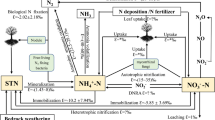
Abstract
Change in land-use practices can result in major shifts in the cycling of various elements, particularly nitrogen (N), which is prone to anthropogenic perturbations. For quantifying these shifts, accurate measurements of rates of biogeochemical transformations of N are needed. We used the (15N) isotope dilution technique to understand the effects of the types of forest alteration on (N) transformation rates by comparing gross N mineralization and ammonium (NH4+) consumption rates in soils of a managed forest, an unmanaged forest, and a rubber plantation in Kerala, India. Overall, nitrate (NO3−) dominated soils of the managed and unmanaged forests, whereas soils in the rubber plantation showed relatively higher NH4+ concentration. Total N (TN) and total organic carbon (TOC) concentrations were the highest under the rubber canopy (TN: 1.49 ± 0.02 mg N g−1; TOC: 7.96 ± 0.86 mg C g−1). In soils of all three forest types, gross N mineralization rates were higher compared to NH4+ consumption rates. Despite high TN and TOC concentrations, the rates of gross N mineralization and NH4+ consumption were considerably lower in the rubber plantation (mineralization: 1.08 ± 0.08 mg N kg−1 d−1; consumption: 0.85 ± 0.09 mg N kg−1 d−) compared to the managed (mineralization: 3.71 ± 0.35 mg N kg−1 d−1; consumption: 2.20 ± 1.41 mg N kg−1 d−1) and unmanaged (mineralization: 2.20 ± 1.07 mg N kg−1 d−1; consumption: 1.39 ± 0.27 mg N kg−1 d−1) forests. The lower NH4+ consumption rates in the rubber plantation led to significantly higher (p < 0.05) residence time of NH4+ (~ 4 days) compared to the managed and unmanaged forests (< 2 days), possibly contributing to acidification of rubber soils (pH ~ 4.8). These results together suggest that replacement of naturally grown forests with a mono-cropped plantation such as rubber negatively impact rates of N transformation processes in tropical soils and imply that change in tree species composition of naturally grown forests can adversely affect soil microbial activity. We recommend intercropping these plantations with commercial crops to maintain soil microbial diversity and biogeochemical cycling for sustainable forest management.






Similar content being viewed by others
References
Allen K, Corre MD, Tjoa A, Veldkamp E (2015) Soil nitrogen-cycling responses to conversion of lowland forests to oil palm and rubber plantations in Sumatra, Indonesia. PLoS ONE 10:e0133325. https://doi.org/10.1371/journal.pone.0133325
Asadiyan M, Hojjati SM, Pourmajidian MR, Fallah A (2013) Impact of land-use management on nitrogen transformation in a mountain forest ecosystem in the north of Iran. J For Res 24:115–119. https://doi.org/10.1007/s11676-012-0291-z
Bargali K, Manral V, Padalia K, Bargali SS, Upadhyay VP (2018) Effect of vegetation type and season on microbial biomass carbon in central himalayan forest soils, India. CATENA 171:125–135. https://doi.org/10.1016/j.catena.2018.07.001
Bargali SS, Singh RP, Joshi M (1993) Changes in soil characteristics in eucalypt plantations replacing natural broad-leaved forests. J Veg Sci 4:25–28. https://doi.org/10.2307/3235730
Booth MS, Stark JM, Rastetter E (2005) Controls on nitrogen cycling in terrestrial ecosystems: a synthetic analysis of literature data. Ecol Monogr 75:139–157. https://doi.org/10.1890/04-0988
Bünemann EK, Schwenke GD, Van Zwieten L (2006) Impact of agricultural inputs on soil organisms—a review. Soil Res 44:379–406
Butterbach-Bahl K, Gundersen P (2011) Nitrogen processes in terrestrial ecosystems. In: Sutton MA, Howard CM, Erisman JW, et al. (eds) The European nitrogen assessment. Cambridge University Press, Cambridge, pp 99–125
Choromanska U, De Luca TH (2002) Microbial activity and nitrogen mineralization in forest mineral soils following heating: evaluation of post-fire effects. Soil Biol Biochem 34:263–271. https://doi.org/10.1016/S0038-0717(01)00180-8
Davidson EA, Hart SC, Shanks CA, Firestone MK (1991) Measuring gross nitrogen mineralization, and nitrification by 15N isotopic pool dilution in intact soil cores. J Soil Sci 42:335–349. https://doi.org/10.1111/j.1365-2389.1991.tb00413.x
Dove MR (2003) The life-cycle of indigenous knowledge, and the case of natural rubber production. Indigenous environmental knowledge and its transformations: critical anthropological perspectives. Taylor & Francis, Amsterdam, pp 213–252
