Abstract
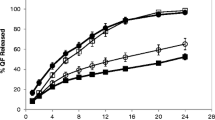
Dimenhydrinate is a therapeutic agent used for the treatment of nausea and vomiting. A conventional dose of dimenhydrinate is 50 mg per 8 h. Once-daily controlled release tablets of dimenhydrinate were formulated intended to treat emesis. The tablets were formulated using hydrophilic polymers Methocel® and hydrophobic polymers Ethocel® and subsequently analyzed for pre-compression and post-compression characteristics, % moisture uptake, dissolution profiles and release behavior of the drug. Identification of active ingredient and compatibility of blends were analyzed through FT-IR. Drug release kinetics of dimenhydrinate was assessed at pH 1.2, 4.5 and 6.8. The results of release profiles were evaluated for different kinetic models including model dependent and model independent. The optimized formulation K100M 30% showed ideal pre-compression properties and in vitro characteristics. Tablets exhibited a controlled drug release pattern due to the formation of a viscous gel layer followed by erosion. Furthermore, zero-order best described the release pattern with the value of r2 ˃ 0.99. Once-daily tablets released 98–100% drug in 24 h. Significantly more concentration and higher viscosity grade of hydrophilic polymer ensured the required pattern of drug release.







Similar content being viewed by others
References
Del Cuvillo A, Mullol J, Bartra J, Davila I, Jauregui I, Montoro J, Sastre J, Valero A (2006) Comparative pharmacology of the H1 antihistamines. J Investig Allergol Clin Immunol 16(1):3–12
Lerner E, Flashner-Barak M, v Achthoven E, Keegstra H, Smit R (2006) Delayed release formulations of 6-mercaptopurine. US Patent, US 20060008520A1
Hutton J, Morris J (1992) Long-acting carbidopa-levodopa in the management of moderate and advanced Parkinson's disease. Neurology 42(1 Suppl 1):51–56 discussion 57-60
Wagstaff AJ, Goa KL (2001) Once-weekly fluoxetine. Drugs 61(15):2221–2228
Perrie Y, Rades T (2009) Pharmaceutics: drug delivery and targeting. Pharmaceutical Press, PA
Aulton ME, Taylor KM (2017) Aulton's pharmaceutics e-book: the design and manufacture of medicines. Elsevier Health Sciences, Amsterdam
Nokhodchi A, Raja S, Patel P, Asare-Addo K (2012) The role of oral controlled release matrix tablets in drug delivery systems. Bioimpacts 2(4):175–187. https://doi.org/10.5681/bi.2012.027
Moore JW (1996) Mathematical comparison of dissolution profiles. Pharm Technol 20:64–75
O'hara T, Dunne A, Butler J, Devane J, Group ICW (1998) A review of methods used to compare dissolution profile data. Pharm Sci Technol Today 1(5):214–223
Shah VP, Tsong Y, Sathe P, Liu J-P (1998) In vitro dissolution profile comparison—statistics and analysis of the similarity factor, f2. Pharm Res 15(6):889–896
Siewert M, Dressman J, Brown CK, Shah VP, Aiache J-M, Aoyagi N, Bashaw D, Brown C, Brown W, Burgess D (2003) FIP/AAPS guidelines to dissolution/in vitro release testing of novel/special dosage forms. Aaps Pharmscitech 4(1):43–52
FDA U (1997) Guidance for industry: dissolution testing of immediate-release solid oral dosage forms. Food and Drug Administration Center for Drug Evaluation and Research (CDER), USA
Zhang Y, Huo M, Zhou J, Zou A, Li W, Yao C, Xie S (2010) DDSolver: an add-in program for modeling and comparison of drug dissolution profiles. AAPS J 12(3):263–271. https://doi.org/10.1208/s12248-010-9185-1
Patra C, Kumar A, Pandit H, Singh S, Devi M (2007) Design and evaluation of sustained release bilayer tablets of propranolol hydrochloride. Acta Pharm 57(4):479–489
The United States Pharmacopeia (2012) USP 35/The National Formulary, NF 30. US Pharmacopeial Convention, Rockville, MD
Commission BP (2016) British Pharmacopoeia 2017. Stationery Office, London
Ghayas S, Shoaib MH, Qazi F, Bushra R, Ali FR, Maboos M, Khalid F (2019) Influence of different viscosity grade cellulose-based polymers on the development of valsartan controlled release tablets. Polym Bull. https://doi.org/10.1007/s00289-019-02802-2
Costa P, Lobo JMS (2001) Modeling and comparison of dissolution profiles. Eur J Pharm Sci 13(2):123–133
Grdešič P, Vrečer F, Ilić I (2016) Flow and compaction properties of hypromellose: new directly compressible versus the established grades. Drug Dev Ind Pharm 42(11):1877–1886
Mehsud SU, Khan GM, Hussain A, Akram M, Akhlaq M, Khan KA, Shakoor A (2016) Controlled release matrix tablets of glipizide: influence of different grades of ethocel and Co-excipient on drug release. Pak J Pharm Sci 29(3):779–787
Hussain T, Saeed T, Mumtaz AM, Javaid Z, Abbas K, Awais A, Idrees HA (2013) Effect of two hydrophobic polymers on the release of gliclazide from their matrix tablets. ACTA Poloniae Pharm-Drug Res 70:749–757
