Abstract
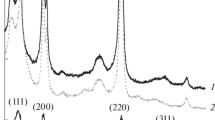
The structure of Pd–Ag/Al2O3 samples with different Ag/Pd ratios has been studied by a complex of physicochemical methods (H2-TPR, XRD, H2-TPD, TEM). Catalysts have been synthesized by supporting an active component on α-Al2O3 and γ-Al2O3. X-ray diffraction analysis has revealed the formation of a Pd–Ag substitutional solid solution with a face-centered cubic crystal lattice owing to alloying of the Pd and Ag components. An increase in the Ag content in the composition of bimetallic nanoparticles hinders the formation of palladium hydride (PdHx), which is completely suppressed at a ratio of Ag/Pd > 0.5. Infrared spectroscopy of adsorbed CO has revealed the formation of single-atom Pd1 sites on the surface of supported Pd–Ag bimetallic nanoparticles isolated from each other by silver atoms. Analysis of the structural stability of isolated Pd1 sites has shown that, under conditions of CO adsorption at 150°C, the stability of the sites can be provided by an increase in the Ag/Pd ratio to ≥2.







Similar content being viewed by others
REFERENCES
Blaser, H.-U., Schnyder, A., Steiner, H., Rössler, F., and Baumeister, P., Handbook of Heterogeneous Catalysis, Ertl, G., Knözinger, H., Scheth, F., and Weitkamp, J., Eds., Weinheim: Wiley, 2008, p. 3284.
Vile, G., Albani, D., Almora-Barrios, N., López, N., and Pérez-Ramírez, J., ChemCatChem, 2016, vol. 8, p. 21.
Liu, X., Mou, C.-Y., Lee, S., Li, Y., Secrest, J., and Jang, B.W.-L., J. Catal., 2012, vol. 285, p. 152.
Trimm, D.L., Cant, N.W., and Liu, I.O.Y., Catal. Today, 2011, vol. 178, p. 181.
Thomas, J.M., Raja, R., and Lewis, D.W., Angew. Chem., Int. Ed. 2005, vol. 44, p. 6456.
Liu, J., ACS Catal., 2017, vol. 7, p. 34.
Thomas, J.M., Phys. Chem. Chem. Phys., 2014, vol. 16, p. 7647.
Pelletier, J.D. and Basset, J.M., Acc. Chem. Res., 2016, vol. 49, p. 664.
Mitchell, S., Vorobyeva, E., and Perez-Ramirez, J., Angew. Chem., Int. Ed., 2018, vol. 57, p. 15316.
Yang, X.F., Wang, A., Qiao, B., Li, J., Liu, J., and Zhang, T., Acc. Chem. Res., 2013, vol. 46, p. 1740.
Wang, L., Huang, L., Liang, F., Liu, S., Wang, Y., and Zhang, H., Chin. J. Catal., 2017, vol. 38, p. 1528.
Qiao, B., Wang, A., Yang, X., Allard, L.F., Jiang, Z., Cui, Y., Liu, J., Li, J., and Zhang, T., Nat. Chem., 2011, vol. 3, p. 634.
Yang, M., Liu, J., Lee, S., Zugic, B., Huang, J., Allard, L.F., and Flytzani-Stephanopoulos, M.A., J. Am. Chem. Soc., 2015, vol. 137, p. 3470.
Hackett, S.F., Brydson, R.M., Gass, M.H., Harvey, I., Newman, A.D., Wilson, K., and Lee, A.F., Angew. Chem., Int. Ed., 2007, vol. 46, p. 8593.
Flytzani-Stephanopoulos, M., Chin. J. Catal., 2017, vol. 38, p. 1432.
Gates, B.C., Flytzani-Stephanopoulos, M., Dixon, D.A., and Katz, A., Catal. Sci. Technol., 2017, vol. 7, p. 4259.
Kyriakou, G., Boucher, M.B., Jewell, A.D., Lewis, E.A., Lawton, T.J., Baber, A.E., Tierney, H.L., Flytzani-Stephanopoulos, M., and Sykes, E.C., Science, 2012, vol. 335, p. 1209.
Zhang, L., Wang, A., Miller, J.T., Liu, X., Yang, X., Wang, W., Li, L., Huang, Y., Mou, C.-Y., and Zhang, T., ACS Catal., 2014, vol. 4, p. 1546.
