachys biroi. Mol Ecol 21:5221–5235. https://doi.org/10.1111/mec.12041
Kulkarni H, Kumari NK, Vastrad AS, Basavanagoud K (2010) Release and recovery of parasitoids in eucalyptus against gall wasp, Leptocybe invasa (Hymenoptyera: Eulophidae) under green house. Karnataka J Agric Sci 23:91–92
Lehtonen J, Jennions MD, Kokko H (2012) The many costs of sex. Trends Ecol Evol 27:172–178. https://doi.org/10.1016/j.tree.2011.09.016
Liebhold A, Bascompte J (2003) The Allee effect, stochastic dynamics and the eradication of alien species. Ecol Lett 6:133–140. https://doi.org/10.1046/j.1461-0248.2003.00405.x
Lockwood JL, Cassey P, Blackburn T (2005) The role of propagule pressure in explaining species invasions. Trends Ecol Evol 20:223–228. https://doi.org/10.1016/j.tree.2005.02.004
Lombaert E, Guillemaud T, Cornuet J-M et al (2010) Bridgehead effect in the worldwide invasion of the biocontrol harlequin ladybird. PLoS ONE 5:e9743
Maatouf N, Lumaret J-P (2012) Eco-éthologie des nouveaux ravageurs invasifs des eucalyptus du Maroc. Ann Société Entomol Fr 48:289–297. https://doi.org/10.1080/00379271.2012.10697778
MacQuarrie CJK, Langor DW, Sperling FAH (2007) Mitochondrial DNA variation in two invasive birch leaf-mining sawflies in North America. Can Entomol 139:545–553. https://doi.org/10.4039/n06-084
Martinez-Sañudo I, Mazzon L, Simonato M et al (2019) Tracking the origin and dispersal of the Asian chestnut gall wasp Dryocosmus kuriphilus Yasumatsu (Hymenoptera, Cynipidae) in Europe with molecular markers. Bull Entomol Res 109:300–308. https://doi.org/10.1017/S000748531800069X
Matalon Y, Katzir N, Gottlieb Y et al (2007) Cardinium in Plagiomerus diaspidis (Hymenoptera: Encyrtidae). J Invertebr Pathol 96:106–108. https://doi.org/10.1016/j.jip.2007.02.010
Mateo Leach I, Ferber S, van de Zande L, Beukeboom LW (2012) Genetic variability of arrhenotokous and thelytokous Venturia canescens (Hymenoptera). Genetica 140:53–63. https://doi.org/10.1007/s10709-012-9657-6
Mattila HR, Reeve HK, Smith ML (2012) Promiscuous honey bee queens increase colony productivity by suppressing worker selfishness. Curr Biol 22:2027–2031. https://doi.org/10.1016/j.cub.2012.08.021
Maynard Smith J (1978) The evolution of sex. Cambridge University Press, Cambridge
McGeoch MA, Genovesi P, Bellingham PJ et al (2016) Prioritizing species, pathways, and sites to achieve conservation targets for biological invasion. Biol Invasions 18:299–314. https://doi.org/10.1007/s10530-015-1013-1
McNally LC, Schneider SS (1992) Seasonal cycles of growth, development and movement of the African honey bee, Apis mellifera scutettata, in Africa. Insectes Soc 39:167–179
McNally LC, Schneider SS (1996) Spatial distribution and nesting biology of colonies of the African honey bee Apis mellifera scutellata (Hymenoptera: Apidae) in Botswana, Africa. Environ Entomol 25:643–652. https://doi.org/10.1093/ee/25.3.643
Mendel Z, Protasov A, Fisher N, La Salle J (2004) Taxonomy and biology of Leptocybe invasa gen. & sp. n. (Hymenoptera: Eulophidae), an invasive gall inducer on Eucalyptus. Aust J Entomol 43:101–113. https://doi.org/10.1111/j.1440-6055.2003.00393.x
Milne WM (1986) The release and establishment of Aphidius ervi Haliday (Hymenoptera: Ichneumonoidea) in lucerne aphids in eastern Australia. Austral Entomol 25:123–130
Miura K, Tagami Y (2004) Comparison of life history characters of arrhenotokous and Wolbachia-associated thelytokous Trichogramma kaykai Pinto and Stouthamer (Hymenoptera: Trichogrammatidae). Ann Entomol Soc Am 97:765–769. https://doi.org/10.1603/0013-8746(2004)097%5b0765:COLHCO%5d2.0.CO;2