Grund J, Koerber M, Walther M, Bodmeier R (2014) The effect of polymer properties on direct compression and drug release from water-insoluble controlled release matrix tablets. Int J Pharm 469(1):94–101
Shah RB, Tawakkul MA, Khan MA (2008) Comparative evaluation of flow for pharmaceutical powders and granules. Aaps Pharmscitech 9(1):250–258
Mathur V, Nagpal K, Singh SK, Mishra DN (2013) Comparative release profile of sustained release matrix tablets of verapamil HCl. Int J Pharm Investig 3(1):60
Wadher K, Kakde R, Umekar M (2011) Formulation and evaluation of a sustained-release tablets of metformin hydrochloride using hydrophilic synthetic and hydrophobic natural polymers. Indian J Pharm Sci 73(2):208
Sun CC, Hou H, Gao P, Ma C, Medina C, Alvarez FJ (2009) Development of a high drug load tablet formulation based on assessment of powder manufacturability: moving towards quality by design. J Pharm Sci 98(1):239–247
Patel P, Dave A, Vasava A, Patel P (2015) Formulation and characterization of sustained release dosage form of moisture sensitive drug. Int J pharm Investig 5(2):92
Ochoa L, Igartua M, Hernandez R, Gascon A, Pedraz J (2005) Preparation of sustained release hydrophilic matrices by melt granulation in a high-shear mixer. J Pharm Pharm Sci a Publ Canadian Soc Pharm Sci, Societe canadienne des sciences pharmaceutiques 8(2):132–140
Raghuvanshi S, Pathak K (2014) Recent advances in delivery systems and therapeutics of cinnarizine: a poorly water soluble drug with absorption window in stomach. J Drug Deliv 2014:479246
Wang Z, Shmeis RA (2006) Dissolution controlled drug delivery systems Design of controlled release drug delivery systems. McGraw-Hill, United States, pp 139–172
Klančar U, Horvat M, Baumgartner S (2012) Correlating cellulose derivative intrinsic viscosity with mechanical susceptibility of swollen hydrophilic matrix tablets. AAPS PharmSciTech 13(3):903–910
Klančar U, Markun B, Baumgartner S, Legen I (2013) A novel beads-based dissolution method for the in vitro evaluation of extended release HPMC matrix tablets and the correlation with the in vivo data. AAPS journal 15(1):267–277
Klančar U, Baumgartner S, Legen I, Smrdel P, Kampuš NJ, Krajcar D, Markun B, Kočevar K (2015) Determining the polymer threshold amount for achieving robust drug release from HPMC and HPC matrix tablets containing a high-dose BCS class I model drug: in vitro and in vivo studies. AAPS PharmSciTech 16(2):398–406
Li CL, Martini LG, Ford JL, Roberts M (2005) The use of hypromellose in oral drug delivery. J Pharm Pharmacol 57(5):533–546
Mallipeddi R (2009) The application of coarse particle ethyl cellulose and high molecular weight polyethylene oxide in the production of beads by extrusion-spheronization. University of the Sciences in Philadelphia, Pennsylvania
Tsong Y, Hammerstrom T, Sathe P, Shah VP (1996) Statistical assessment of mean differences between two dissolution data sets. Drug Inf J 30(4):1105–1112
Zhang H, Surian JM (2010) Biopharmaceutic consideration and assessment for oral controlled release formulations Oral Controlled Release Formulation Design and Drug Delivery Theory to Practice. Wiley, New Jersey, pp 33–45
Ritger PL, Peppas NA (1987) A simple equation for description of solute release I Fickian and non-fickian release from non-swellable devices in the form of slabs, spheres, cylinders or discs. J Controlled Release 5(1):23–36
Bae Y (2010) Drug delivery systems using polymer nanoassemblies for cancer treatment. Ther deliv 1(3):361–363
Fotaki N, Aivaliotis A, Butler J, Dressman J, Fischbach M, Hempenstall J, Klein S, Reppas C (2009) A comparative study of different release apparatus in generating in vitro–in vivo correlations for extended release formulations. Eur J Pharm Biopharm 73(1):115–120
Kostewicz ES, Abrahamsson B, Brewster M, Brouwers J, Butler J, Carlert S, Dickinson PA, Dressman J, Holm R, Klein S (2014) In vitro models for the prediction of in vivo performance of oral dosage forms. Eur J Pharm Sci 57:342–366
Shoaib MH, Siddiqi SAS, Yousuf RI, Zaheer K, Hanif M, Rehana S, Jabeen S (2010) Development and evaluation of hydrophilic colloid matrix of famotidine tablets. Aaps Pharmscitech 11(2):708–718
Yuksel N, Kanık AE, Baykara T (2000) Comparison of in vitro dissolution profiles by ANOVA-based, model-dependent and-independent methods. Int J Pharm 209(1–2):57–67
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zeb-un-Nisa, Shoaib, M.H., Yousuf, R.I. et al. Influence of different viscosity grade Methocel and Ethocel polymers for the development of controlled release dimenhydrinate matrix tablets. Polym. Bull. 78, 5525–5546 (2021). https://doi.org/10.1007/s00289-020-03369-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-020-03369-z