Jin, Y., Datye, A.K., Rightor, E., Gulotty, R., Waterman, W., Smith, M., Holbrook, M., Maj, J., and Blackson, J., J. Catal., 2001, vol. 203, p. 292.
Nikolaev, S.A., Zanaveskin, L.N., Smirnov, V.V., Aver’yanov, V.A., and Zanaveskin, K.L., Russ. Chem. Rev., 2009, vol. 78, p. 231.
Teschner, D., Borsodi, J., Wootsch, A., Revay, Z., Havecker, M., Knop-Gericke, A., Jackson, S.D., and Schlogl, R., Science, 2008, vol. 320, p. 86.
Ellert, O.G., Tsodikov, M.V., Nikolaev, S.A., and Novotortsev, V.M., Russ. Chem. Rev., 2014, vol. 83, p. 718.
Furukawa, S. and Komatsu, T., ACS Catal., 2016, vol. 7, p. 735.
Föttinger, K., Catalysis, 2013, vol. 25, p. 77.
Furukawa, S., Endo, M., and Komatsu, T., ACS Catal., 2014, vol. 4, p. 3533.
Furukawa, S. and Komatsu, T., ACS Catal., 2016, vol. 6, p. 2121.
Wencka, M., Hahne, M., Kocjan, A., Vrtnik, S., Koželj, P., Korže, D., Jagličić, Z., Sorić, M., Popčević, P., Ivkov, J., Smontara, A., Gille, P., Jurga, S., Tomeš, P., Paschen, S., Ormeci, A., Armbrüster, M., Grin, Y., and Dolinšek, J., Intermetallics, 2014, vol. 55, p. 56.
Kovnir, K., Osswald, J., Armbrüster, M., Teschner, D., Weinberg, G., Wild, U., Knop-Gericke, A., Ressler, T., Grin, Y., and Schlögl, R., J. Catal., 2009, vol. 264, p. 93.
Feng, Q., Zhao, S., Wang, Y., Dong, J., Chen, W., He, D., Wang, D., Yang, J., Zhu, Y., Zhu, H., Gu, L., Li, Z., Liu, Y., Yu, R., Li, J., and Li, Y., J. Am. Chem. Soc., 2017, vol. 139, p. 7294.
Kovnir, K., Armbrüster, M., Teschner, D., Venkov, T.V., Jentoft, F.C., Knop-Gericke, A., Grin, Y., and Schlögl, R., Sci. Technol. Adv. Mater., 2007, vol. 8, p. 420.
Kovnir, K., Armbruster, M., Teschner, D., Venkov, T.V., Jentoft, F.C., Knop-Gericke, A., Grin, Y., and Schlogl, R., J. Catal., 2008, vol. 258, p. 219.
Pei, G.X., Liu, X.Y., Wang, A., Lee, A.F., Isaacs, M.A., Li, L., Pan, X., Yang, X., Wang, X., Tai, Z., Wilson, K., and Zhang, T., ACS Catal., 2015, vol. 5, p. 3717.
Zafeiratos, S., Piccinin, S., and Teschner, D., Catal. Sci. Technol., 2012, vol. 2, p. 1787.
McCue, A.J. and Anderson, J.A., J. Catal., 2015, vol. 329, p. 538.
Stakheev, A.Yu., Smirnova, N.S., Markov, P.V., Baeva, G.N., Bragina, G.O., Rassolov, A.V., and Mashkovsky, I.S., Kinet. Catal., 2018, vol. 59, p. 310.
Kachala, V.V., Khemchyan, L.L., Kashin, A.S., Orlov, N.V., Grachev, A.A., Zalesskiy, S.S., and Ananikov, V.P., Russ. Chem. Rev., 2013, vol. 82, p. 648.
Izumi, F. and Momma, K., Solid State Phenom., 2007, vol. 130, p. 15.
Iwasa, N. and Takezawa, N., Top. Catal., 2003, vol. 22, p. 215.
Fagherazzi, G., Benedetti, A., Polizzi, S., Di Mario, A., Pinna, F., Signoretto, M., and Pernicone, N., Catal. Lett., 1995, vol. 32, p. 293.
Gu, J., Wang, S., He, Z., Han, Y., and Zhang, J., Catal. Sci. Technol., 2016, vol. 6, p. 809.
Odoom-Wubah, T., Li, Q., Adilov, I., Huang, J., and Li, Q., Mol. Catal., 2019, vol. 477, p. 110558.