Drury CF, Hart SC, Yan XM (2008) Nitrification techniques for soils. In: Carter MR, Gregorich EG (eds) Soil sampling and methods of analysis, 2nd edn. Canadian Society of Soil Science, CRC Press Taylor and Francis Group, Boca Raton, pp 495–513
Fokom R, Adamou S, Teugwa MC, Begoude Boyogueno AD, Nana WL, Ngonkeu MEL, Tchameni NS, Nwaga D, Tsala Ndzomo G, Amvam Zollo PH (2012) Glomalin related soil protein, carbon, nitrogen and soil aggregate stability as affected by land use variation in the humid forest zone of south Cameroon. Soil Tillage Res 120:69–75. https://doi.org/10.1016/j.still.2011.11.004
Giovannini G, Poggio G, Sequi P (1975) Use of an automatic CHN analyzer to determine organic and inorganic carbon in soils. Commun Soil Sci Plant Anal 6:39–49. https://doi.org/10.1080/00103627509366544
Guimarães DV, Gonzaga MIS, da Silva TO, da Silva TL, da Silva DN, Matias MIS (2013) Soil organic matter pools and carbon fractions in soil under different land uses. Soil Tillage Res 126:177–182. https://doi.org/10.1016/j.still.2012.07.010
Haynes RJ, Naidu R (1998) Influence of lime, fertilizer and manure applications on soil organic matter content and soil physical conditions: a review. Nutr Cycl Agroecosyst 51:123–137. https://doi.org/10.1023/A:1009738307837
Hicks WK, Kuylenstierna JCI, Owen A, Dentener F, Seip HM, Rodhe H (2008) Soil sensitivity to acidification in Asia: status and prospects. AMBIO J Hum Environ 37:295–303. https://doi.org/10.1579/0044-7447(2008)37[295:SSTAIA]2.0.CO;2
Kellman L, Kumar S, Diochon A (2014) Soil nitrogen dynamics within profiles of a managed moist temperate forest chronosequence consistent with long-term harvesting-induced losses. J Geophys Res Biogeosci 119:1309–1321. https://doi.org/10.1002/2013JG002469
Kirkham D, Bartholomew WV (1954) Equations for following nutrient transformations in soil, utilizing tracer data1. Soil Sci Soc Am J 18:33–34. https://doi.org/10.2136/sssaj1954.03615995001800010009x
Kooch Y, Rostayee F, Hosseini SM (2016) Effects of tree species on topsoil properties and nitrogen cycling in natural forest and tree plantations of northern Iran. CATENA 144:65–73. https://doi.org/10.1016/j.catena.2016.05.002
Li D, Yang Y, Chen H, Xiao K, Song T, Wang K (2017) Soil gross nitrogen transformations in typical karst and non-karst forests, southwest China. J Geophys Res Biogeosci 122:2831–2840. https://doi.org/10.1002/2017JG003850
Li H, Ma Y, Liu W, Liu W (2012) Soil changes induced by rubber and tea plantation establishment: comparison with tropical rain forest soil in Xishuangbanna, SW China. Environ Manag 50:837–848. https://doi.org/10.1007/s00267-012-9942-2
Ma Z, Zhang X, Zhang C, Wang H, Chen F, Fu X, Fang X, Sun X, Lei Q (2018) Accumulation of residual soil microbial carbon in Chinese fir plantation soils after nitrogen and phosphorus additions. J For Res 29:953–962. https://doi.org/10.1007/s11676-017-0522-4
Mathew J (2006) The impact of new economic policy on Indian agriculture: a study of selected cash crops. Doctoral thesis, University of Calicut, Kerala, pp 1–15
Michopoulos P, Kaoukis K, Karetsos G, Grigoratos T, Samara C (2020) Nutrients in litterfall, forest floor and mineral soils in two adjacent forest ecosystems in Greece. J For Res 31(1):291–301. https://doi.org/10.1007/s11676-019-00952-7
Myrold DD, Tiedje JM (1986) Simultaneous estimation of several nitrogen cycle rates using 15N: theory and application. Soil Biol Biochem 18:559–568. https://doi.org/10.1016/0038-0717(86)90076-3
Nsabimana D, Klemedtson L, Kaplin BA, Wallin G (2008) Soil carbon and nutrient accumulation under forest plantations in southern Rwanda. Afr J Environ Sci Technol 2:142–149
Ollinger SV, Smith ML, Martin ME, Hallett RA, Goodale CL, Aber JD (2002) Regional variation in foliar chemistry and N cycling among forests of diverse history and composition. Ecology 83:339–355. https://doi.org/10.1890/0012-9658(2002)083[0339:RVIFCA]2.0.CO;2
Owen JS, Wang MK, Wang CH, King HB, Sun HL (2003) Net N mineralization and nitrification rates in a forested ecosystem in northeastern Taiwan. For Ecol Manag 176:519–530. https://doi.org/10.1016/S0378-1127(02)00225-6
Pandey CB, Singh GB, Singh SK, Singh RK (2010) Soil nitrogen and microbial biomass carbon dynamics in native forests and derived agricultural land uses in a humid tropical climate of India. Plant Soil 333:453–467. https://doi.org/10.1007/s11104-010-0362-x
Peng Y, Yang W, Yue K, Tan B, Wu F (2019) Impacts of soil fauna on nitrogen and phosphorus release during litter decomposition were differently controlled by plant species and ecosystem type. J For Res 30:921–930. https://doi.org/10.1007/s11676-018-0664-z