Mochiah MB, Ngi-Song AJ, Overholt WA, Stouthamer R (2002) Wolbachia infection in Cotesia sesamiae (Hymenoptera: Braconidae) causes cytoplasmic incompatibility: implications for biological control. Biol Control 25:74–80. https://doi.org/10.1016/S1049-9644(02)00045-2
Muller HJ (1932) Some genetic aspects of sex. Am Nat 66:118–138
Niemelä P, Mattson WJ (1996) Invasion of North American forests by European phytophagous insects. Bioscience 46:741–753. https://doi.org/10.2307/1312850
Nonacs P (2006) Interspecific hybridization in ants: at the intersection of ecology, evolution, and behavior. Ecology 87:2143–2147
North A, Pennanen J, Ovaskainen O, Laine A-L (2011) Local adaptation in a changing world: the role of gene-flow, mutation and sexual reproduction. Evolution 65:79–89. https://doi.org/10.1111/j.1558-5646.2010.01107.x
Nyamukondiwa C, Kleynhans E, Terblanche JS (2010) Phenotypic plasticity of thermal tolerance contributes to the invasion potential of Mediterranean fruit flies (Ceratitis capitata). Ecol Entomol 35:565–575. https://doi.org/10.1111/j.1365-2311.2010.01215.x
Nyeko P, Mutitu EK, Day RK (2007) Farmers’ knowledge, perceptions and management of the gall-forming wasp, Leptocybe invasa (Hymenoptera: Eulophidae), on Eucalyptus species in Uganda. Int J Pest Manag 53:111–119. https://doi.org/10.1080/09670870601185230
Oldroyd BP, Fewell JH (2007) Genetic diversity promotes homeostasis in insect colonies. Trends Ecol Evol 22:408–413. https://doi.org/10.1016/j.tree.2007.06.001
Orlova-Bienkowskaja M, Bienkowski A, Karpun NN (2018) Plant quarantine alarm: as much as 20 new alien insect pest species including Drosophila suzukii appeared in the Caucasus in the last seven years. BioRxiv
Otto SP, Lenormand T (2002) Resolving the paradox of sex and recombination. Nat Rev Genet 3:252–261. https://doi.org/10.1038/nrg761
Page RE (1980) The evolution of multiple mating behavior by honey bee queens (Apis mellifera L.). Genetics 96:263–273
Pannebakker BA, Zwaan BJ, Beukeboom LW, Van Alphen JJM (2004) Genetic diversity and Wolbachia infection of the Drosophila parasitoid Leptopilina clavipes in western Europe. Mol Ecol 13:1119–1128. https://doi.org/10.1111/j.1365-294X.2004.02147.x
Pearcy M, Goodisman MAD, Keller L (2011) Sib mating without inbreeding in the longhorn crazy ant. Proc R Soc B Biol Sci 278:2677–2681. https://doi.org/10.1098/rspb.2010.2562
Pearcy M, Hardy O, Aron S (2006) Thelytokous parthenogenesis and its consequences on inbreeding in an ant. Heredity 96:377–382. https://doi.org/10.1038/sj.hdy.6800813
Peterson AT (2003) Predicting the geography of species’ invasions via ecological niche modeling. Q Rev Biol 78:419–433
Pinto MA (2005) Africanization in the United States: replacement of feral European honeybees (Apis mellifera L.) by an African hybrid swarm. Genetics 170:1653–1665. https://doi.org/10.1534/genetics.104.035030
Plantard O, Rasplus J-Y, Mondor G et al (1998) Wolbachia–induced thelytoky in the rose gallwasp Diplolepis spinosissimae (Giraud) (Hymenoptera: Cynipidae), and its consequences on the genetic structure of its host. Proc R Soc Lond B Biol Sci 265:1075–1080
Poorjavad N, Goldansaz SH, Leeuwen TV (2018) Fertility life table parameters, COI sequences and Wolbachia infection in populations of Trichogramma brassicae collected from Chilo suppressalis. Bull Insectology 71:89–96
