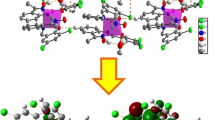
Abstract
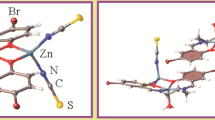
The structure of the ZnII complex with NN’O-donor ligand N-(2-pyridyl)methyl-2-hydroxy-3,5-di-tert-butylbenzaldimine (1H) [Zn(C21H27N2O)2]·3CH3OH is described. The Zn (II) metallic centre presents a five-coordinate environment with a square-pyramidal distorted geometry (τ-factor 0.14). One ligand acts as tridentate and another ligand as bidentate [Zn(1-κ3N,N’,O)(1-κ2N,O)]·3CH3OH. The three methanol solvent molecules interact with the zinc complex by hydrogen-bonds (H-bonds). This structure was modelled with Density Functional Theory (DFT) calculations and a good correlation between the experimental parameters and the calculated ones was obtained.
Graphic Abstract
The X-ray diffraction single crystal structure of the Zn(II) complex with NN’O-donor pyridino-imino-phenolate ligand (1H) [Zn(1)2]·3CH3OH is described and compared with the modelled by DFT calculations.




Similar content being viewed by others
References
Darensbourg DJ, Karroonnirun O (2010) Ring-opening polymerization of lactides catalyzed by natural amino-acid based zinc catalysts. Inorg Chem 49:2360–2371. https://doi.org/10.1021/ic902271x
Huang Y, Kou X, Duan Y-L, Ding F-F, Yin Y-F, Wang W, Yang Y (2018) Magnesium and zinc complexes bearing NNO-tridentate ketiminate ligands: synthesis, structures and catalysis in the ring-opening polymerization of lactides. Dalt Trans 47:8121–8133. https://doi.org/10.1039/C8DT00888D
Ebrahimi T, Mamleeva E, Yu I, Hatzikiriakos SG, Mehrkhodavandi P (2016) The role of nitrogen donors in zinc catalysts for lactide ring-opening polymerization. Inorg Chem 55:9445–9453. https://doi.org/10.1021/acs.inorgchem.6b01722
Cuesta-Aluja L, Campos-Carrasco A, Castilla J, Reguero M, Masdeu-Bultó AM, Aghmiz A (2016) Highly active and selective Zn(II)-NN′O Schiff base catalysts for the cycloaddition of CO2 to epoxides. J CO2 Util 14:10–22. https://doi.org/10.1016/j.jcou.2016.01.002
Shit S, Nandy M, Saha D, Zhang L, Schmitt W, Rizzoli C, Row TNG (2016) Synthesis, crystal structure and fluorescence properties of two dinuclear zinc(II) complexes incorporating tridentate (NNO) Schiff bases. J Coord Chem 69:2403–2414. https://doi.org/10.1080/00958972.2016.1197390
Sarwar M, Madalan AM, Tiseanu C, Novitchi G, Maxim C, Marinescu G, Luneau D, Andruh M (2013) A new synthetic route towards binuclear 3d–4f complexes, using non-compartmental ligands derived from o-vanillin. Syntheses, crystal structures, magnetic and luminescent properties. New J Chem 37:2280–2292. https://doi.org/10.1039/c3nj00199g
Zheng Z-P, Ou Y-J, Hong X-J, Wei L-M, Wan L-T, Zhou W-H, Zhan Q-G, Cai Y-P (2014) Anion-dependent assembly of four sensitized near-infrared luminescent heteronuclear Zn II –Yb III Schiff base complexes from a trinuclear Zn II complex. Inorg Chem 53:9625–9632. https://doi.org/10.1021/ic501118b
Mitchell JM, Finney NS (2001) New molybdenum catalysts for alkyl olefin epoxidation. Their implications for the mechanism of oxygen atom transfer. J Am Chem Soc 123:862–869. https://doi.org/10.1021/ja002697u
Bruker (2012) APEX2. Bruker AXS Inc., Madison
Bruker (2015) SAINT V8.37A. Bruker AXS Inc., Madison
Sheldrick GM (2015) Crystal structure refinement with SHELXL. Acta Crystallogr Sect C Struct Chem 71:3–8. https://doi.org/10.1107/S2053229614024218
Hübschle CB, Sheldrick GM, Dittrich B (2011) ShelXle: a Qt graphical user interface for SHELXL. J Appl Crystallogr 44:1281–1284. https://doi.org/10.1107/S0021889811043202