Philip A (2015) Soil characterization of rubber ecosystems with special reference to soil organic matter. Doctoral thesis, Mahatma Gandhi University, Kottayam, Kerala, pp 8–24
Rajasekharan P, Veeraputhran S (2002) Adoption of intercropping in rubber smallholdings in Kerala, India: a tobit analysis. Agrofor Syst 56:1–11. https://doi.org/10.1023/A:1021199928069
Rayment GE, Higginson FR (1992) Australian laboratory handbook of soil and water chemical methods, 3rd edn. Inkata Press, Melbourne, pp 33–39
Reddy CS, Jha CS, Dadhwal VK (2016) Assessment and monitoring of long-term forest cover changes (1920–2013) in Western Ghats biodiversity hotspot. J Earth Syst Sci 125:103–114
Rustad L, Campbell J, Marion G, Norby R, Mitchell M, Hartley A, Cornelissen J, Gurevitch J, GCTE-NEWS (2001) A meta-analysis of the response of soil respiration, net nitrogen mineralization, and aboveground plant growth to experimental ecosystem warming. Oecologia 126:543–562. https://doi.org/10.1007/s004420000544
Schimel JP, Bennett J (2004) Nitrogen mineralization: challenges of a changing paradigm. Ecology 85:591–602. https://doi.org/10.1890/03-8002
Stark JM, Hart SC (1996) Diffusion technique for preparing salt solutions, Kjeldahl digests, and persulfate digests for nitrogen-15 analysis. Soil Sci Soc Am J 60:1846–1855. https://doi.org/10.2136/sssaj1996.03615995006000060033x
Tiedje JM, Sorenson J, Chang YYL (1981) Assimilatory nitrate reduction: perspective and methodology for simultaneous measurements of several nitrogen cycle processes, terrestrial nitrogen cycles. Ecol Bull 33:331–342
Tripathi N, Singh RS (2012) Impact of savannization on nitrogen mineralization in an Indian tropical forest. For Res Open Access 1:2–11. https://doi.org/10.4172/2168-9776.1000108
Turrión M-B, Schneider K, Gallardo JF (2009) Carbon accumulation in Umbrisols under Quercus pyrenaica forests: effects of bedrock and annual precipitation. CATENA 79:1–8. https://doi.org/10.1016/j.catena.2009.04.004
Wang C, Chen Z, Unteregelsbacher S et al (2016) Climate change amplifies gross nitrogen turnover in montane grasslands of Central Europe in both summer and winter seasons. Glob Change Biol 22:2963–2978. https://doi.org/10.1111/gcb.13353
Wang Q, Wang W, He X, Zhang W, Song K, Han S (2015) Role and variation of the amount and composition of glomalin in soil properties in farmland and adjacent plantations with reference to a primary forest in north-eastern China. PLoS ONE 10:1–19. https://doi.org/10.1371/journal.pone.0139623
Wang Q, Wang WJ, Zhong ZL, Wang HM, Fu YJ (2020) Variation in glomalin in soil profiles and its association with climatic conditions, shelterbelt characteristics, and soil properties in poplar shelterbelts of Northeast China. J For Res 31(1):279–290. https://doi.org/10.1007/s11676-019-00909-w
White CS (1991) The role of monoterpenes in soil nitrogen cycling processes in ponderosa pine. Biogeochemistry 12:43–68. https://doi.org/10.1007/BF00002625
Yang K, Zhu J, Gu J, Xu S, Yu L, Wang Z (2018) Effects of continuous nitrogen addition on microbial properties and soil organic matter in a Larix gmelinii plantation in China. J For Res 29:85–92. https://doi.org/10.1007/s11676-017-0430-7
Zhang H, Zhang GL, Zhao YG, Zhao WJ, Qi ZP (2007) Chemical degradation of a ferralsol (Oxisol) under intensive rubber (Hevea brasiliensis) farming in tropical China. Soil Tillage Res 93:109–116. https://doi.org/10.1016/j.still.2006.03.013
Acknowledgements
We sincerely thank V. Sudheesh for help in nutrient measurements and the Cochin University of Science and Technology for logistics support. The funding for the present study was provided by the Department of Space, Government of India, India, under ISRO-GBP program.
Author information
Authors and Affiliations
Corresponding author
Additional information
Corresponding editor: Zhu Hong.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project funding: The funding for the present study was provided by the Department of Space, Government of India, India, under ISRO-GBP program.
The online version is available at http://www.springerlink.com.
Rights and permissions
About this article
Cite this article
Sharma, N., Kumar, S. Effects of the type of forest alteration on gross nitrogen mineralization in soils of southern India. J. For. Res. 32, 1689–1697 (2021). https://doi.org/10.1007/s11676-020-01225-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-020-01225-4