Powell W, Wright AF (1988) The abilities of the aphid parasitoids Aphidius ervi Haliday and A. rhopalosiphi De Stefani Perez (Hymenoptera: Braconidae) to transfer between different known host species and the implications for the use of alternative hosts in pest control strategies. Bull Entomol Res 78:683–693
Prasanth Jacob J, Senthil K, Sivakumar V et al (2015) Gall wasp Leptocybe invasa (Hymenoptera: Eulophidae) management in eucalypts. J Biol Control 29:20. https://doi.org/10.18641/jbc/29/1/75789
Pucci C, Spanedda AF, Minutoli E (2003) Field study of parasitism caused by endemic parasitoids and by the exotic parasitoid Copidosoma koehleri on Phthorimaea operculella in Central Italy. Bull Insectology 56:221–224
Pusey A, Wolf M (1996) Inbreeding avoidance in animals. Trends Ecol Evol 11:201–206
Quang Thu P, Dell B, Burgess TI (2009) Susceptibility of 18 eucalypt species to the gall wasp Leptocybe invasa in the nursery and young plantations in Vietnam. ScienceAsia 35:113–117
Rabeling C, Kronauer DJC (2013) Thelytokous parthenogenesis in eusocial Hymenoptera. Annu Rev Entomol 58:273–292. https://doi.org/10.1146/annurev-ento-120811-153710
Rankin DJ, Kokko H (2007) Do males matter? The role of males in population dynamics. Oikos 116:335–348. https://doi.org/10.1111/j.0030-1299.2007.15451.x
Ross KG, Fletcher DJC (1986) Diploid male production: a significant colony mortality factor in the fire ant Solenopsis invicta (Hymenoptera: Formicidae). Behav Ecol Sociobiol 19:283–291. https://doi.org/10.1007/BF00300643
Ruf D, Dorn S, Mazzi D (2012) Unexpectedly low frequencies of diploid males in an inbreeding parasitoid with complementary sex determination. Biol J Linn Soc 108:79–86
Sakai AK, Allendorf FW, Holt JS et al (2001) The population biology of invasive species. Annu Rev Ecol Syst 32:305–332. https://doi.org/10.1146/annurev.ecolsys.32.081501.114037
Salin C, Deprez B, Van Bockstaele DR et al (2004) Sex determination mechanism in the hymenopteran parasitoid Aphidius rhopalosiphi De Stefani-Peres (Braconidae: Aphidiinae). Belg J Zool 134:15–22
Schilthuizen M, Stouthamer R (1997) Horizontal transmission of parthenogenesis-inducing microbes in Trichogramma wasps. Proc R Soc B Biol Sci 264:361–366
Schneider MV, Beukeboom LW, Driessen G et al (2002) Geographical distribution and genetic relatedness of sympatrical thelytokous and arrhenotokous populations of the parasitoid Venturia canescens (Hymenoptera). J Evol Biol 15:191–200
Schneider MV, Driessen G, Beukeboom LW et al (2003) Gene flow between arrhenotokous and thelytokous populations of Venturia canescens (Hymenoptera). Heredity 90:260–267. https://doi.org/10.1038/sj.hdy.6800245
Schneider SS, DeGrandi-Hoffman G, Smith D (2004) The African honeybee: factors contributing to a successful biological invasion. Annu Rev Entomol 49:351–376
Sheppard WS, Rinderer TE, Mazzoli JA et al (1991) Gene flow between African- and European-derived honey bee populations in Argentina. Nature 349:782–784
Skinner SW (1982) Maternally inherited sex ratio in the parasitoid wasp Nasonia vitripennis. Science 215:1133–1134. https://doi.org/10.1126/science.215.4536.1133
Slatyer RA, Mautz BS, Backwell PRY, Jennions MD (2012) Estimating genetic benefits of polyandry from experimental studies: a meta-analysis. Biol Rev 87:1–33. https://doi.org/10.1111/j.1469-185X.2011.00182.x
Smith DR, Taylor O, Brown W (1989) Neotropical africanized honey bees have African mitochondrial DNA. Nature 339:213–215