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Chesseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr., Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09. Inc, Wallingford
Orio M, Philouze C, Jarjayes O, Neese F, Thomas F (2010) Spin interaction in octahedral zinc complexes of mono- and diradical Schiff and Mannich bases. Inorg Chem 49:646–658. https://doi.org/10.1021/ic901846u
Gallaway JBL, Mcrae JRK, Decken A, Shaver MP (2012) Ring-opening polymerization of rac-lactide and ε-caprolactone using zinc and calcium salicylaldiminato complexes. Can J Chem 426:419–426. https://doi.org/10.1139/V2012-012
Sheng N (2009) Chlorido[2-methoxy-6-(2-pyridylmethyliminomethyl)phenolato]zinc(II). Acta Crystallogr Sect E Struct Reports Online 65:m1295–m1295. https://doi.org/10.1107/S1600536809037015
Trösch A, Vahrenkamp H (2004) Zinkkomplexe eines neuen N,N,O-liganden. Zeitschrift fur Anorg und Allg Chemie 630:2031–2034. https://doi.org/10.1002/zaac.200400249
Huang H-W (2011) (4-Chloro-2-{[(pyridin-2-ylmethyl)imino]methyl}phenolato)iodido(methanol)zinc(II). Acta Crystallogr Sect E Struct Reports Online 67:m313–m313. https://doi.org/10.1107/S160053681100417X
Maheswari PU, Barends S, Özalp-Yaman S, de Hoog P, Casellas H, Teat SJ, Massera C, Lutz M, Spek AL, van Wezel GP, Gamez P, Reedijk J (2007) Unique ligand-based oxidative DNA cleavage by zinc(II) complexes of hpyramol and hpyrimol. Chem A Eur J 13:5213–5222. https://doi.org/10.1002/chem.200601525
Wu GP, Ren WM, Luo Y, Li B, Zhang WZ, Lu XB (2012) Enhanced asymmetric induction for the copolymerization of CO2 and cyclohexene oxide with unsymmetric enantiopure SalenCo(III) complexes: synthesis of crystalline CO2-based polycarbonate. J Am Chem Soc 134:5682–5688. https://doi.org/10.1021/ja300667y
Zhang C, Wang ZX (2008) Aluminum and zinc complexes supported by functionalized phenolate ligands: synthesis, characterization and catalysis in the ring-opening polymerization of ε-caprolactone and rac-lactide. J Organomet Chem 693:3151–3158. https://doi.org/10.1016/j.jorganchem.2008.07.002
You Z-L (2005) catena -Poly[[[4-bromo-2-(2-pyridylmethyliminomethyl)phenolato]zinc(II)]-µ-chloro]. Acta Crystallogr Sect C Cryst Struct Commun 61:m456–m458. https://doi.org/10.1107/S010827010502932X
Addison AW, Rao TN, Reedijk J, van Rijn J, Verschoor GC (1984) Synthesis, structure, and spectroscopic properties. J Chem Soc Dalt Trans 251:1349–1356
Acknowledgements
The authors are thankful to Ministerio de Economia y Competitividad and AEI/FEDER UE (CTQ2016-75016-R and CTQ2017-83566-P), Departament d’Economia i Coneixement (2017 SGR 1472 and 2017 SGR 629) and Xarxa d’R + D + I en Química Computacional (XRQTC). Authors would like to thank the use of Rede de Infraestruturas de Apoio á Investigación e ao Desenvolvemento Tecnolóxico, Universidade de Santiago de Compostela (RIAIDT-USC) analytical facilities.
Funding
Generalitat de Catalunya (2017 SGR 1472, 2017 SGR 629 and Xarxa d'R+D+I en Química Computacional XRQTC), the Ministerio de Economia y competitividad (AEI/FEDER UE CTQ2016-75016-R and CTQ2017-83566-P) and the Ministerio de Asuntos Exteriores (Programa de Cooperación Internacional C030686-10).
Author information
Authors and Affiliations
Contributions
Not applicable.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Iksi, S., Guemmout, F.E., Reguero, M. et al. Crystal Structure and DFT Calculations of Zn(II)-NN’O Schiff Base Complex. J Chem Crystallogr 51, 432–437 (2021). https://doi.org/10.1007/s10870-020-00865-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-020-00865-y