Kay Lup, A.N., Abnisa, F., Wan Daud, W.M.A., and Aroua, M.K., J. Chin. Chem. Soc., 2019, vol. 66, p. 1.
Rozanov, V.V. and Krylov, O.V., Russ. Chem. Rev., 1997, vol. 66, p. 117.
Satterfield, C.N., Heterogeneous Catalysis in Practice, New York: McGraw-Hill, 1980.
Li, Z., Wu, K., Cao, J., and Wang, Y., IOP Conf. Ser.: Mater. Sci. Eng., 2017, vol. 207, p. 012004.
Matori, K.A., Wah, L.C., Hashim, M., Ismail, I., and Zaid, M.H., Int. J. Mol. Sci, 2012, vol. 13, p. 16812.
Bondarchuk, I.S. and Mamontov, G.V., Kinet. Catal., 2015, vol. 56, p. 379.
Benipal, N., Qi, J., Liu, Q., and Li, W., Appl. Catal., B, 2017, vol. 210, p. 121.
Mitsudome, T., Urayama, T., Yamazaki, K., Maehara, Y., Yamasaki, J., Gohara, K., Maeno, Z., Mizugaki, T., Jitsukawa, K., and Kaneda, K., ACS Catal., 2016, vol. 6, p. 666.
Karakhanov, E.A., Maximov, A.L., Zolotukhina, A.V., Yatmanova, N.N., and Rosenberg, E., Appl. Organomet. Chem., 2015, vol. 29, p. 777.
Johansson, M., Skúlason, E., Nielsen, G., Murphy, S., Nielsen, R.M., and Chorkendorff, I., Surf. Sci., 2010, vol. 604, p. 718.
Lear, T., Marshall, R., Lopez-Sanchez, J.A., Jackson, S.D., Klapotke, T.M., Baumer, M., Rupprechter, G., Freund, H.J., and Lennon, D., J. Chem. Phys., 2005, vol. 123, p. 174706.
Cabilla, G.C., Bonivardi, A.L., and Baltanás, M.A., Catal. Lett., 1998, vol. 55, p. 147.
Primet, M., J. Catal., 1976, vol. 44, p. 324.
González, S., Neyman, K.M., Shaikhutdinov, Sh., Freund, H.-J., and Illas, F., J. Phys. Chem C, 2007, vol. 111, p. 6852.
Walle, L.E., Grönbeck, H., Fernandes, V.R., Blomberg, S., Farstad, M.H., Schulte, K., Gustafson, J., Andersen, J.N., Lundgren, E., and Borg, A., Surf. Sci., 2012, vol. 606, p. 1777.
Tang, J., Deng, L., Deng, H., Xiao, S., Zhang, X., and Hu, W., J. Phys. Chem. C, 2014, vol. 118, p. 27850.
Anderson, J.A., Fernández-García, M., and Haller, G.L., J. Catal., 1996, vol. 164, p. 477.
Svenum, I.H., Herron, J.A., Mavrikakis, M., and Venvik, H.J., Catal. Today, 2012, vol. 193, p. 111.
ACKNOWLEDGMENTS
The authors thank G.I. Kapustin for analyzing the samples by the H2-TPR. The electron microscopy studies of the samples were conducted by employees of the Department of Structural Studies of Zelinsky Institute of Organic Chemistry of the Russian Academy of Sciences.
Funding
This work was supported by the Russian Science Foundation (project no. 19-13-00285). The synthesis procedure for bimetallic Pd–Ag catalysts is based on the results obtained in studies supported by a grant of the Russian Science Foundation (no. 16-13-10530).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by M. Timoshinina
Abbreviations: H2-TPR, temperature-programmed reduction with hydrogen; XRD, X-ray diffraction analysis; H2-TPD, temperature-programmed desorption of hydrogen; TEM, transmission electron microscopy; ICP–AES, inductively coupled plasma atomic emission spectroscopy; fcc, face-centered cubic crystal lattice
Rights and permissions
About this article
Cite this article
Rassolov, A.V., Bragina, G.O., Baeva, G.N. et al. Formation of Isolated Single-Atom Pd1 Sites on the Surface of Pd–Ag/Al2O3 Bimetallic Catalysts. Kinet Catal 61, 758–767 (2020). https://doi.org/10.1134/S0023158420050080
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158420050080