Starý P (2002) Field establishment of Aphidius colemani Vier. (Hym., Braconidae, Aphidiinae) in the Czech Republic. J Appl Entomol 126:405–408. https://doi.org/10.1046/j.1439-0418.2002.00663.x
Starỳ P (1974) Taxonomy, origin, distribution and host range of Aphidius species (Hym., Aphidiidae) in relation to biological control of the pea aphid in Europe and North America. J Appl Entomol 77:141–171
Stebbins G (1957) Self fertilization and population variability in the higher plants. Am Nat 91:337–354
Stone GN, Atkinson RJ, Rokas A et al (2007) Evidence for widespread cryptic sexual generations in apparently purely asexual Andricus gallwasps. Mol Ecol 17:652–665. https://doi.org/10.1111/j.1365-294X.2007.03573.x
Stone GN, Schönrogge K, Atkinson RJ et al (2002) The population biology of oak gall wasps (Hymenotera: Cynipidae). Annu Rev Entomol 47:633–668
Stouthamer R (1993) The use of sexual versus asexual wasps in biological control. Entomophaga 38:3–6
Stouthamer R, Luck RF (1993) Influence of microbe-associated parthenogenesis on the fecundity of Trichogramma deion and T. pretiosum. Entomol Exp Appl 67:183–192
Suarez AV, Tsutsui ND (2008) The evolutionary consequences of biological invasions. Mol Ecol 17:351–360. https://doi.org/10.1111/j.1365-294X.2007.03456.x
Suckling DM, Tobin PC, McCullough DG, Herms DA (2012) Combining tactics to exploit Allee effects for eradication of alien insect populations. J Econ Entomol 105:1–13. https://doi.org/10.1603/EC11293
Sundström L (1993) Genetic population structure and sociogenetic organisation in Formica truncorum (Hymenoptera; Formicidae). Behav Ecol Sociobiol. https://doi.org/10.1007/BF00172934
Takada H (1968) Aphidiidae of Japan. Insecta Matsumurana 30:67–124
Takada H, Tada E (2000) A comparison between two strains from Japan and Europe of Aphidius ervi. Entomol Exp Appl 97:11–20
Tarpy DR, Page RE (2001) The curious promiscuity of queen honey bees (Apis mellifera): evolutionary and behavioral mechanisms. Ann Zool 38:11
Taylor CM, Hastings A (2005) Allee effects in biological invasions. Ecol Lett 8:895–908. https://doi.org/10.1111/j.1461-0248.2005.00787.x
Thiel A, Weeda AC, De Boer JG, Hoffmeister TS (2013) Genetic incompatibility drives mate choice in a parasitic wasp. Front Zool 10:43
Tomasetto F, Tylianakis JM, Reale M et al (2017) Intensified agriculture favors evolved resistance to biological control. Proc Natl Acad Sci 114:3885–3890. https://doi.org/10.1073/pnas.1618416114
Tougeron K, van Baaren J, Llopis S et al (2018) Disentangling plasticity from local adaptation in diapause expression in parasitoid wasps from contrasting thermal environments: a reciprocal translocation experiment. Biol J Linn Soc 124:756–764
Tregenza T, Wedell N (2002) Polyandrous females avoid costs of inbreeding. Nature 415:71–73. https://doi.org/10.1038/415071a
Unruh T, Short R, Herard F et al (2003) Introduction and establishment of parasitoids for the biological control of the apple ermine moth, Yponomeuta malinellus (Lepidoptera: Yponomeutidae), in the Pacific Northwest. Biol Control 28:332–345. https://doi.org/10.1016/S1049-9644(03)00101-4
Van Driesche R, Lyon S, Sanderson J et al (2008) Greenhouse trials of Aphidius colemani (Hymenoptera: Braconidae) banker plants for control of aphids (Hemiptera: Aphididae) in greenhouse spring floral crops. Fla Entomol 91:588–591. https://doi.org/10.1653/0015-4040-91.4.583
Van Driesche RG, Hoddle M (1997) Should arthropod parasitoids and predators be subject to host range testing when used as biological control agents? Agric Hum Values 14:211–226
Vavre F, Fleury F, Varaldi J et al (2002) Infection polymorphism and cytoplasmic incompatibility in Hymenoptera-Wolbachia associations. Heredity 88:361–365. https://doi.org/10.1038/sj.hdy.6800063
Venette RC, Kriticos DJ, Magarey RD et al (2010) Pest risk maps for invasive alien species: a roadmap for improvement. Bioscience 60:349–362. https://doi.org/10.1525/bio.2010.60.5.5
Vrijenhoek RC, Parker ED Jr (2009) Geographical parthenogenesis: General purpose genotypes and frozen niche variation. Lost sex: the evolutionary biology of parthenogenesis. Springer, Dordrecht, pp 99–132
Wang Z, Smith SM (1996) Phenotypic differences between thelytokous and arrhenotokous Trichogramma minutum from Zeiraphera canadensis. Entomol Exp Appl 78:315–323. https://doi.org/10.1111/j.1570-7458.1996.tb00796.x
Ward DF, Stanley MC, Toft RJ et al (2008) Assessing the risk of invasive ants: a simple and flexible scorecard approach. Insectes Soc 55:360–363. https://doi.org/10.1007/s00040-008-1013-6
Watmough R, Broodryk S, Annecke D (1973) The establishment of two imported parasitoids of potato tuber moth [Phthorimaea operculellai] in South Africa. Entomophaga 18:237–249
Werren J, Skinner S, Huger A (1986) Male-killing bacteria in a parasitic wasp. Science 231:990–992. https://doi.org/10.1126/science.3945814
Wetterer JK (2008) Worldwide spread of the longhorn crazy ant, Paratrechina longicornis (Hymenoptera: Formicidae). Myrmecol News 11:137–149
Wetterer JK, Guénard B, Booher DB (2015) Geographic spread of Vollenhovia emeryi (Hymenoptera: Formicidae). Asian Myrmecol 7:105–112
Wetterer JK, Porter SD (2003) The little fire ant. Wasmannia auropunctata: distribution, impact and control. 41:41
Wiernasz DC, Perroni CL, Cole BJ (2004) Polyandry and fitness in the western harvester ant, Pogonomyrmex occidentalis. Mol Ecol 13:1601–1606. https://doi.org/10.1111/j.1365-294X.2004.02153.x
Wilson EE, Mullen LM, Holway DA (2009) Life history plasticity magnifies the ecological effects of a social wasp invasion. Proc Natl Acad Sci 106:12809–12813. https://doi.org/10.1073/pnas.0902979106
Wilson EO (1963) Social modification related to rareness in ant species. Evolution 17:249–253. https://doi.org/10.1111/j.1558-5646.1963.tb03274.x
Yamauchi K, Ogata K (1995) Social structure and reproductive systems of tramp versus endemic ants (Hymenoptera: Formicidae) of the Ryukyu Islands. Pac Sci 49:55–68
Zayed A, Packer L (2005) Complementary sex determination substantially increases extinction proneness of haplodiploid populations. Proc Natl Acad Sci USA 102:10742–10746
Zepeda-Paulo F, Lavandero B, Mahéo F et al (2015) Does sex-biased dispersal account for the lack of geographic and host-associated differentiation in introduced populations of an aphid parasitoid? Ecol Evol 5:2149–2161. https://doi.org/10.1002/ece3.1504
Zúñiga E, Van den Bosch R, Drea JJ, Gruber F (1986) Control biologico de los afidos (Hom. II. Obtencion, introduccion y cuarentena de depredatores y parasitoides, Aphididae) de los cereales en chile
Acknowledgements
We thank the Tree Protection Cooperative Programme (TPCP), Natural Resources Canada and the U.S. Department of Agriculture for funding.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Queffelec, J., Allison, J.D., Greeff, J.M. et al. Influence of reproductive biology on establishment capacity in introduced Hymenoptera species. Biol Invasions 23, 387–406 (2021). https://doi.org/10.1007/s10530-020-02375-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-020-02375-6